Anton von Storck an Austrian physician ( 1731- 1803) was convinced that plants regarded as poisonous still had medicinal applications if employed in carefully controlled quantities. His experiments with the plants such as hemlock, henbane etc. involved a three-step process. Initially it was used on animals, followed by a personal trial, and finally given to his patients, following a “sliding-scale” approach to determine the optimum dosage. Hahnemann got interested in the works of Storck for his research on various herbs, including their toxicity and medicinal properties.Following the innovative works of Anton von Storck, Hahnemann tested substances for the effects they produced on a healthy individual, presupposing that they may heal the same ills that they caused. His researches led him to agree with von Störck that the toxic effects of ingested substances are often broadly parallel to certain disease states. He later devised methods of diluting the drugs he was testing in order to lessen their toxic effects. He claimed that these dilutions, when prepared according to his technique of ‘potentization’ using dilution and succussion (vigorous shaking), were still effective in alleviating the same symptoms in the sick. His more systematic experiments with dose reduction really commenced around 1800-1801.
Homeopathic remedies were challenged only after the discovery of Avogadro’s number, a number of years after homeopathy came into use. It is true that homeopathic dilutions above the 12c potency are devoid of any medicinal molecules as shown in Table 1 below:
The mother tincture/solution is prepared by dissolving 1gm of NaCl in 9 ml of distilled water( i.e.1:9 w/v). For preparation of a 1C potency 1 ml of mother tincture is added to 9ml of distilled water and then succussed. The 2c potency is prepared by adding 1ml of 1C potency to 99ml of distilled water and succussing. Thereafter, following similar steps further serial dilutions are prepared. The calculations are made considering the density of water 1gm/cc and molecular mass of NaCl=23+35.45=58.45]
Table1
| Type of dilution | Solute/Foreign content(in ml) | Solvent i.e. Distilled water (in ml) | Total Solution/content(in ml) | Molecules of NaClpresent | Molecules of H2Opresent | Number of H2O molecules having altered molecular orientations, added to distilled H2O |
| MotherSolution (M.S) | 1gmNaCl | 9ml | 9ml | 1.0304X1022 | 3.0115X1023 | Nil |
| 1C | 1ml of MS | 9ml | 10ml | 1.1449X1021 | 3.3461X1023 | 3.3461 X1022 of MS |
| 2C | 1ml of 1C | 99ml | 100ml | 1.1449X1020 | 3.3461X1024 | 3.3461 X1022 of 1C |
| 3C | 1ml of 2C | 99ml | 100ml | 1.1449X1018 | 3.3461X1024 | 3.3461 X1022 of 2C |
| 4C | 1ml of 3C | 99ml | 100ml | 1.1449X1016 | 3.3461X1024 | 3.3461 X1022 of 3C |
| 5C | 1ml of 4C | 99ml | 100ml | 1.1449X1014 | 3.3461X1024 | 3.3461 X1022 of 4C |
| 6C | 1ml of 5C | 99ml | 100ml | 1.1449X1012 | 3.3461X1024 | 3.3461 X1022 of 5C |
| 7C | 1ml of 6C | 99ml | 100ml | 1.1449X1010 | 3.3461X1024 | 3.3461 X1022 of 6C |
| 8C | 1ml of 7C | 99ml | 100ml | 1.1449X1008 | 3.3461X1024 | 3.3461 X1022 of 7C |
| 9C | 1ml of 8C | 99ml | 100ml | 1.1449X1006 | 3.3461X1024 | 3.3461 X1022 of 8C |
| 10C | 1ml of 9C | 99ml | 100ml | 1.1449X1004 | 3.3461X1024 | 3.3461 X1022 of 9C |
| 11C | 1ml of 10C | 99ml | 100ml | 1.1449X1002 | 3.3461X1024 | 3.3461 X1022 of 10C |
| 12C | 1ml of 11C | 99ml | 100ml | 1.1449X100 | 3.3461X1024 | 3.3461 X1022 of 11C |
| 13C | 1ml of 12C | 99ml | 100ml | Nil | 3.3461X1024 | 3.3461 X1022 of 12C |
| ***** | ||||||
| nC | 1ml of (n-1)C | 99ml | 100ml | Nil | 3.3461X1024 | 3.3461 X1022 of (n-1)C |
The working principle of homeopathic medicine which should have been embraced as a great discovery for mankind was instead called unscientific. In modern times researchers have tried to discover the force behind homeopathy. One such researcher was Jacques Benveniste, who was pilloried by the scientific community for his efforts.
With due regards to Avogadro’s concept in homeopathic dilution i.e. finding no molecules of the parent compound beyond 12c dilution, we started searching for peculiar properties of water. After going through different physical and chemical properties we started our work with the electrical properties of water. To our surprise we noticed that even two steel electrodes when touched to some water show some minute voltage. At first we thought it might be due to the presence of ions in the water. When we repeated our experiment with distilled water ( as it does not contains any other ions) we were surprised to find a similar result.
We tried experiments aimed at different aspects of water’s generation, conduction and retention of electrical energy (using distilled water) and below we briefly reproduce some of them:
[A] Generation of electrical energy in various cases:
According to the existing theory, the generation of emf by an electrochemical cell is either due to the
- chemical reactions i.e. at the cost of the electrodes
or
- Concentration cells wherein the electrodes on the two sides are the same but they are dipped into solutions differing in concentrations of the reversible ions, i.e. Electrolyte concentration cell
or the Electrode concentration cell or Amalgam Cells ,wherein the electrodes of different activities are dipped in the same solution of an electrolyte containing an ion of the electrode material. .
Case 1: Generation of electrical energyin an irreversible cell Zn | H2SO4 | Cu wherein Zn and Cu metals are dipped in Sulphuric acid thereby generating emf of about 1 volt. In this cell electricity is produced due to a chemical reaction at the cost of the zinc electrode.
Zn → Zn+++ 2e ( At cathode) [ Zinc goes into solution as ZnSO4 ]
2H+ + 2e → H2 ( At anode)
Cu & Zn electrodes when dipped in distilled water record an emf of about 850mV in distilled water and 500mV (approximately) in the human body3 as shown at sl no. 12 of Table: 2 below. As there is hardly any possibility for zinc to dissolve in distilled water or to go into the human body, the generation of such emf through chemical reaction undoubtedly indicates a contradiction with the existing theory.
Case 2: Generation of electrical energy in the Leclanche Cell, wherein the Zn plate is the –ve electrode and graphite is the +ve electrode, the electrolyte being a paste of MnO2, NH4Cl and ZnCl2.
Here, also an emf of about 1.5 volts is generated at the cost of the Zn electrode as shown by the following reactions:
At the –ve electrode :Zn+ 2MnO2+ H2O→ Zn++ +Mn2O3 + 2OH –
At the +ve electrode : 2MnO2 + H2O +2e → Mn2O3+ 2OH–
Subsequently, secondary chemical reactions occur which consume the OH– and Zn++ through NH4Cl as :
2NH4Cl+ 2OH– = 2NH3+2Cl– +2H2O
Zn++ + 2NH3+2Cl– = Zn(NH3) 2Cl2
This cell, made up of Carbon (Gr) & Zn electrodes, still generates sufficient emf when the electrolyte is replaced by distilled water and the human body3 as shown at sl no. 2 of Table: 2. In distilled water it shows 1150mV and in the human body about 800mV. Here, also as in case 1 there is no possibility of Zinc going into solution or into the human body, and thus clearly indicates the contradiction with the existing theory.
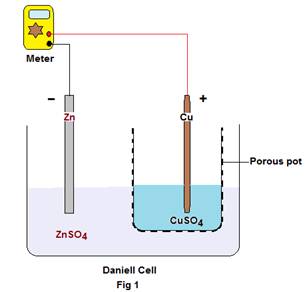
Case 3: Generation of electrical energy in the Daniel Cell wherein a zinc rod is partially immersed in ZnSO4 solution and a copper rod in CuSO4 solution as the two electrodes. The two solutions are separated by a porous barrier that allows the electrical current to pass through but prevents mass diffusion (Fig.1).
During the production of current, Zn atoms pass into solution as Zn++ ions leaving behind two electrons each in the metal electrode. On the other hand, copper ions from the solution are deposited on the copper electrode by accepting two electrons each from the metal. Thus there is an excess of electrons in the Zn electrode and a deficit of the same at the Cu electrode. When the circuit is closed, the electrons from Zn flow to the copper through the wire to feed the copper ions in the process of its discharge.
The cell being represented as: (-) Zn | ZnSO4 : CuSO4 | Cu (+)
The reaction at – ve electrode is: Zn= Zn++ + 2e (i.e: Loss of electrons or oxidation occurs)
The reaction at + ve electrode is :Cu++ + 2e= Cu (i.e: Gain of electrons or reduction occurs)
The total cell reaction being: Zn +Cu++ =Zn+++Cu
So for production of 1 Faraday of electricity, 1 gm-equivalent of Zn goes into solution and 1gm-equivalent of Cu is deposited on the Cu electrode. The emf of the cell is about 1.10volts. From the above it is clear that for generation of emf some amount of zinc goes into solution and copper ions are deposited on the copper electrode, i.e the Zn electrode becomes thin while the copper becomes thicker.
Now when Cu& Zn are placed in distilled water or the human body they are found to produce a remarkable emf of about 850 mV(0.850 volts) in distilled water and about 500mV on human body3, showing at least some amount of Zn must have gone into distilled water and some amount of copper ions from distilled water have got deposited on the copper electrode. But as per prevailing scientific norms, neither is it possible for zinc to get dissolved in distilled water nor distilled water to supply copper ions to be deposited on the copper electrode for the generation of emf. Here also lies a sound contradiction to the existing theory behind generation of electromotive force (emf).
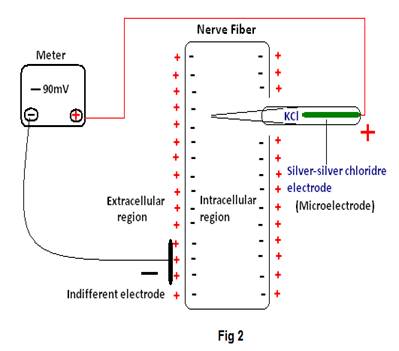
Case 4) The measurement of cell membrane potential using one micro electrode (Silver –silver chloride electrode) & another “indifferent electrode” (Fig2) seems interesting7.The emf
Recorded is -90mV. Here, the case is similar to the electrolyte concentration cell in chemistry where electrodes must be placed into solutions differing in the concentrations of the reversible ion to measure the generated emf.The general equation for the emf of any concentration cell without transference (i.e.no direct transfer of electrolyte from one solution to the other) can be obtained from the Nernst equation:
Where E=emf of the cell, R= Gas constant (8.3143 J), F(in coulombs) = 96500, ln=2.303log, a1 &a2 are activities of the reversible ion in the solution.
Moreover, is the total number and the number of positive or negative ions produced by the ionization of one molecule of electrolyte ,and Z+ or Z- is the valence of the ion with respect to which the extreme electrodes are reversible1.
From the above, it is seen that the Nernst equation can only be used to calculate the emf of a cell with electrodes reversible to one of the ions of the electrolyte. But, it is quite astonishing how potentials due to Na+ or K+ ions present in the electrolytes (extracellular & intracellular fluids) are calculated using the Nernst equation, by using one “indifferent electrode” & one micro electrode (Silver –silver chloride electrode) instead of using a sodium or potassium (amalgamated) electrode as stated in chemistry. In other words, as we are concerned with the presence of Na+ & K+ ions in the intracellular and extracellular region it seems quite contradictory, not using amalgamated electrodes as per existing theories in chemistry, and instead using such electrodes that do not contain any reversible ions.
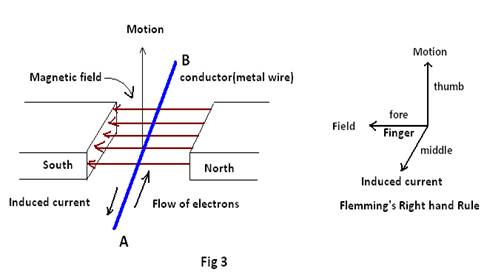
Case 5) The electromagnetic flow meter7in medical science is a well developed device that can be used to measure blood flow in a blood vessel without opening it.The principle for this instrument is in accordance with generation of electromotive force in a wire that is moved rapidly through a magnetic field as per Faraday’s law of electromagnetic induction.The movement of electrons in a metal conductor (due to relative motion between metal conductor and magnet) generates induced emf, hence induced current. But in the electromagnetic flow meter, the conductor is water, that when placed in a magnetic field is able to show induced current due to induced emf. What generates induced current in water? It is believed that such generation of emf is due to the presence of ions in water. This suggests that ions, like electrons are also deflected in the magnetic field, thereby generating induced emf and hence induced current. Therefore, in water ions seem to flow from A to B (Fig 3) in lieu of electrons, while generating emf in the presence of a magnetic field. Further it implies that the difference in concentration of ions at A & B generates emf by pushing ions through the water2. This shows that ions not only can move towards the opposite electrodes as in electrolysis, but also can move even to the neutral electrodes for generating emf , as in the electromagnetic flow meter!
Case6: The different/dissimilar pairs of electrodes are found to Generate electrical energy not only with distilled water but also in the human body3. The results are presented in Table 2 below:
Table2
| Sl No. | Electrode pairs | Observed emf (in mV)In Distilled water | Observed emf (in mV)In human body | |
| (A) | (B) | |||
| 1 | Gr – Mg (Graphite – Magnesium) | 1970 | 1180 | 1500 |
| 2 | Gr – Zn (Graphite – Zinc) | 1150 | 740 | 900 |
| 3 | Gr – Pb (Graphite – Lead) | 778 | 534 | 600 |
| 4 | Gr – Cu (Graphite – Copper) | 240 | 250 | 270 |
| 5 | Gr – Ag (Graphite – Silver) | 230 | 240 | 262 |
| 6 | Gr – Au (Graphite – Gold) | 101 | 210 | 220 |
| 7 | Au – Zn (Gold – Zinc) | 920 | 500 | 700 |
| 8 | Ag – Zn (Silver – Zinc) | 960 | 548 | 735 |
| 9 | Au – Ag (Gold – Silver) | 75 | 30 | 50 |
| 10 | Ag – Cu (Silver – Copper) | 25 | 44 | 85 |
| 11 | Au – Cu (Gold – Copper) | 65 | 77 | 90 |
| 12 | Cu – Zn (Copper – Zinc) | 850 | 490 | 596 |
To our astonishment not only two dissimilar electrodes but even two similar electrodes, including a non-metal pair (i.e. Graphite- Graphite) generated emf in distilled water4. (Table 3).
Table3
| Sl No. | Electrode pairs | Type of electrode | Emf in mV |
| 1 | Cu-Cu | Metal-Metal (Same) | 06 |
| 2 | Zn-Zn | Metal-Metal (Same) | 10 |
| 3 | Gr-Gr | Non metal-Non metal | 09 |
Our findings cannot be explained by any of the two stated theories:
- none of the electrodes used have any chemical reaction with water i.e. electrodes cannot be used up or consumed during generation of emf as required under existing theory.
- Moreover, as the electrodes are placed in distilled water, there is no possibility of the second condition, i.e. formation of concentration cells that involves electrodes dipped in solutions containing reversible ion (i.e. solution containing ions of the electrode material).
Thus it gives clear indication that distilled water is able to generate emf with the same or different electrode pairs in which does not fit with the stated theories in science. Here lies the contradiction in theory of generation of emf in distilled water. It therefore shows that there is some property of water that makes it able to generate emf beyond the stated theories.
We explained the same as being the result of repulsion of electrons of distilled water (lone pair & bond pair) with the free electrons in case of metal and pi ( ) electrons in case of non-metal Graphite electrodes through orientations of water molecules5 .
[B] Conduction of electrical energy in distilled water/water
It is well known that ions present in water/ distilled water are responsible for conduction of electrical energy .We conducted the following experiments to see how electrical energy is conducted in water.
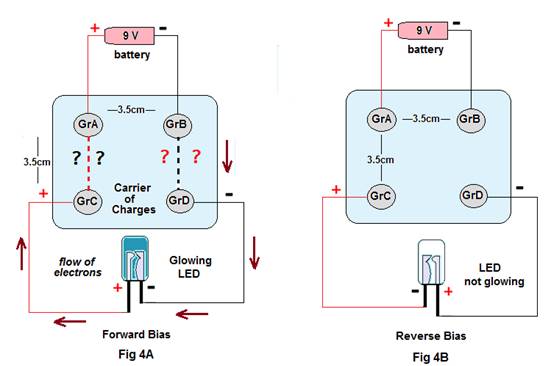
Experiment 1.Four graphite electrodes (GrA, GrB, GrC&Gr D) are placed in a bowl containing 30 ml of distilled water at a distance of about 3.5 cm as shown in fig 4A. The graphite electrodes GrC & GrD are placed just at the opposite side of GrA & GrB respectively. Now an LED is connected to GrC & GrD while a 9 volt battery is connected to GrA & GrB (connections shown in Figure4A).
Discussion: The emf shown at the GrC- Gr D pair is about 1.46 volts. The LED glows only when its negative terminal is connected to the Graphite electrode that is in front of the Graphite electrode connected to the negative terminal of the battery (i.e. negative terminal of LED is connected to GrD). Thus in this case GrD becomes the negative end while GrC becomes the positive end and it seems the LED is forward biased (Fig4A) . But when the negative terminal of the LED is connected to the Graphite C (GrC) the LED does not glow and thus seems to be reversed biased. If the result is owing to conduction of electrical energy by ions, it is not understood how the ions approach to neutral electrodes that too selectively (i.e.GrC in both cases behaves as positive electrode while Gr D as a negative electrode, but never the reverse.
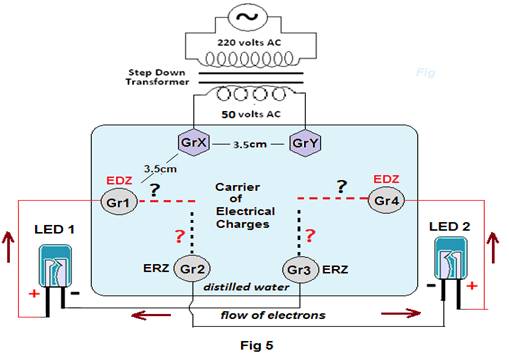
Experiment 2: Six graphite electrodes are placed in a bowl containing 30 ml of distilled water as shown in figure at a distance of about 3.5cm. Now using a step down transformer, 220 volts AC is step down to about 50 volts AC which is then connected to GrX & GrY (Fig5).
The AC emf shown by a combination of pairs of electrodes from 1 to 4 is recoded in table4.
Table4
| Sl no. | Electrode pairs | AC emf in volts | |
| 1 | Gr1-Gr2 | 7.8 | |
| 2 | Gr1-Gr3 | 14.2 | |
| 3 | Gr1-Gr4 | 15.3 | |
| 4 | Gr2-Gr3 | 8.0 | |
| 5 | Gr2-Gr4 | 10.1 | |
| 6 | Gr3-Gr4 | 3.2 | |
Next LED1 is connected with its positive terminal to Gr1 while its negative terminal to Gr3. The LED1 glows. Thereafter LED2 is connected to different combinations and result shown in tabular form.
Table5
| Sl no. | Graphite electrodes to which terminals of LED1 are connected | Graphite electrodes to which terminals of LED2 are connected | Result | ||
| Positive(+) terminal of LED1 | Negative (-) terminal of LED1 | Positive(+) terminal of LED2 | Negative (-) terminal of LED2 | ||
| 1 | Gr1 | Gr3 | Gr2 | Gr3 | Both LED1 &LED2 glows simultaneously |
| 2 | Gr1 | Gr3 | Gr2 | Gr4 | Both LED1 &LED2 glows simultaneously |
| 3 | Gr1 | Gr3 | Gr3 | Gr2 | Both LED1 &LED2 glows simultaneously |
| 4 | Gr1 | Gr3 | Gr4 | Gr2 | Both LED1 &LED2 glows simultaneously(Fig 5) |
Discussions:This experiment again shows that For LED1, Gr3 is ERZ (Electron rich zone) while Gr1 is EDZ( electron deficient zone). But for LED2, Gr3 acts as ERZ (as shown in sl. 1) and EDZ ( at sl.3). Similarly Gr2 & Gr4 also acts as both ERZ &EDZ depending on the situation. As both the LEDs glow for all the conditions stated above, this clearly questions then how the ions are able to make an electrode (i.e.Graphite) positive (EDZ) or negative(ERZ) according to the load (i.e. here LEDs) to which it is connected.As the existing theory allows ions to approach and get neutralized to its reverse electrode only (i.e. cations and anions to the cathode and anode respectively), how the AC emf is conducted through water to all the four electrodes Gr1 to Gr4 certainly seems a mystery.
[C] Retention of electrical energy bydistilled water due to DC emf /alteration of electrical strain in distilled water due to AC emf
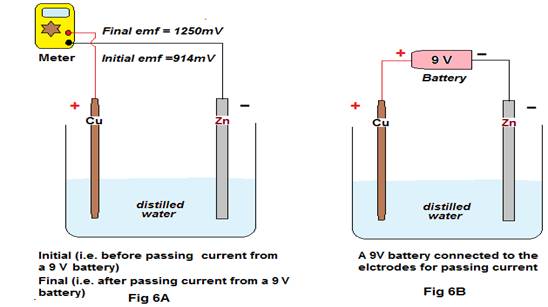
Experiment 3: The experiment is performed with the following six (6) pairs of electrodes with varying combinations. The 1st pair of electrodes Cu & Zn was put in a beaker containing pure/ distilled water and its emf was recorded as “Initial” emf (Fig. 6A). After recording of initial emf , external emf of about 9 volts was allowed to pass through it, such that the positive terminal of the external source (i.e 9 volt battery) is attached to the positive electrode of the 1st pair (i.e: Cu) & negative terminal to the negative electrode of the 1st pair (i.e: Zn)(Fig 6B).The external emf was allowed to pass for 60 seconds and thereafter disconnected. The Final reading (Fig 6A) is recorded after 15 seconds.
Table 6
| Sl No. | Pairs of Electrode | Type of electrode | Initial ( i.e:Before passing External emf)(in mV) | Final (i.e: After passing External emf of 9 volts for 60 seconds through similar Poles)(in mV) | |
| Positive (+) | Negative(-) | ||||
| 1 | Cu | Zn | Metal-Metal (Different) | 914 | 1250 |
| 2 | Gr. | Zn | Non metal-Metal | 1203 | 2030 |
| 3 | Gr | Cu | Non metal-Metal | 150 | 1290 |
| 4 | Cu | Cu | Metal-Metal (Same) | 10 | 482 |
| 5 | Zn | Zn | Metal-Metal (Same) | 30 | 238 |
| 6 | Gr | Gr | Non metal-Non metal | 11 | 1580 |
Discussion:This experiment is performed to show the effect of passing of DC emf through electrodes dipped in water4.
Here, Cu = Copper (Metal), Zn = Zinc (Metal), Gr = Graphite (Non-metal). In the above six cases it is found that even pure distilled water can generate a remarkable emf with different combinations/types of electrodes. The generation of such emf is therefore either due to chemical reactions or to the presence of ions in water (H3O+& OH–). As there is hardly any chemical reaction with the stated pairs and water, the generation of such emf may be attributed to the presence of H3O+& OH– ions in water (as even the purest form of distilled water contains these ions). However,
a) If the generation of emf in water is due to the presence of H3O+& OH– ions only,there should not be such extreme variations in emf for the above six pairs (i.e. emf of Cu-Cu being 10 mV while 1203 mV in case of Gr-Zn). The range of (1203-10)=1193mV is quite notable.
b) the increase in emf of Cu-Cu pair from 10 mV to 482 mV and Gr-Gr pair from 11mV to 1580mV on passing of current from an external source certainly seems a challenging one to be explained by the present concepts of charging of a cell/battery wherein a definite chemical reaction takes place while charging and discharging e.g. in Lead Accumulator the chemical reaction while charging/discharging is shown by .
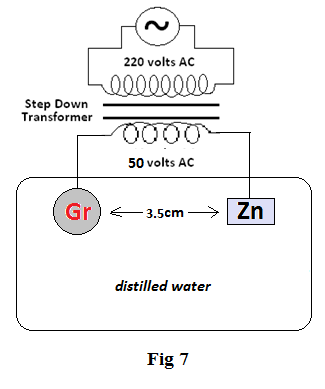
Experiment 4: Two electrodes Gr & Zn are placed in about 20ml of distilled water (Fig7) . The DC emf of Gr & Zn is recorded as initial emf . The two terminals of the transformer producing 50 volts AC (actually 56 volts AC) are then connected to Gr & Zn and alternating current is allowed to pass for 3 minutes. The initial /final emf of Gr & Zn is recorded in the table 7 below. The experiment is then repeated with Gr-Gr and Gr-Pb.
Observations: The observations are noted in table 7. below:
Table 7
| Electrode+ – | Initial(ACemf in Volts)Before passing of AC emf | Final(ACemf in Volts)After passing of AC emf | Initial(DCemf in mV)Before passing of AC emf | Final(DCemf in mV)After passing of AC emf | Difference(in mV) |
| Gr- Zn | 0 | 0 | 802 | 674 | –128 |
| Gr- Gr | 0 | 0 | -14 | –26 | –12 |
| Gr- Pb | 0 | 0 | 725 | 698 | –27 |
Discussion: This experiment is performed to show the effect of passing of AC emf (alternating current) through electrodes dipped in water6.
For all the three pairs (i.e. Gr- Zn , Gr- Gr &Gr- Pb ) though there is no change in AC emf found after passing of AC emf through the pairs for 3 minutes, but sufficient change in DC emf is recorded for the said pairs. The decrease in original emf of the pairs on passing of alternating current undoubtedly highlights the effect of passing of alternating current through distilled water. Finally, it shows that passing of AC (alternating current) through water, though not helping generation of AC emf, certainly helps generation (i.e. bringing change) of DC emf. The generation of DC emf seems remarkable on restricting flow of electrons suitably. Now, it is well known that the change in DC emf of water on passing of electrical energy is owing to the change in molecular orientation of water4. It is therefore, obvious that alteration of the DC emf is certainly the result of the molecular orientations induced by Alternating Current in distilled water
Explanations in support of electrical strain concept in water;
Now, we present our views explaining the efficacy of homeopathic dilution through some peculiar electrical properties of water and being totally independent of Avogadro’s number in regards to the presence of medicinal molecules in the dilution.
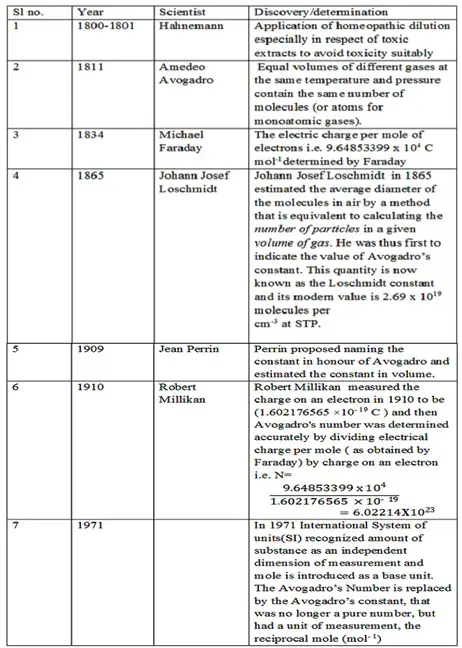
- Homeopathic dilutions have been used since 1800 and remained unchallenged until determination of Avogadro’s number by Millikan in 1910 (as presented below in tabular form). A mathematical calculation based on Avogadro’s Number led to the conclusion that homeopathic dilution must be nothing but placebo after homeopathy had already been used for 109 years !!
Summary: Hahneman devised methods of diluting the drugs he was testing in order to lessen their toxic effects. He started using his homeopathic dilutions as medicines around 1800-1801 by nullifying the toxicity of toxics through dilutions. Avogadro in 1811 gave us the concept of having the same number of molecules (or atoms for monoatomic gases) in equal volumes of different gases at the same temperature and pressure. Though he proposed the idea of the same number of molecules in equal volume of different gases under certain conditions, he didn’t determine the actual number.
In 1865 Loschmidt found the value of Avogadro’s number by estimating the number of particles in a given volume of gas. The number is now known as the Loschmidt constant and its modern value is 2.69 x 1019 molecules per cm-3 at STP. In 1909 Jean Perrin proposed naming the constant in honour of Avogadro and estimated the constant with respect to volume. In 1910 on estimating the charge on an electron, Robert Millikan determined the Avogadro’s number accurately by dividing electrical charge per mole – (i.e. 9.64853399 x 104 C mol-1 as determined by Michael Faraday in1834) by the charge on an electron.
Since 1910, further progressive calculations have made accurate determination of the values for the Faraday constant and the elementary charge that in turn resulted in more accurate determination of Avogadro’s number by the scientists. Possibly on recognition of amount of substance as an independent dimension of measurement and introduction of mole [i.e. the amount of any substance that contains as many elementary units (atoms molecules etc ) as there are atoms in 12 gms of 12C, corresponding to Avogadro constant, which has a value of 6.02214129×1023 elementary units ] as a base unit by the International System of units (SI) in 1971 , the amount of substance present in the Homeopathic dilution has been challenged.
It is not proper to discount homeopathy on the basis of Avogadro’s number without explaining the fundamental contradictions in science. The main vehicle of homeopathic remedies being water, we have shown its various electrical properties in relation to generation, conduction and retention of electrical energy. Some of the apparent contradictions in regards to generation , conduction and retention of electrical energy by water are presented below:
Generation:
- How distilled water generates emf with different metals/non-metal electrodes without chemical reactions or having concentration difference of electrode /electrolyte (e.g. as shown in Table2 )
- How the human body generates emf with different electrodes without chemical reactions or having concentration difference electrode /electrolyte (e.g. as shown in Table2 )
- How emf in case of Einthoven triangle is generated7e. whether there is any chemical reaction between the human body and electrodes placed on the body or existence of any concentration gradient in respect of electrode /electrolyte.
- How emf in the case of the Electromagnetic Flowmeter is generated7 In the electromagnetic flow meter, the conductor is water, that when placed in a magnetic field is able to show induced current due to induced emf. What then generates induced current in water? It is believed that such generation of emf is due to the presence of ions in water. This suggests that ions, like electrons are also deflected in the magnetic field, thereby generating induced emf and hence induced current. Therefore, in water ions seem to flow between electrodes of Electromagnetic Flowmeter, in lieu of electrons, while generating emf in presence of a magnetic field. Further it implies that the difference in concentration of ions at the electrodes of the Flowmeter generates emf by pushing ions through the water. This shows that ions not only can move towards the opposite electrodes as in electrolysis, but also can move even to the neutral electrodes for generating emf , as in the electromagnetic flow meter!
- The method used to measure emf in case of Cell membrane potential is quite amazing 7.
As we are concerned with the potentials due to Na+ & K+ ions in the intracellular and extracellular region it seems quite contradictory, using such electrodes that do not contain any reversible ions. Why amalgamated electrodes, as per existing theories in chemistry are not used, certainly seems a reasonable question.
Conduction:
- How Dc emf is conducted from GrA & G rB to the neutral electrodes GrC & Gr D i.e. why the ions approach to neutral electrodes for generating emf at GrC –GrD cannot be explained by the existing theories that allow only approach of any ions to its opposite electrodes while conduction i.e. cations (positive ion) to cathode(negative electrode) and vice versa.
- How AC emf is conducted from GrA & GrB to all the neutral electrodes 1,2,3 & 4 i.e. why the ions approach to neutral electrodes for generating emf at Gr1 to Gr4 cannot be explained by the existing theories. How the emf is generated at the four electrodes simultaneously is also surprising.Moreover ,if the generation of emf at electrodes Gr1 to Gr4 is owing to ionization and subsequent migration of ions then why there is so much difference in generation of emf, Gr1-Gr2=7/8 volts AC while Gr1-Gr4=15.3 volts AC (Table:4)
- How alternating current (AC) is conducted through water while earthing /grounding8.
Retention:
- How DC emf is retained even in distilled water i.e. distilled water is able to hold electrical charge similar to that of a rechargeable battery/accumulators.
- How AC em falters DC emf of a pair of electrodes in distilled water.
All the stated electrical properties of water remain unexplained by the existing theories of science. We explained the same considering the fact that:
- Orientations of water molecules change according to induced electrical strain 4,9.
- Water re-orients its molecules as per necessity during conduction or retention or generation of electrical energy10.
- It is not ions but delocalization of electrons in water molecules that conduct electrical energy through orientations of water molecules11.
– In conventional medicines the molecules are believed to convey the medicinal power in the living body but in homeopathic dilutions it is not the molecules of medicine but the electrical strain induced in the vehicle,by the substance, that conveys the medicinal power of a substance in the living body12.
– Micro current therapy (i.e. supplying of electrical currents less than 1micro ampere (1µA), to nerves using MENS(Micro current Electrical Neuromuscular Stimulator) through electrodes placed on the skin was especially used in sports medicine but owing to its outstanding efficacy it now has extensive use even in chronic cases.
– The micro current is often used to relieve pain instead of analgesics (i.e. paracetamol , NSAID etc). It means that the pain can be get relieved either by molecules ( i.e. by molecules of analgesics) or without any molecules (i.e by applying suitable electrical energy only) as found in homeopathic dilutions. It is thus evident that application of suitable electrical energy has the same effect as chemicals; the mechanism however may differ. It is believed that as the micro currents used are in close proximity to the body’s natural electrical current, at cellular level they are able to restore normal activity. Thus the mode of action of the micro current therapy and homeopathic dilution seems to be same i.e. without any molecules and exclusively due to micro current /electrical strain induced at the cellular level.
– When preparing serial homeopathic dilutions the electrical strain of the latter differs from the former dilution in regards to difference in molecular orientations of water13 (Table1) but cannot be determined due to non-availability of any such scientific instruments and for which the homeopathic dilution cannot be held responsible.
Thus for the reasons cited above we suggest homeopathic dilution to be independent of Avogadro’s number in regards to presence of active medicinal molecules and instead dependent on electrical strain of water molecules induced by the medicinal molecules.
Inference: Homeopathy has no relation to Avogadro’s number, in regards to molecules, but to the electrical strain in water molecules induced by the medicinal molecules through molecular orientations of water.
Reference:
[1] Origin of Cell membrane Potential : Homeopathy Explained as Orientation of Water Molecules,Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, December 2013hpathy.com
[2] Electromagnetic Flow Meters Generate Emf Through Orientations of Water Molecules,Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, May2012hpathy.com
[3] Untold Facts of Science –The Science Behind Homeopathy,Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, May2012hpathy.com
[4] Water the Magic Liquid: Retaining Electrical Energy, Ruhul Amin &BiplabChakraborty (Published in Homeopathy for Everyone, April 2012 hpathy.com
[5] Generation of electrode Potential of an electrode –an explanation, Ruhul Amin &BiplabChakraborty (Simillimum,vol.XXIII.2010, www.hanp.net)
[6] The Origin of Homeopathic Imponderabilia – A.C. Current Changes Molecular Orientation of Water,Ruhul Amin &BiplabChakraborty (Published in Homeopathy for Everyone Oct 2014hpathy.com)
[7] Textbook of Medical Physiology (Eleventh Edition) -Guyton and Hall
[8] The Phenomenon of Electrical Earthing Explained By Orientations of Water Molecules,Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, Jan 2014hpathy.com
[9] Solutions Ionic or Non-ionic Conducts & Retain Electrical Energy Ruhul Amin &BiplabChakraborty (Published in Homeopathy for Everyone July 2012 hpathy.com)
[10].Orientations of water molecules-the force behind Homeopathy, RuhulAmin ,BiplabChakraborty (Published in Homeopathy For Everyone Apr 2013 hpathy.com)
[11] Delocalization of Electrons Through Orientations of Water Molecules Helps Electrical Conduction In Water,Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, March 2014hpathy.com
[12].The electrical energy of substances –the secret of homeopathy, Ruhul Amin &BiplabChakraborty (Simillimum,vol.XXII, Summer Fall/ 2009 www.hanp.net)
[13] The Homeopathic Dilution-A New Explanation, Ruhul Amin, BiplabChakraborty&FarookRahaman (Simillimum,vol.XXI, Summer Fall/2008). www.hanp.net
[14] Wikipedia

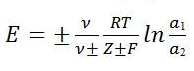


Thanks for the article on the explanation and preparation of Homeopathic medicine showing the dynamic curative force at the base level of energy of a substance with an experiment. But, at one place a doubt has arisen in my mind in the sentence of the article, ‘1C potency 1 ml of mother tincture is added to 9ml of distilled water and …… In this regard I have a question. ‘ Is 1 ml of mother tincture added to 9 ml of distilled water to produce 1C potency? Perhaps, instead of ‘9 ml’ it will be ’99 ml’ so far as 1C = 1 part of 100th part. Thanks to you once again.
Tariqul Alam
Sir,
Thanks!
Actually as per Homeopathic Pharmacy Natrum Mur (Sodium chloride) is included in Class V(A) and 1st potency of Natrum Mur is stated to be prepared by adding 90 minims of purified water to the 10 minims of the mother solution i.e. in the ratio 1:9 . In this regard you may kindly refer to “ A text book of Homeopathic Pharmacy” by Dr D D Banerjee published by B Jain Publishers (P) Ltd.
Moreover, to see how we categorically explained “ How and why Homeopathy is scientific” please visit our blog “Recent Homeopathic Research Dr. Md. Ruhul Amin” (www.aminchakraborty.blogspot.com) and share your views for development of this research.
Thanks for sharing your views.
Ruhul Amin & Biplab Chakraborty
Being as Avogadro’s number only applies to gases in any case, surely it’s irrelevant when discussing liquid dilutions as is the case in homeopathic remedies?
This all looks very scientific but I, along with most of its readers will have insufficient expertise to properly evaluate your methods or conclusions.
Has this paper been published and peer reviewed so that those with sufficient expertise can properly examine it?