Most metals are very good conductors of electricity. In metals, electrical conduction arises by the movement of electrons, unlike the mechanism of conduction in aqueous solutions or fused melts of ionic compounds, where ions migrate during conduction.
The very property of water to convey the medicinal effect of a substance through retention and conveyance of electrical impulses is of great importance, not only for homeopathy but for the development of Science1. It is well known that conductance of electrical energy is a flow of electrons for metals, through ions in aqueous solution or water, and through holes in semiconductors.
It is said that the metallic bond which is responsible for holding/bonding atoms of a metal or metals (for alloys) together, helps movement of electrical energy through available electrons in its conduction band. In other words, delocalized electrons in the conduction band, help the flow of electrical energy. In the Molecular Orbital Theory the valence electrons are considered to be associated with all the nuclei concerned, as atomic orbitals combine to produce molecular orbitals. A brief discussion regarding conduction in conductors and semiconductors is presented here:
Conductors:
In electrical conductors (metals), either the valence band is only partly full, or the valence and conduction bands overlap. There is therefore no significant gap between filled and unfilled Molecular Orbitals (MO), and perturbation can occur readily.
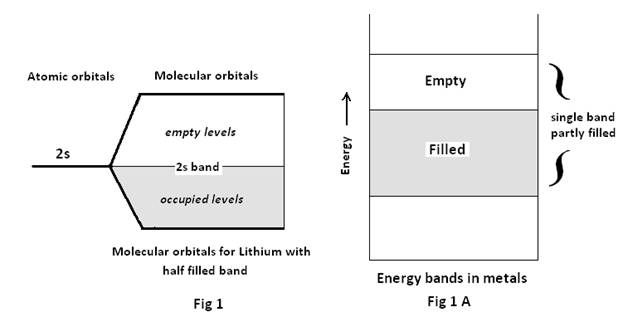
For the Lithium molecule with configuration 1S2 2S1, if n atoms of Li are present in a Lithium crystal, then n 2S atomic orbitals combine to form n 2S molecular orbitals. Now the total valence electrons being n, they will find their place in the n/2 MO (as each MO can accommodate 2 electrons) and hence there will be n/2 filled MO and n/2 vacant MO Fig 1.
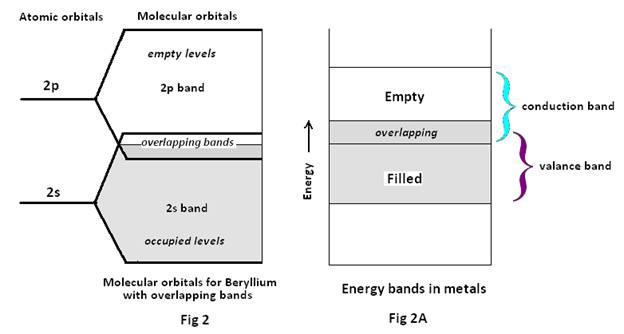
But in Beryllium with electronic configuration 1S2 2S2 , if there are n atoms of Be in a crystal combine they will combine to form n 2S MO. As the total number of valence electrons in the Be crystal is nX 2 it may be assumed that they all will be accommodated in the n2S MO (as each MO can accommodate 2 electrons). But this is not the fact, since in an isolated Be atom the 2S and 2p atomic orbitals are some 60 Kj mol-1 different in energy and in the same way as 2s AOs form a band of MOs, the 2p AOs (atomic orbitals) form a 2p band of MOs. The upper part of the 2s band overlaps with the lower part of the 2p band. Because of this overlap of the bands, some of the 2p band is occupied and some of the 2s band is empty Fig 2.
In both the cases it is found as if there is a single energy band with no forbidden gap (Fig 1A & Fig 2A). Consequently if any electron at the top of the filled portion of the band gains a little extra energy, say under the influence of electric field, it can easily move up into the empty part of the energy band. So now the electron being free to move in the crystal, may be termed a free electron.
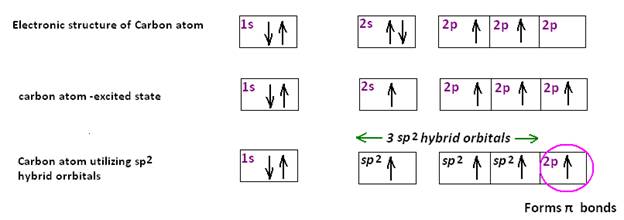
Non-Metal conductor: In graphite only three of the valence electrons of each carbon atom are involved in forming sp2 hybrid bonds, the fourth electron forming a π bond. The π electrons in graphite are mobile, shared by a whole layer of carbon atoms and hence conduct electricity.
Insulators: The conditions for a crystalline substance to be an insulator at room temperature are
a) Its valence band must be full
b) The conduction band must be normally empty
c) The forbidden gap must be greater than about 1eV.
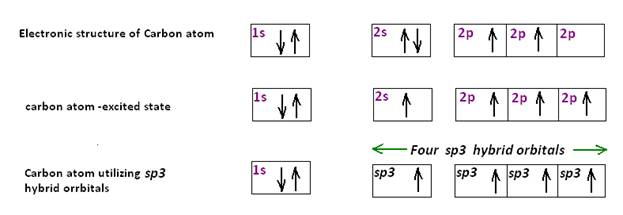
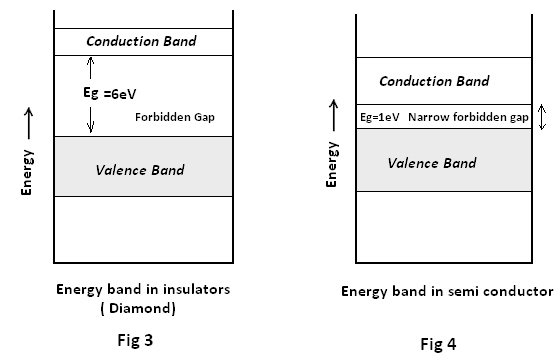
In diamond, which is colourless, each carbon atom utilizes sp3 hybrid orbitals to form four bonds. Thus each carbon atom is tetrahedrally bonded to four other carbon atoms and a three dimensional polymer is formed .
The energy band diagram (Fig 3) shows that it requires about 6eV of energy to move an electron from the valence band to the conduction band, whereas average energy possessed by an electron at room temperature i.e 300K is somewhat less than the required value. Therefore, diamond is a very good insulator at room temperature. An electric field cannot give this amount of energy to an electron in the solid (diamond).Thus no electron could acquire sufficient kinetic energy to cross the forbidden gap in the electric field. Therefore there will be no flow of electrical energy and the solid will be an insulator. But as average energy possessed by the electron rises with temperature, quite a good number of electrons cross the forbidden gap and thus conductivity of diamond increases with temperature.
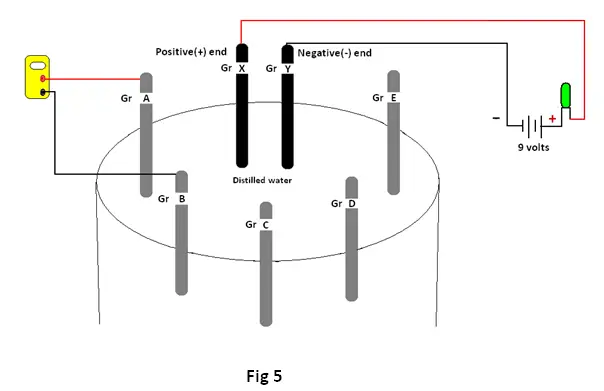
Semi conductors : Actually a semiconductor has the energy band structure very similar to that of an insulator, with the only difference that in the semi conductor , the conduction and the valence band are separated by a narrow forbidden gap of about 1eV (fig 4). As in diamond, an energy gap separates the top of the filled valence energy band from a vacant higher band (conduction band). But the forbidden gap for Germanium and Silicon are 0.76eV and 1.1eV respectively. At low temperatures the conductivity of Silicon is very low. Since the energy gap is small even at room temperature, a small fraction of electrons in the valence band have sufficient kinetic energy owing to thermal agitation to cross the relatively small energy gap to enter the conduction band above it. The fractions are however sufficient to permit flow of limited amounts of electrical energy when an electric field is applied. Thus Silicon, having electrical conductivity lying between that of conductors and insulators, is considered a semiconductor.
The theory of conduction of electrical energy in water or aqueous solution got questioned when it was shown that solutions ionic or non-ionic conduct electrical energy through the orientation of water molecules2. But still the vital question remained how the orientation of water molecules could help such conduction of electrical energy. To help explain conductivity of electrical energy in water the following experiment is performed:
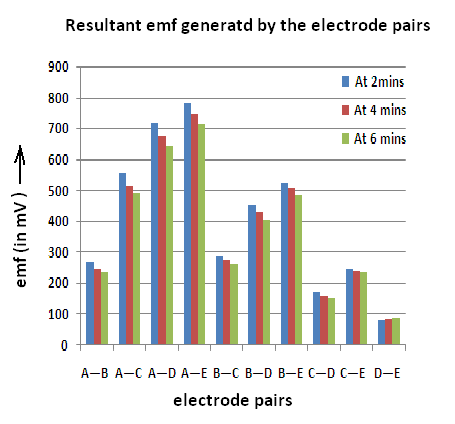
Experiment: An LED is connected to a 9 volt battery through a pair of graphite electrodes (Gr X & Gr Y) immersed in distilled water. Five Graphite electrodes Gr A, Gr B, Gr C, Gr D & Gr E are placed at different positions (about 3cm apart) in a beaker as shown in Fig 5.The emf of different pairs of electrodes (A,B,C,D &E) are measured before and after passing electrical energy through the circuit containing the LED. Results are recorded in the table below at 2minutes, 4mins & 6 mins:
| Electrode | Initial emf |
Final emf on passing electrical energy(in mV) |
Resultant emf (in mV) |
||||
| Pairs | (in mV) | At 2minutes | At 4 minutes | At 6 minutes | At 2mins | At 4 mins | At 6 mins |
| A—B |
36 |
303 |
279 |
270 |
267 |
243 |
234 |
| A—C |
19 |
575 |
532 |
508 |
556 |
513 |
489 |
| A—D |
26 |
745 |
699 |
665 |
719 |
673 |
639 |
| A—E |
76 |
857 |
824 |
791 |
781 |
748 |
715 |
| B—C |
-17 |
269 |
253 |
242 |
286 |
270 |
259 |
| B—D |
-10 |
440 |
417 |
395 |
450 |
427 |
405 |
| B—E |
42 |
563 |
548 |
526 |
521 |
506 |
484 |
| C—D |
8 |
176 |
166 |
156 |
168 |
158 |
148 |
| C—E |
61 |
302 |
300 |
293 |
241 |
239 |
232 |
| D—E |
54 |
129 |
135 |
140 |
75 |
81 |
86 |
Discussion: In this experiment when the current is allowed to pass through the circuit containing the LED, it is believed that the LED glows due to the passage of electrical energy through the ions present in distilled water. The ions being neutralized at the opposite electrodes while conveying current i.e, H3O+ ions move towards Cathode (negative electrode) while OH– move to the Anode (positive electrode). From the result shown in the table, two things are not clear:
i) Why the ions move to the relatively neutral electrodes ( A,B,C,D &E) to generate such emf ?
ii) Why and how the presence of ions in water generates different emf for different pairs of graphite electrodes?
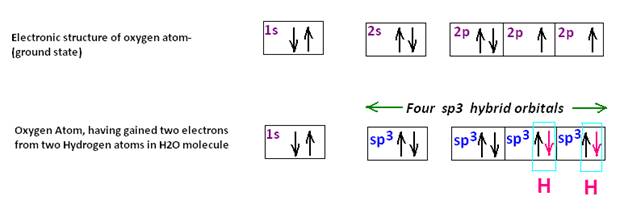
Had the emf shown by different pairs of Graphite electrodes been due to ions in water, they should have been nearly the same, if not equal. It is also not clear why Graphite A behaves always as a positive electrode while Graphite E is never positive but always negative. A graph is plotted for emf generated by a pair of electrodes at 2 mins, 4mins & 6mins.
It shows that emf generated by the electrode pairs A to E is in the order A-E>A-D>A-C>A-B. That shows that negativity of the electrodes is in the order E>D>C>B>A. i.e, E is at relatively highest negative potential .
For the pairs B to E, emf generated is in the order B-E>B-D>B-C that again shows that negativity of the electrodes is in the order E>D>C>B. i.e, E is at relatively highest negative potential.
Similarly for the pairs C to E where C-E> D-E, again E is at relatively highest negative potential.
It therefore appears that Gr A being closest to Positive electrode of the circuit containing the LED is always positive and is always at relatively positive potential whereas Gr E being closest to the negative electrode of the circuit containing the LED is always at relatively negative potential, while the rest are intermediate in nature. So, B, C & D are negative with respect to A but are positive with respect to E.
It is believed that conduction of electrical energy takes place through delocalization of electrons in both conductors and semi conductors and through delocalization of π electrons in the case of Graphite. While for water and aqueous solution it is believed to be conducted by ions (i.e; through cations and anions present in the water / aqueous solution moving to the opposite electrode). As it is already shown that conduction of electrical energy in water is not due to ions 3 the option left is delocalization of electrons within water molecules, which can explain such peculiar electrical properties. That electrons in water molecules are not localized but rather delocalized is further supported by the following facts;
i) That an electrode behaves as both cathode & anode simultaneously during conduction of electrical energy 3 .
ii) That in a molecule of water, oxygen, the central atom, has two unpaired electrons, which are sufficient to form the two bonds required. The molecule being triatomic must be either linear with a bond angle of 1800 , or else angular, that is, bent. But as there are four electron pairs (2 bond pairs and 2 lone pairs) in the outer orbital of oxygen in the water molecule, they should be tetrahedrally arranged to minimize electron repulsion and the resulting molecule would be angular. The bond angle HOH in water is 1040 27’ instead of 1090 28’ (as for a true tetrahedral) due to the distortion by two lone pairs. In the water molecule, each oxygen atom utilizes sp3 hybrid orbitals to form two bond pairs with two H-atoms and other two having lone pairs. As the four sp3 hybrid orbitals are formed by the combination of one “s” and three “p” orbitals, they are of equal energy and thus can allow shifting of electrons for any minor energy change as found in self ionization of water molecules: H2O + H2O=H3O+ + OH–, wherein both the ions are stabilized by the delocalization of electrons.
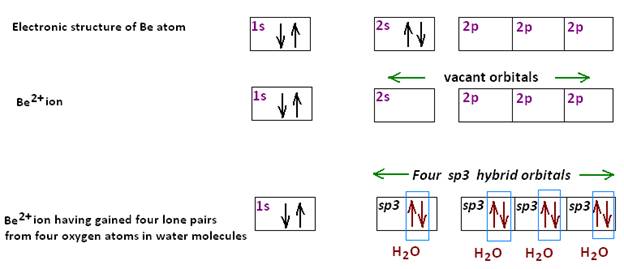
iii) Metals are electropositive and have a tendency to lose electrons when energy is supplied: M→M+ + e .The electropositive nature of a metal is also shown in the degree of hydration of the ions. This is due to the fact that in the change: M+ to [(H2O) n →M]+ the positive charge becomes spread over the whole complex ion. Since the charge is no longer localized on the metal, this is almost the same as the change M+→M. In other words the process of hydration also involves delocalization of electrons of water molecules simultaneously. As In the Beryllium salt, such as [Be(H2O)4]Cl2, Be ion(Be2+) forms the tetrahedral complex ion [Be(H2O)4]2+ with water having gained four lone pairs from four oxygen atoms in water molecules.
iv) The abnormal properties of liquid water are attributed to the presence of associated water molecules. The presence of (H2O) 2 and (H2O) 3 in liquid water is due to hydrogen bonds . X-ray studies indicate that ice has a crystalline structure, so that each H2O molecule in it is surrounded tetrahedrally by 4 others –the water molecules being held by H-bonds. Even liquid water has a partly broken down ice structure. As the lifetime of Hydrogen bonds is on the order of some pico seconds i.e; 10-12 sec, H-bonds are broken and formed at the rate of 1012 times per second. Moreover owing to non-electrical strains and electrical imbalances (due to formation of hydronium ions as a result of self ionization of water molecules: H2O + H2O=H3O+ + OH–) formation of H-bonds take place at random. Thus breaking and formation of H-bonds at random along with non-electrical strains & electrical imbalances also helps delocalization of electrons in water molecules.
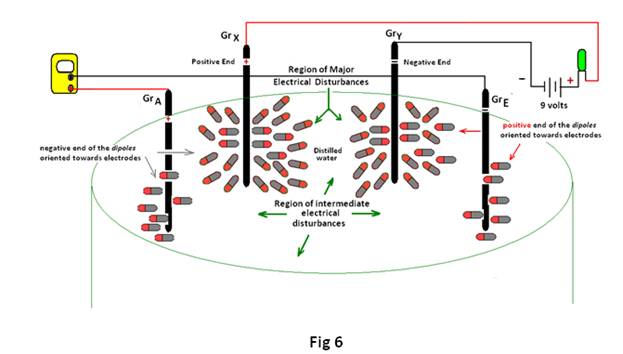
v) When current from any external source is allowed to pass through distilled water, as in electrolysis, hydrogen is evolved at the negative electrode while oxygen at the positive electrode, since cations and anions move to the opposite electrodes. Similarly when any circuit is made with an LED it glows, which is again said to be due to movement of ions to the opposite electrodes for conveying electrical energy. But it is already shown that conveyance of electrical energy is possible even below the decomposition potential of water i.e 1.23 volts 4. As the water molecules are polar (i.e having negative and positive poles) they definitely have some specific orientation when subjected to a particular electric field. So, when two electrodes connected to some external source are placed in distilled water, at the positive electrode (Anode) water molecules in its close proximity point their negative end towards the electrode (i.e oxygen is pointed towards the anode) while at the negative electrode (Cathode) water molecules in close proximity point their positive end towards the electrode (i.e hydrogen is pointed towards cathode) as shown in Fig 6.
Thus the external emf orients the water molecules specifically which in turn helps delocalization of electrons throughout the water molecules and can explain the generation of emf by the relatively neutral electrodes. In other words, when two electrodes connected to an external source of electrical energy are placed in distilled water, it divides the entire region into three distinct regions that are as follows:
a) region of major electrical disturbances near Cathode (negative electrode) where water molecules in the close proximities are oriented towards the cathode with its positive end (i.e Hydrogen) directed towards the electrode.
b) region of major electrical disturbances near Anode (positive electrode) where water molecules in the close proximities are oriented towards the anode with its negative end (i.e Oxygen) directed towards the electrode.
c) region of intermediate electrical disturbances that results to counter the electrical imbalances due to the regions of major electrical disturbances. This act of counterbalancing the electrical imbalances at the two electrodes by the water molecules present in the intermediate region, help delocalization of electrons in the entire region. The delocalization of electrons throughout the entire region is not uniform, as the orientation of water molecules is greatly influenced by the nature of its nearest electrode (i.e cathode or anode). This accounts for the always positivity of Gr A (nearest to the positive electrode), negativity of Gr E (nearest to the negative electrode) and intermediary of the rest (B,C & D) that causes generation of varying emf 5 even at the relatively neutral electrodes .
Inference: It is not ions but delocalization of electrons in water molecules that conduct electrical energy through orientations of water molecules.
References:
1) Orientations of water molecules-the force behind Homeopathy. Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone Apr 2013 hpathy.com
2) Solutions Ionic or Non-ionic Conduct & Retain Electrical Energy Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone July 2012 hpathy.com)
3) The key to the Homeopathic dilution, Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone Nov 2012 hpathy.com
4) The Human Body And Water Both Retain Electrical Energy Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone Sep2012 hpathy.com
5) Water the Magic Liquid: Retaining Electrical Energy, Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone April 2012, hpathy.com


As a patient who has found in homeopathy the most fascinating path towards healing ever experienced in his lifetime, I set out years ago to try and understand a bit more about it. I found some books by Scholten and De Schepper especially eye-opening, not to mention the great “The Genius of Homeopathy” by Stuart Close. Yet none of them said much about HOW homeopathy works.
I’m also interested in physics, and through the years I made the effort to read quite a few books on the subject. Little did I know that as I delved deeper into both physics and homeopathy, I’d come to realize that the former holds the key to understanding at least part of the mystery that surrounds the latter.
I find current research in homeopathy extremely interesting, since it’s trying to bridge the gap between the two disciplines, leveraging the latest discoveries in physics to explain how homeopathy is able to achieve its miraculous effects – provided the correct remedy is chosen and administered in the right potency and dose.
Nowadays, studies on the memory of water – brilliantly synthesized by Massimo Citro in his recent book “The Basic Code of the Universe”, which will remain among my favourite books ever – seem to reveal so much more than we thought we could know about the mystery of homeopathy. And what I like so much about this field of research is that, at least in my opinion, it’s not depriving this medicine of its magic: on the contrary, once you learn about the unexpected properties of water, you’re left even more in awe with respect to the laws of the magnificent universe we inhabit.
Having said that, I would have enjoyed this particular article even more if it had stated more explicitly the relationship between the presented findings and the inner workings of homeopathy. Although this is a quite technical piece, one should keep in mind that the general public might also be interested enough to read it – and those of us who are neither physicists, nor certified homeopaths might need a little more guidance to grasp the significance of these discoveries for the field of homeopathy.
This minor detail won’t stop me from expressing my heartfelt congratulations to the writers, and to anyone committed to finally laying a firm scientific foundation for homeopathy. Since centuries of successful clinical experience apparently are not enough to neutralize the usual naysayers, let’s see if some groundbreaking discovery does the work! 🙂
A very well researched article, Late Dr Jacques Benveniste did some excellent research on memory of water but the world instead of encouraging him treated him so inhumanly that he died later with a broken heart.But such research one day will establish the working of Homeopathy and pave the way for its future glory.
Dr Wequar Ali Khan
Miami Florida