A group of tropical disease caused by a range of parasitic roundworms and larvae that transmit disease to humans. About 200 million people are affected by filariasis, which occurs in tropic and subtropics areas of Southeast Asia, South America, Africa, and the Pacific, Mosquitoes inject the worm larvae when they bite, which migrate to the lymph nodes where they develop into mature worms in about a year.
Some of the species live in the lymphatic vessels and block them, causing ELEPHANTIASIS (swelling of limbs with thickened, coarse skin). Another type of worm can be seen and felt just underneath the skin, which produces irritating and painful swellings called calabar swellings.
Bancroftian filariasis (Wuchereria bancrofti filarial nematode) is spread widely throughout Africa, southern and southeastern Asia, the Pacific, and the tropical and subtropical regions of South America.
Malayan filariasis (Brugia malayi filarial nematode) is found only in southern and southeastern Asia. Imported States, especially among immigrants from the Caribbean and Pacific islands.
Bancroftian and Malayan filariasis are transmitted to humans by the bite of an infected mosquito. The infective larvae that are transmitted into humans via the bite pass into the human lymph system, where they develop to maturity during a six to 12 month period. Fertilized mature female worms release embryos that develop into moving larvae (microfilariae). Which appear in the human blood system at night only
Causes of filariasis
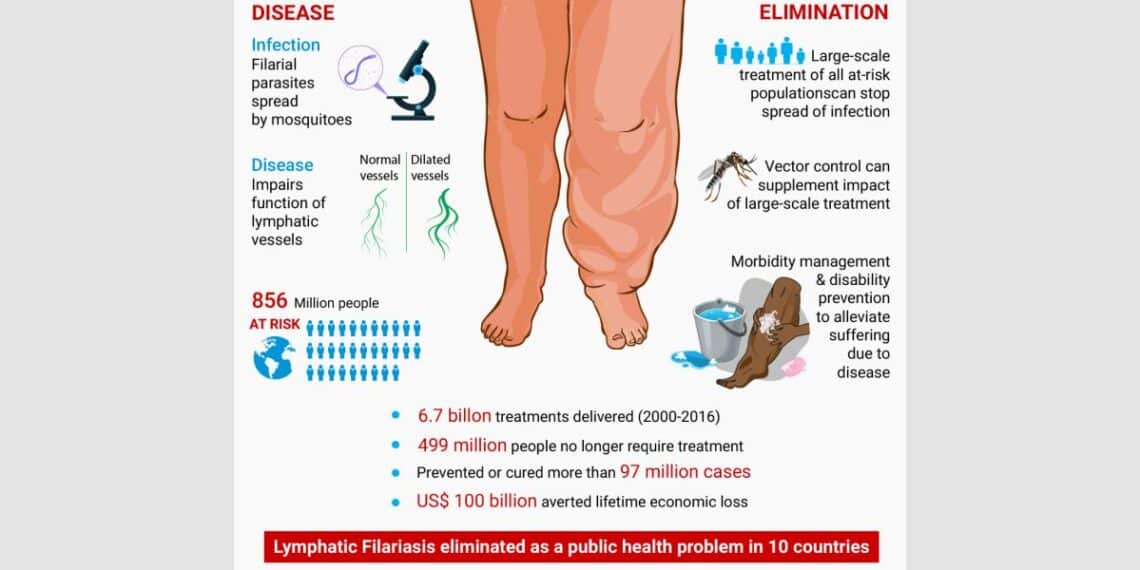
Lymphatic filariasis is caused by threadlike worms (nematodes) that live in the lymphatic system; it is transmitted by the bite of a mosquito carrying infective larvae. The larvae develop into adult worms in the lymphatic vessels, causing severe damage and swelling (lymphodema).
The most common filarial species is Wuchereria bancrofti, but Brugia malayi and Brugia timori cause the infection in Asia. It takes several mosquito bites over many months to cause the infection. People living in tropical and subtropical settings are at the greatest risk for infection.
Cutaneous filariasis caused by Loa loa is transmitted by the mango fly or deer fly; onchocerca transmits microfilariae via the black fly; and Manosonella streptocerca transmits infection via a midge. Dirofilaria immitis may cause lesions in the lung periphery. Depending on the species, incubation may be 3 to 12 months.
Symptoms of filariasis
Lymphatic filariasis is otherwise called Elephantiasis (Homeopathy Treatment for Elephantiasis). Here the patient presents with swollen genitals and a swollen scrotum. Patients have difficulty in mobility and prefer to sit on their enlarged scrotum.
In addition, the backflow of lymphatic fluid causes the legs to enlarge and the leakage of the lymphatic fluid causes the verrucous and thick appearance of the skin mimicking elephant legs.
Other symptoms are testicular and inguinal pain, slight fever, exfoliation of the affected skin and inguinal lymphadeonpathies. In subcutaneous filariasis, the symptoms are limited to the skin and the eye. The skin manifests with erythematous, pruritis, edematous, with scab like eruptions.
The skin thickness, described as hanging groin and leopard skin. The eye manifestations are corneal fibrosis, keratitis, choroiditis, glaucoma, iridocyclitis and optic atrophy.
All of these eye findings can cause blindness. Patients with serous cavity filariasis are usually asymptomatic. If ever they have symptoms, they are mild fever, pruritus and mild abdominal pain.
Diagnosis of filariasis
The adult worms residing in the lymphatics are inaccessible, so diagnosis is based on finding microfilariae in the peripheral blood. The level of filaria is inversely related to the clinical signs because much of the damage is due to immunological responses to the microfilariae rather than to the organisms themselves.
Furthermore the presence of microfilariae does not necessarily mean that they are causing clinical problems and conversely a lack of microfilariae in the blood does not exclude a diagnosis of filariasis. The peripheral blood finding must therefore be assessed in the context of the clinical picture.
To optimize the chances of findings scanty microfilariae in the blood, the sample should be taken at the appropriate time for the expected peak concentration of microfilariae (i.e. around midnight or midday for nocturnally and diurnally periodic forms respectively).
There are many techniques for demonstrating microfilariae in the laboratory. The simplest method is a wet preparation of fresh blood. Microfilariae will survive in venous blood collected into EDTA for 2 days at room temperature. Motile microfilariae can be seen on a slide under low power and can be counted in a counting chamber. Numbers of microfilariae may be low, so concentration techniques are often required.
Sensitivity can be increased by passing the whole blood sample through a 3 µm Nuclepore filter membrane and then stating the microfilariae trapped in the filter.
For species identification, thick and thin blood films should be stained with Giemsa or haematoxylin and the microfilariae differential according to the pattern of their sheaths, nuclei distributions and size. The edges of the film should be examined carefully as microfilariae tend to be concentrated at the periphery and are easily missed if the microscopist goes straight onto high power in the center of film.
Detection of circulating antigen by enzyme linked immunosorbent assay (ELISA) or ICT has replaced microscopy for the diagnosis of bancroftian, but not brugian, filariasis.
An antigen immunochromatography card test is available for the detection of W. bancrofti, which does not react with other filariae and is highly sensitive (100%) and specific (92%).
Filarial DNA can be detected by PCR, and ultrasound can help to identify adult worms within the lymphatic system. Serological tests are not very helpful in the diagnosis as most individuals from endemic areas have antibodies to crude filarial antigens and there is cross reactivity with other filariae and nematodes.
Complications of filariasis
Filarial infection can cause personal and socioeconomic hardships due to its debilitating consequences. Meningoencephalopathy and localized necrotizing granulomas are severe complications of L. loa infection. D. immitis may cause pulmonary infarction if the larvae lodge in the branches of the pulmonary arteries. Lymphatic filariasis may result in Hydroceles or elephantiasis of the extremities or genitals. A secondary bacterial infection may cause elephantiasis, blindness, and infection of the skin and lymphatic system
Treatment of filariasis
Individual chemotherapy
Diethylcarbamazine remains the treatment of choice. Adequate dosage will kill adult worms. Even a small single dose will clear blood microfilariae temporarily. Sensitivity reactions to filarial antigen, both local and systemic, are common in infected people and simulate some of the acute manifestation of the infection; they necessitate care and supervision in the initial stages, especially in Brugia infections.
Treatment should be started at 1 mg/kg on the first day, increasing over 3 or more days to 6 mg/kg in divided doses; this dose then being continued for 21 days. Coinfection with Loa loa and Onchocerca volvulas must be excluded before diethylcarbamazine is given to avoid dangerous reactions.
Indication for curative treatment is acute manifestation with or without microfilariaemia, and chronic disease in patients who are either microfilaria positive or positive for filarial antigen identified serologically. Treatment often reduces the size of Hydroceles but has little effect on chronic lymph edema.
Surgical and supportive treatment
Acute manifestation of filariasis can mimic strangulated hernia and testicular torsion. Surgical treatment of filarial Hydrocele is the same as that for non-filarial disease.
Scrotal lymphodema can be treated surgically, usually with preservation of the testes. Lymphosaphenous anastomosis is being used for leg elephantiasis; may other procedures have been used in the past, often with disappointing results.
Bacterial infection is common in those with lymphodema, especially when the skin is fissured, breached in an inter-digital cleft, or when there is minor injury, ulcer, or insect bite. Early use of antibiotics and resting of the affected limb lessens the risk of increasing lymphodema; supportive bandaging applied each morning or wearing elastic stocking reduces chronic oedema.
Vector control
These campaigns are targeted at the local vector. Larval Aedes breeding sites such a discarded tins, tyres, or coconut shells can be removed.
Culex numbers can be reduced by improved sanitation, larvicides, and polystyrene beads applied to the water surface of latrines and cesspits. Bed nets and repellants are universally applicable. Where Anopheles is the vector, malaria control can interrupt filariasis transmission as in Samoa, Vanuatu, and part of southern China.
Homeopathic treatment of filariasis symptoms– Homeopathy is one of the most popular holistic systems of medicine. The selection of remedy is based upon the theory of individualization and symptoms similarity by using holistic approach. This is the only way through which a state of complete health can be regained by removing all the sign and symptoms from which the patient is suffering. The aim of homeopathy is not only to treat filariasis but to address its underlying cause and individual susceptibility. As far as therapeutic medication is concerned, several remedies are available to cure filariasis symptoms that can be selected on the basis of cause, sensations and modalities of the complaints. For individualized remedy selection and treatment, the patient should consult a qualified homeopathic doctor in person. There are following remedies which are helpful in the treatment of filariasis symptoms:
Apis Mel, Clematis E, Silicea, Natrum Mur, Thuja Occidentalis, Lycopodium, Sulphur, Cannabis Sativa, Aurum Met, Hydrastis and many other medicines.
Reference
Carol Turkington, Bonnie Ashby- The encyclopedia of infectious disease; 2007; 114
Lippincott Williams & Wilkins, Lippincott- Lippincott’s Guide to infectious Diseases; 2010; 126
David Arieti MS- Prognosis Disaster: The Environment, Climate Change, Human Influences…; 2011; 319
Hoffbrand- Postgraduate Haematology; 2008; 987
D. A. Warrell, Timothy M. Cox, John D. Firth- Oxford textbook of medicine, Volume 1; 2003; 795


Key remedies found successful in Filariasis in the order of importance:
1. Mercurius sulphuricus
2. Apis mellifica
3. Hydrocotyle asiatica
4. Bryonia
What is the doses of prescribed filarial homoeopathy drug
1. Mercurius sulphuricus
2. Apis mellifica
3. Hydrocotyle asiatica
4. Bryonia
Arsenic alba
Surabhi sharma hospital address for filarial treatment
I am suffering for filarisis from last 7 years.I treat a lot, but there is no use for.
Please send me,accurate address of Hospital that can cure filarisis.
i have seen best result with Arsenic alba with all 4 above medicines to cure filaria