Failure of either contractility or deglutitive inhibition will result in failure of bolus progression and will be felt by the patient as Dysphagia. Excessive peristaltic contraction of the esophagus (nutcracker esophagus) and diffuse esophageal spasm are usually associated with pain, although Dysphagia may be a feature. More commonly Dysphagia occurs because of uncoordinated or hypotensive esophageal contraction. The most well defined example of such abnormalities would be achalasia of the cardia.
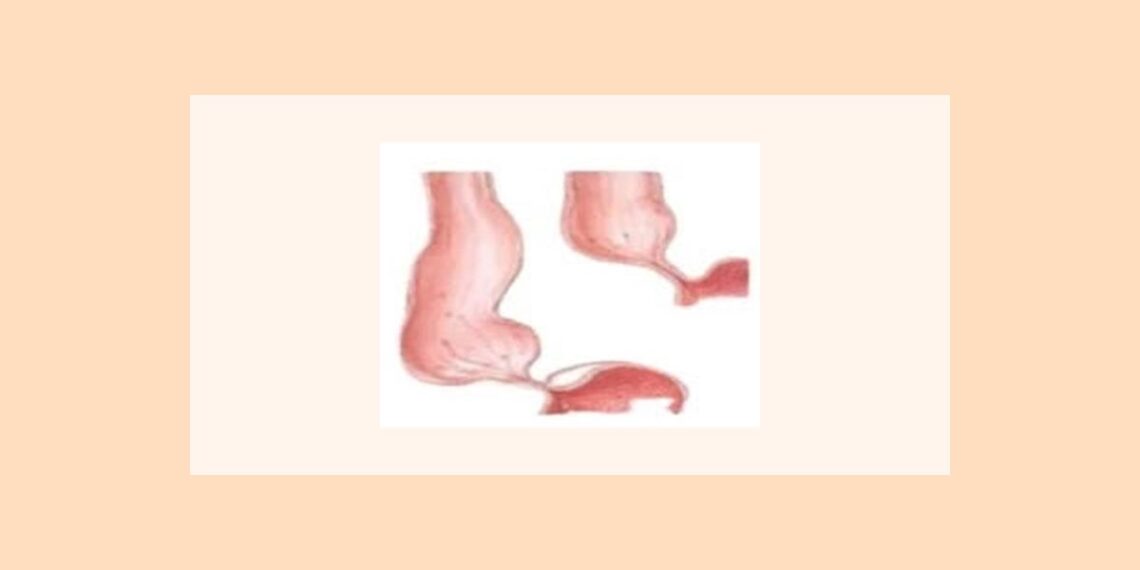
Achalasia, literally ‘failure to relax’, is characterized by uncoordinated, non-progressive contraction within the esophageal body. In addition the LOS fails to relax on swallowing, and is usually hypertensive at rest.
Achalasia presents with Dysphagia as the primary symptom. As the disease progresses the esophagus dilates proximal to the obstructing LOS, and can harbor large volumes of food, which may be regurgitated or even aspirated if it doesn’t pass distally. Patients often lose weight as the symptoms progress. Dysphagia in Achalasia is variable and can occur to solids and liquids. Patients may be able to assist esophageal emptying by employing manoeuvres such as straightening up or lifting their arms.
The cause of idiopathic Achalasia is unknown, but Chagas disease, which is a trypanosomal infection most prevalent in South America, closely mimics the esophageal changes of Achalasia. Achalasia may also be mimicked by tumor infiltration around the gastric cardia. It is important therefore to obtain an endoscopic evaluation in all cases and to maintain a high index of suspicion in older patients presenting with apparent Achalasia.
Oesophageal manometry is the cornerstone for the diagnosis of Achalasia. The pathognomonic features are failure of LOS relaxation and disordered or absent esophageal contraction, particularly in the distal esophageal body.
The finding of resting LOS hypertension is a common additional feature. Early in the evolution of the disease, some patients may exhibit strong esophageal contractions on swallowing, although the contractions will yield an isobaric waveform with non- progression. This is known as vigorous Achalasia.
Also Read: Best Homeopathic Remedies for Acid Reflux
Etiology of achalasia cardia
There is derangement of the neuromuscular mechanism responsible for the normal working of the cardiac sphincter which fails to relax when food reaches it. Degeneration of the ganglionic cells of the nerve plexus of the esophageal wall or the vagus nerve (or its nuclei), or an abnormality in the post ganglionic neuromuscular transmission are some of the possible causes.
The fact that the trouble often starts or is reactivated during or after mental stress (e.g. fear or mental shock) has led to the belief that it is a psychosomatic disorder. Rarely, achalasia occurs secondary to Chagas disease, gastric carcinoma infiltrating into the esophagus, lymphoma, irradiation, or due to certain toxins, drugs or infection due to neurotropic viruses.
The condition is more common in females and majority of cases occur between 25 to 35 years of age.
Pathogenesis of achalasia cardia
The disease is thought to be due to a neuromuscular disorder, possibly degeneration of the myenteric plexus (Auerbach plexus). Psychogenic or hormonal factors are also possible.
Clinical features of Achalasia Cardia
Dysphagia is the commonest presenting symptom in patients with achalasia cardia, and occurs in more than 95% of patients. In general, the Dysphagia due to motor disorders of the esophagus occurs with solids as well as liquids. However, most patients with Achalasia complain predominantly, if not exclusively, of Dysphagia to solid food. The converse, Dysphagia for liquids only, almost never occurs.
Patients often localize the Dysphagia to the region of the LES. The natural history of Dysphagia varies. Some patients notice that the Dysphagia reaches to certain degree of severity, and then stops progressing. In others, the Dysphagia continues to worsen to the extent that it results in markedly reduced oral intake, malnutrition and inanition. Emotional stress and the ingestion of cold liquids are well known exacerbating and precipitating factors. The duration of Dysphagia averages two years at presentation.
Eighty to ninety percent of patients with Achalasia experience spontaneous regurgitation of food from the esophagus. The regurgitation of food from the esophagus, the regurgitation is because of progressive stasis of food that the patient has eaten hours, or even days, previously. Some patients learn to induce regurgitation to relieve the retrosternal discomfort related to the distended esophagus. As the disease progresses, the likelihood that aspiration will occur increase. Patients may present with serious pulmonary complication like pneumonia, lung abscesses, bronchiectasis and hemoptysis.
Chest pain is present in two thirds of patients. Typically, patients describe chest pain as being retrosternal. This is a more common feature in patients with early or so called vigorous achalasia, which is characterized by prominent contractions in the body of the esophagus, documented by radiography or manometry. These contractions are simultaneous and hence esophageal musculature gets fatigued, chest pain tends to abate or disappear.
The chronic obstruction at the level of the LES may occasionally result in the development of an esophageal diverticulum in the distal esophagus. Identification of this is important as its presence increases the risk of esophageal perforation during pneumatic dilation to treat the achalasia.
It is important to rule out secondary cause of achalasia (secondary achalasia, pseudoachalasia), such as gastroesophegeal cancer and paraneoplastic syndromes, prior to making a diagnosis of idiopathic achalasia. It is better to rule out with endoscopy with judicious use of CT scan and endosonography.
If a patient with achalasia presents with significant loss of weight over a short period, esophageal cancer should be ruled out. When cancer develops, it is usually squamous, and arises in the dilated middle part of esophagus, rendering it relatively silent until late. Heartburn, and response to antacids, is indistinguishable from that seen in patients with gastroesophegeal reflux disease (GERD). In fact, some patients with achalasia are mistakenly diagnosed with GERD for many years. Other rare symptoms of achalasia include airway compromise and stridor because of compression of the trachea by the dilated esophagus.
Differential Diagnosis of achalasia cardia
The differential diagnosis of achalasia begins with the broad differential diagnosis of Dysphagia and exclusion of mechanical causes of Dysphagia. Strictures, benign neoplasm, vascular rings, webs, foreign bodies, and severe oesophagitis (peptic, infectious, chemical, drug induced) are among the frequently encountered entities.
Achalasia must be distinguished from other motility disorders such as diffuse esophageal spasm and scleroderma that has been complicated by a peptic stricture. The next step is to determine whether it is due to an identifiable cause. These cases, termed secondary achalasia, may be due to infection with T cruzi (Chagas disease) or neural degeneration as a paraneoplastic syndrome associated with many malignancies.
Gastroesophegeal junction carcinoma may produce myenteric plexus degeneration by local tumor invasion, resulting in achalasia. The term pseudoachalasia is used when there is no functional cause of achalasia but the gastroesophegeal junction is narrowed due to outside compression or fibrosis and the esophageal body is dilated due to obstruction.
Patients with secondary achalasia due to tumor at the gastroesophegeal junction present with more recent and progressive onset of Dysphagia or with progressive weight loss. Careful retroflexed inspection of the cardia and gastroesophegeal junction by upper endoscopy is necessary.
Laboratory Findings for achalasia cardia
Some patients with idiopathic achalasia have increased anti-neuronal antibodies, including anti Hu-1 anti-neuronal antibodies. Enzyme linked immuno-sorbent assay (ELISA) test, agglutination tests, and confirmatory assays, including immuno-fluorescence, immuno blot, Western blot, and radio-immuno-precipitation tests, may aid in identifying T cruzi in patients with achalasia caused by Chagas disease.
Imaging Studies for achalasia cardia
Barium esophagogram, the preferred initial method of evaluation in patients with Dysphagia, may reveal characteristically smooth, symmetric narrowing or “bird-beaking” of the distal esophagus, and often a dilated esophagus with no peristaltic activity and poor esophageal emptying.
In severe achalasia, chest radiographs may reveal a dilated esophagus containing food, possibly air fluid level within the esophagus in the upright position, absence of gastric air bubble, and sometimes a tubular mediastinal mass beside the aorta.
Administration of a smooth muscle relaxant such as sublingual nitroglycerin or inhaled amyl nitrate may cause relaxation of the LES and distinguish achalasia from pseudoachalasia arising from mechanical causes. Severe cases may reveal a markedly dilated and tortuous esophagus, called a sigmoid esophagus.
Endoscopy
Esophagogastroduodenoscopy is not sensitive in the diagnosis of esophageal motility disorders. However, it is very useful in excluding mechanical disorders, particularly those that may be the cause of the motility disorder (e.g., infiltration by gastroesophegeal) junction cancer, causing secondary achalasia).
Manometry
The diagnosis is confirmed by esophageal manometry, which typically reveals complete absence of peristalsis, incomplete LES relaxation (<50% of baseline pressure), and often but not necessarily increased lower esophageal basal tone (>30 mm Hg). Weak contraction s may be noted in the esophageal body, which are simultaneous or appear simultaneous but identical if the esophageal body becomes a single lumen (common cavity effect). Esophageal pressure may also exceed gastric pressures when the esophagus is filled with food or fluid.
Complications of achalasia cardia
These include an increased risk of esophageal carcinoma, and aspiration can cause pneumonia.
Treatment of achalasia cardia
The aim of treatment is to relieve the functional obstruction at the cardia. The two main methods are forceful dilation of the cardia and Heller’s myotomy.
Pneumatic forceful dilatation involves stretching the cardia with a balloon to disrupt the muscle and render it less competent. The results have, however, been variable. Perforation is the major complication and has been reported in about 3 to 5% cases. The risk of perforation increases with the bigger balloons and they should be used cautiously for progressive dilatation over a period of weeks.
Heller’s myotomy involves cutting the muscles of the lower esophagus and cardia. Heller’s myotomy may be performed through an abdominal route or trans-thoracically. The left thoracotomy is the preferred approach. After the lower end of the esophagus is exposed and carefully mobilized to preserve the vagal nerve trunks. The muscle coat of the distal esophagus is divided longitudinally down to the esophageal mucosa. This incision extends 5 cm proximally and 2 cm distally to the oesopahgogastric junction. The incision is deepened up to the submucous coat only taking care not to damage the mucosa which bulges out throughout the gap. Carefully measured myotomy does not increase the incidence of gastro-esophageal reflux. In case there is a possibility that the lower sphincter has got destroyed and antireflux procedure may need to be added. It is desirable to perform a partial rather than a total fundoplication in this situation because of the risk of causing Dysphagia in the presence of an aperistaltic esophagus.
Heller’s myotomy is now being performed by a minimal access approach by either thoracoscopy or laparoscopy. It is a pain free procedure and requires a short hospital stay.
Botulinum toxin is given by endoscopic injection into the cardio esophageal sphincter. It is a new form of treatment whose place is not yet established. It acts rapidly and strongly binds to presynaptic cholinergic nerve terminals and produces paralysis within a few hours. Transmission of neuromuscular impulses resumes after the growth of new axon terminals and clinical weakening of muscles is seen for several months.
Drugs, such as calcium channel antagonists may decrease pressure in the lower sphincter and may improve swallowing but its long term efficacy is doubtful. It may be useful for transient relief of symptoms.
Homeopathic treatment of achalasia cardia – Homeopathy is one of the most popular holistic systems of medicine. The selection of remedy is based upon the theory of individualization and symptoms similarity by using holistic approach. This is the only way through which a state of complete health can be regained by removing all the sign and symptoms from which the patient is suffering. The aim of homeopathy is not only to treat achalasia cardia but to address its underlying cause and individual susceptibility. As far as therapeutic medication is concerned, several remedies are available to cure achalasia cardia symptoms that can be selected on the basis of cause, sensations and modalities of the complaints. For individualized remedy selection and treatment, the patient should consult a qualified homeopathic doctor in person. There are following remedies which are helpful in the treatment of achalasia cardia:
Phosphorous, Arsenic, Nitric Acid, Digitalis, Sulphur, Sulphuric Acid, Natrum Mur, Hydrastis, Chamomilla, Drosera, Aesculus, Hyoscyamus, Ferrum Met, Podophyllum, and many other medicines.
References:
Graeme Duthie, Angela Gardiner: Physiology of the Gastrointestinal Tract; 2004; 26
Prasad: Principles and Practice of Medicine a TB for students and Practitioners; 346
Pathogenesis; 2009; 371
Tandon: Tropical Hepato- Gastroenterology; 2007; 83
Norton J. Greenberger, Richard Steven Blumb: Current Diagnosis and Treatment in Gastroenterology, Hepatology, and Endoscopy; 2009; 167
John S. Axford, C. A. O’Callaghan; Medicine; 2004; 701
Gupta: Textbook of Surgery; 2003; 687


I have suffered from achalasia for 35 years which initially was diagnosed as GERD and mis-treated with PPI’s and H2 blockers which made everything worse. I have recently had further manometry tests which have revealved the latest diagnosis and have been offered the Botox treatment etc. or ultimately the surgical Myotomy.
I am Homoeopath and Nutritional Therapist and think I’ve been put on this planet to discover every therapy that is going , and thankfully been able to help many many people over 25 years of practice. However…I am despairing of my own health at this minute …just about to take some Phosphorous 200C as I know that it is a constitutional of mine , yet which , for some reason I have avoided taking??? Any recommendations would be gladly taken on board.
Thanks
Hi rosalin,
Greetings from Perth, Australia. I would love to get in touch with you. My daughter 3 years old is exhibiting similar symptoms. I’m a desperate father !
Thanks
Pusker
Rosalind, are you able to advise as to the success of Phosporous 200C.