Overview: This is the 6th paper to resolve the mystery of Homeopathy scientifically. In our previous works we have explained that each Homeopathic medicine is different from each other and every potency of a medicine is also different, in respect to the electrical energy content of water5. In those papers it is shown that whenever any substance is introduced into water, it produces a strain, due to which dipoles of water molecules are forced to rearrange themselves thus this water becomes different from any other water in respect of its electrical energy content4.
Abstract: Whenever two electrodes are placed in a beaker containing pure /distilled water it generates emf. Here we have applied some external emf to the electrodes in water and have got some remarkable changes in its generation of emf, after removal of the external source of emf. From our experiments it is obvious that there is a change in water from passing an external emf, as there is hardly any possibility of changes in electrode pairs on passing of external emf . By now it is crystal clear that electrical energy is solely responsible for causing changes in water, which is again the main criteria to differentiate various Homeopathic Medicines. In other words, water can hold electrical energy.
Experiment:1
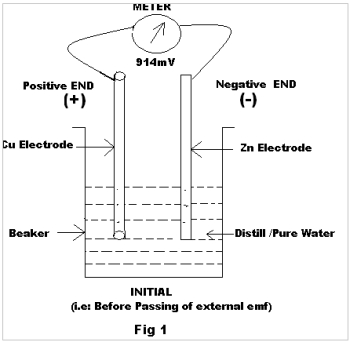
An experiment is performed with the following six (6) pairs of electrodes with varying combinations. The 1st pair of electrodes Cu & Zn was put in a beaker containing pure/ distilled water and its emf was recorded as “Initial” emf (Fig. 1).
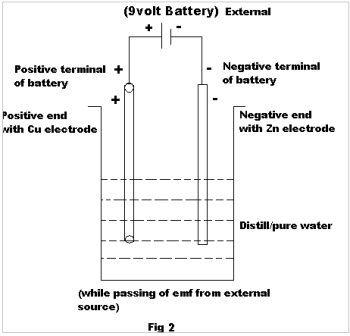
After recording of initial emf , external emf of about 9 volts was allowed to pass through it, such that the positive terminal of the external source(i.e 9 volt battery) is attached to the positive electrode of the 1st pair (i.e: Cu) & negative terminal to the negative electrode of the 1st pair (i.e: Zn) as shown in Fig-2
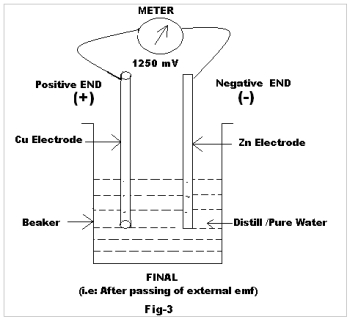
The external emf was allowed to pass for 60 seconds and thereafter disconnected. The Final reading (Fig-3) is recorded after 15 seconds.
A similar process is done for the rest of the five pairs and readings are recorded in the table below:
| Sl No. | Pairs of Electrode | Type of electrode | Initial ( i.e:Before passing External emf)(in mV) | Final (i.e: After passing External emf of 9 volts for 60 seconds through similar Poles)(in mV) | |
| Positive (+) | Negative (-) | ||||
| 1 | Cu | Zn | Metal-Metal (Different) | 914 | 1250 |
| 2 | Gr. | Zn | Non metal-Metal | 1203 | 2030 |
| 3 | Gr | Cu | Non metal-Metal | 150 | 1290 |
| 4 | Cu | Cu | Metal-Metal (Same) | 10 | 482 |
| 5 | Zn | Zn | Metal-Metal (Same) | 30 | 238 |
| 6 | Gr | Gr | Non metal-Non metal | 11 | 1580 |
Here, Cu = Copper (Metal), Zn = Zinc (Metal), Gr = Graphite (Non-metal) In the above six cases it is found that even pure/distilled water can generate a remarkable emf with different combinations/types of electrodes. The generation of such emf is therefore either due to chemical reactions or to the presence of ions in water (H3O+ & OH–). As there is hardly any chemical reaction with the stated pairs and water the generation of such emf may be attributed to the presence of H3O+ & OH– ions in water (as even the purest form of distilled water contains these ions). If the generation of emf is due to the presence of only H3O+ & OH– ions in water —
a) there should not be such extreme variations for the above six pairs (the variation of emf from 10 mV (in case of Cu-Cu) to 1203 mV( in case of Gr-Zn) is quite remarkable).
b) an electrode playing different roles for different combination pairs i.e; at Sl.no. 1. Copper (Cu) is behaving as Positive electrode with Zinc (Zn) whereas at Sl.no.3 the same metal Copper (Cu) is behaving as a Negative electrode with Graphite (Gr.).
c) Moreover it is found that when these three electrodes are dipped in distilled water simultaneously, Cu behaves as a positive electrode with Zn while as a negative electrode with Graphite.1
In all the six cases above it is found that the final emf is always greater than the initial emf, which implies that the polarity of the electrode pairs remains the same even after passing of an external emf.
For the 1st pair, Cu & Zn in experiment 1, it increases from 914 mV to 1250 mV. Similar results for the other 5 pairs shows that either
i. positive electrode becomes more positive & negative electrode becomes more negative
or ii. changes, in charge, occurred at any one of the electrodes.
To confirm the changes that occurred at electrode ends on passing of external emf, the following experiment is done. Herein three electrodes (Cu, Zn & Gr.) are placed in a beaker containing distilled water and their readings are recorded separately for each pair as in the table below. External emf of about 9 volts is passed through Cu & Zn for about 1 minute. After stopping of external emf , final emf’s of all three pairs are recorded separately after 15 seconds and noted in the table below:
| Sl No. | Pairs of Electrode | Type of electrode | Initial ( i.e:Before passing External emf)(in mV) | Final (i.e: After passing External emf of 9 volts for 60 seconds through Cu & Zn with similar polarity)(in mV) | |
| Positive (+) | Negative ( –) | ||||
| 1 | Cu | Zn | Metal-Metal (Different) | 840 | 1059 |
| 2 | Gr. | Zn | Non metal-Metal | 1147 | 1129 |
| 3 | Gr | Cu | Non metal-Metal | 227 | 190 |
The initial and final readings for the pairs Gr – Zn & Gr – Cu clearly shows that changes occurred at both the electrodes on passing of external emf through Cu & Zn in water.
So it can be inferred here that the positive electrode becomes more positive & the negative electrode becomes more negative on passing of an external emf through similar poles.
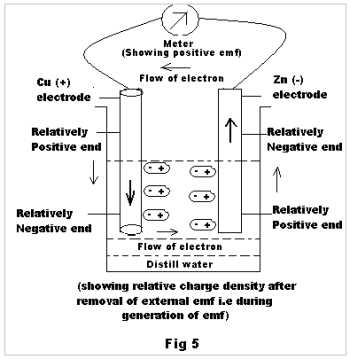
Now the question arises about what makes the positive electrode become more positive & the negative electrode more negative on allowing emf to pass through similar poles. For the 1st pair Cu was positive and Zn was negative during generation of emf , but the question is how the immersed part of the said electrodes behaves within water. Actually the immersed portion of the Cu electrode behaves as more electron dense than the Zn electrode, i.e reverse of outside or emf producing end as shown in Fig. 5. If this result was due to ionization H3O+ & OH– ions would have gone to the more electron dense end (Cu end) and the less electron dense end (Zn end) respectively and would have been neutralized there. Thus the generated emf (Final) would have approached Zero, which is not the case.
Experiment:2
Another experiment is performed with the following six (6) pairs of electrodes with varying combinations. The 1st pair of electrodes Cu & Zn was placed in a beaker contaning distilled water and its emf was recorded as “Initial” emf . After recording of initial emf , an external emf of about 9 volts was allowed to pass through it such that the negative terminal of the external source (i.e 9 volt battery) is attached to the positive electrode of the 1st pair (i.e: Cu) & positive terminal to the negative electrode of the 1st pair (i.e: Zn). The external emf was allowed to pass for 60 seconds and thereafter disconnected. The Final reading is recorded after 15 seconds. A similar process is carried out for the rest of the five pairs and readings are recorded in the table below:
| Sl No. | Pairs of Electrode | Type of electrode | Initial ( i.e:Before passing External emf)(in mV) | Final (i.e: After passing External emf of 9 volts for 60 seconds through opposite Poles)(in mV) | |
| Positive (+) | Negative (—) | ||||
| 1 | Cu | Zn | Metal-Metal (Different) | 850 | 518 |
| 2 | Gr. | Zn | Non metal-Metal | 1110 | 189 |
| 3 | Gr | Cu | Non metal-Metal | 259 | –864 |
| 4 | Cu | Cu | Metal-Metal (Same) | 06 | –585 |
| 5 | Zn | Zn | Metal-Metal (Same) | 10 | –278 |
| 6 | Gr | Gr | Non metal-Non metal | 09 | –1532 |
Here, Cu = Copper (Metal), Zn = Zinc (Metal), Gr = Graphite (Non-metal)
In all the six cases above it is found that the final emf is always less than the initial emf, which implies that the polarity of the electrode pairs get altered after passing of external emf.
For the 1st pair,Cu & Zn in experiment 2 it decreases from 850 mV to 518 mV. Similar results for other 5 pairs shows that
i) positive electrode becomes less positive & negative electrode becomes less negative
or ii) changes in charge occurred at any one of the electrodes.
It is already shown above that on passing of external emf through a pair of electrodes dipped in water, changes occur at both the electrodes in water. Now the question arises about what makes the positive electrode becomes less positive & the negative electrode less negative on allowing emf to pass through opposite poles. If this result was due to ionization H3O+ & OH–ions would have gone to the more electron dense end (Cu end) and less electron dense end (Zn end) respectively, and would have been neutralized there as discussed above. Thus the generated emf (Final) would have approached Zero, which is again not the case. And, it is always far from Zero, i.e; either Positive emf (For pairs 1&2) or Negative emf (for pairs 3,4,5 &6).
Explanation: In experiments1 & 2 wherein the same external emf’s were allowed to pass through a given pair of electrodes for the same time, through similar poles and dissimilar poles respectively, it is found that the final emf differs greatly, i.e increases in the former but decreases in the latter. Say for the 1st pair,Cu & Zn in experiment 1 it increases from 914 mV to 1250 mV whereas in exp. 2 for the same pair it decreases from 850 mV to 518 mV. The result is similar for the rest of the 5 pairs, so it is obvious that had the generation of the final emf been due to the ionization, the Final emf would have been the same irrespective of direction of passing of external emf. The generation of different emf’s for each of the pairs for the above two experiments ,after passing of external emf, suggests that there is a certain change in one of the following conditions as detailed below:
1st Condition: any changes within the electrodes itself. or
2nd Condition: any change within the water.
1st Condition: It is a well known fact that the electrodes being made up of metal/non-metal (graphite), are unable to generate any emf in the absence of water.
A simple experiment would however confirm the same. Two electrodes Cu & Zn were placed on the aluminium wire (instead of water), Initial emf was measured. Then external emf of about 9 volts was passed through Cu & Zn for 1 minute and thereafter disconnected. Final emf was measured. Both the initial and final emf were found to be Zero, thereby suggesting that there is certainly no change in electrodes on passing of external emf .
2nd Condition: As the 1st condition is ruled out, it is obvious that the 2nd condition is solely responsible for the change of emf on passing of external emf through a given pair of electrodes.
Explanation for the findings: From the above experiments it is clear that water not only produces emf with metal/non-metal electrodes immersed in it, but also can retain electrical energy when some external emf is passed through this electrodes i.e: water has both the properties of generation of emf and retention of electrical energy.
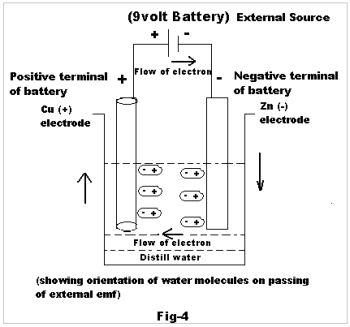
To explain this, let us consider the case when external emf is passed through two electrodes dipped in distilled water. It is well known to us that all polar molecules have positive & negative ends. Normally in the absence of an electric field, the molecules would be randomly oriented in all directions due to a thermal effect. But when such molecules are placed in an electric field between two charged plates , there will arise two distinct effects .Firstly, the usual distortion of the positive and negative charges would occur, giving rise to induced polarization; secondly, the field would tend to orient all the molecules, being polar and having permanent moment, in the direction of the field. Had the molecules been stationary, these would be oriented at 1800 to the direction of the field, the positive end pointing towards the negative plate. But owing to thermal agitation, which opposes such orientation, the molecules would occupy some mean position between the direction of the field and their original position, giving rise to orientation polarization (as shown in Fig-4).2,3
When the external emf is stopped, it is found that the electrodes through which the positive terminal of the external source of emf was joined, remains positive and the electrode through which the negative terminal of the external source of emf was joined remained negative. Had this result being solely due to ionization, at least some of the result would have been reversed. Say, for the pair Gr & Cu in expt.2 at sl no.3 the initial emf is found to be 259 mV, showing that here Gr is a positive electrode and Cu is a negative electrode. But after passing of external emf , the final emf for this pair is found to be –864 mV, which shows that here Cu is the positive electrode and Gr is the negative electrode. Here, a question arises regarding the change of polarity of the electrodes on passing of external emf. Actually, as soon as an external emf source is disconnected the orientation of dipoles of water molecules prefer polarity to the electrodes. The electrode through which the positive terminal of the external source of emf was connected, behaves as positive (as shown in Fig 5 ) as the portion dipped in water behaves as a relatively negative zone and the portion above water behaves as a relatively positive zone. Similarly other electrodes behave as negative electrode and emf is generated. From the above fact it is clear that even in absence of any external source of emf, water is able to generate some emf with same/different electrodes due to different orientation of dipoles of water molecules.
Inference: It therefore proves that water holds electrical energy through change of its orientation of dipoles.
References:
[1] Generation of electrode Potential of an electrode –an explanation , Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXIII.2010, www.hanp.net)
[2] Physical Chemistry, P C Rakshit ( Revised 4th Edition) 1980, Published by S C Rakshit, Kolkata , India
[3] Physical Chemistry, S Glasstone, 2nd Edition, 1946
[4]. The Homeopathic Dilution-A New Explanation, Ruhul Amin ,Biplab Chakraborty & Farook Rahaman (Simillimum,vol.XXI,Summer Fall/2008. www.hanp.net)
[5]. The electrical energy of substances –the secret of homeopathy, Ruhul Amin & Biplab Chakraborty(Simillimum,vol.XXII, Summer Fall/ 2009. www.hanp.net)


Dear Dr Ruhul Amin,
Very good experimental work, ” Water the magic liquid – retaining electrical energy”.
I like it, keep it up.
Prof Shaikh Shamsur Rahman,
Abu Dhabi,
UAE.
Dr Amin, I am quite certain you will be very interested in the research of Dr Shui Yin Lo, which led to the discovery of stable water clusters, or double helix water, as he has chosen to name it. Visit his websites at and http://doublehelixwater.com . This is cutting edge research and what I consider the discovery of the century…see if you agree!
DEAR DR,
YOU HAVE PROVED THAT REMEDY HOWEVER DILUTED CONTAIN THE POWER OF THAT REMEDY.THE POWER IN CREASES WITH EVERY SUCCESSION. SO THE REMEDY WHEN BECOMES MORE POWERFUL THEN THE POISON OF BACTERIA OR VIRUS IN THE BODY IS DESTROYED BY THAT REMEDY AND THE PATIENT IS CURED. IT HAS BEEN ALREADY PROVED THAT LIKE KILLS THE LIKE THAT IS LIKE CURES THE LIKE SO HOMEOPATHY IS TOTALLY SCIENTIFIC PATHY. TO FIND SIMILAR REMEDY MAY BE DIFFICULT BUT NOT IMPOSSIBLE. HOMEOPATH MAY FAIL BUT HOMEOPATHY NEVER FAILS
THANKS
DR SHEKHAR
Water is indeed a magic solution! I alwayz kept wondering how & why a little vial of water with pungent smell could cure for sure all our troubles until I get to know that you, two prodigal sons of Almighty,have already decoded the one great mystery of Nature. Pray 4 U, keep growing.