The study of water has revealed its mysterious behavior not only in respect of its physical or chemical properties but its electrical properties too. The theory of homeopathy being directly related to the electrical properties of water, much attention is being given to explore those properties, including conduction, retention and generation of electrical energy. In our earlier work we have shown that the mode of action of micro current therapy and homeopathic dilution seem to be the same, i.e. without any involvement of molecules and exclusively due to micro current /electrical strain induced at the cellular level.1
Delocalization of valence electrons and Pi electrons are said to conduct electrical energy in metals and graphite (non-metals) respectively. We have shown in our previous publications that ions do not conduct electrical energy2 in water or aqueous solution and it is yet to be identified which electrons are actually delocalized through orientations of water molecules for conducting electrical energy. Now, in search of the same we proceeded with the following experiments.
Materials Used: Adapter (220 volts AC to 12 volts DC, 500mA), A step down transformer (220 volts AC to 50 volts AC, 500mA), graphite rods, Zinc plate LED’s (low voltage range), glass bowl, meter, distilled water and NaCl.
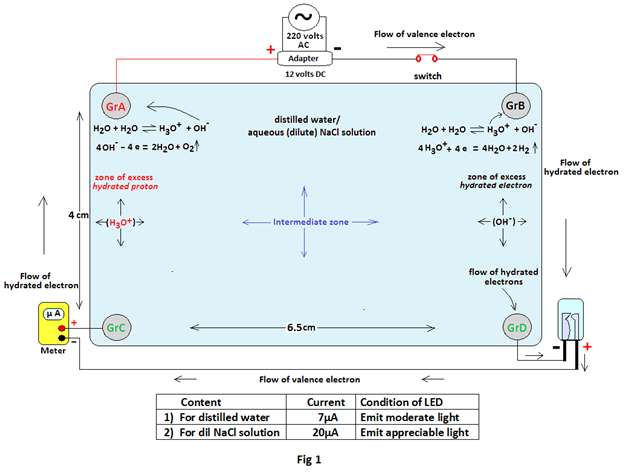
Experiment 1: A source of 12 volts DC (actually about 14.5 volts DC,500mA) from an adapter is connected to two Graphite electrodes (GrA & GrB) ,placed in 15 ml of distilled water contained in a glass bowl, through a switch. The positive terminal of the battery is connected to GrA while the negative terminal to Gr B. Two Graphite rods GrC & GrD are placed just opposite to GrA & GrB respectively . An LED (Light Emitting Diode) is also connected to GrC & GrD along with a meter as shown in Fig 1.
Observations: When the circuit is closed and the current is allowed through the circuit, observations are noted in the tabular form below. The observations are noted once again after addition of 2 drops of 8% of Sodium Chloride solution in the distilled water with gentle mixing to obtain a dilute salt solution (0.0533% or 0.00091N) in the container.
Table 1:
| Sl No. | Applied emf from external source at
GrA & GrB |
Content of the
Glass bowl |
Observed potential difference (p.d) at
GrC & GrD |
Condition of the LED connected to
GrC & GrD |
Current through the circuit |
| 1 | 14.48 volts dc | Distilled water | 2.48 volts | Emit moderate light | 7µA |
| 2.40 volts | ,, | 5µA | |||
| 2.45 volts | ,, | 7µA | |||
| 2 | 14.40 volts dc | 0.0533% of NaCl solution | 3.04 volts | Emit appreciable light | 25µA |
| 2.98 volts | ,, | 28µA | |||
| 3.02 volts | ,, | 35µA |
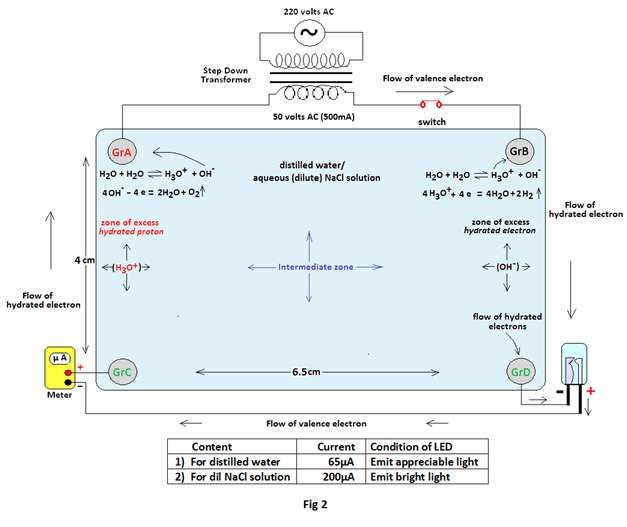
Experiment 2: The above experiment is repeated by replacing the source of emf to 50 volts AC (actually about 53.6 volts AC,500mA) by using a step down transformer (Fig 2).
Observations: The observations are noted below in tabular form.
| Sl No. | Applied emf from external source at
GrA & GrB |
Content of the
Glass bowl |
Observed potential difference (p.d) at
GrC & GrD |
Condition of the LED connected to
GrC & GrD |
Current through the circuit |
| 1 | 53.6 volts AC | Distilled water | 6.9volts ac | Emits appreciable light | 65µA |
| 7.5volts ac | ,, | 68µA | |||
| 7.8volts ac | ,, | 68 µA | |||
| 2 | 53.4 volts AC | 0.0533% of NaCl solution | 12.8volts ac | Emits bright light | 200µA |
| 13.5 volts ac | ,, | 256µA | |||
| 14.2 volts ac | ,, | 315µA |
Next, to see the effect of electrical conduction in water below the decomposition potential of water, the following simple experiments are performed:
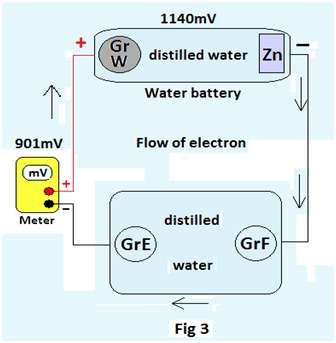
Experiment 3: A water battery is prepared by placing a graphite rod(GrW) and a zinc plate (Zn) in distilled water. The emf of this cell is noted. The water battery is then connected to a meter through two Graphite rods GrE & GrF dipped in distilled water contained in a glass bowl as shown in Fig 3.The potential difference between GrW & Gr E is noted.
Observations: The observations are noted in a tabular form
| Sl. No. | Emf of the water battery | Potential difference shown between GrW & GrE |
| 1 | 882 mV | 618 mV |
| 2 | 1140 mV | 901 mV |
| 3 | 1141 mV | 900 mV |
The potential difference of about 618mV to 900mV between GrW & GrE confirms the passage of electrical current through the distilled water between GrE & GrF even below the decomposition potential of water (i.e.1.23volts).
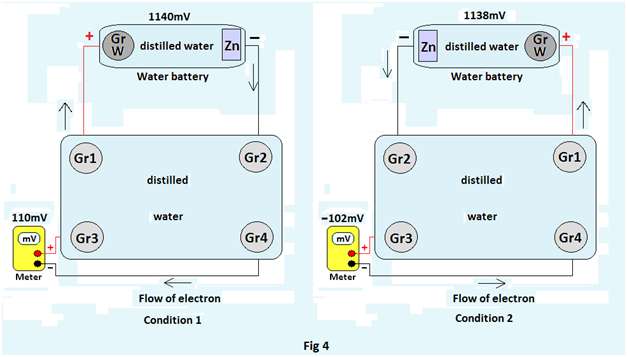
Experiment 4: A water battery is connected to two graphite rods Gr1(connected to GraphiteW) & Gr2( connected to Zinc). Another pair of graphite Gr3(connected to positive end of the meter) & Gr4(connected to negative end of the meter) is placed in 15 ml of distilled water contained in a bowl and its emf is measured as its initial emf. Now Gr1 & Gr 2 are placed in the same bowl such that Gr1 is in front of Gr3 while Gr2 in front of Gr4 and emf of Gr3 & Gr4 is again measured & noted as final emf (Fig4, condition 1) The experiment is repeated by reversing the order of placement of Gr1& Gr2( i.e. now,Gr2 is placed in front of Gr3 and Gr1 in front of Gr4) and final emf is noted again(Fig4, condition 2).
Observations: The observations are recorded in tabular form
| Condition no. | Condition type | emf of the water battery | emf of Gr3—Gr4
(initial emf) |
emf of Gr3—Gr4
(final emf) |
Difference |
| 1 | Gr1 placed in front of Gr3 & Gr2 in front of Gr4 | 988mV | -35mV | 20mV | 55mV |
| 1141mV | -20mV | 108mV | 128mV | ||
| 1140mV | -25mV | 110mV | 135mV | ||
| 2 | Gr2 placed in front of Gr3 & Gr1 in front of Gr4 | 990mV | -26mV | -70mV | -44mV |
| 1140mV | -18mV | -112mV | -94mV | ||
| 1138mV | -20mV | -102mV | -82mV |
The varying difference in initial and final emf of Gr3 & Gr4 confirms the flow of electrical energy from Gr1 & Gr 2 to Gr3 & Gr4 through the distilled water. Therefore, it is rather clear that the final emf observed is certainly not the emf generated by Gr3& Gr4 itself but the potential difference due to conduction of electrical energy from Gr1 & Gr2 to the graphite pair Gr3 &Gr4.Here also electrical conduction through distilled water is found even below decomposition potential of water.
Discussions
In experiment 1 & 2, the potential difference found at GrC & GrD that helps power the LED confirms the flow of electrical energy through the circuit. Though two graphite rods placed in distilled water show some emf acting like an energy source3 (in the range of (10 to 20mV), from the potential difference found in these experiments (about 2.40 volts dc and about 6 volts AC) it is evident that the LED uses the electrical energy applied at GrA & GrB from the external source. But it remains a question how such electrical energy is conveyed to the rods GrC & GrD connected to the LED both for the distilled water & the salt solution.
In experiment 3 & 4, it is found that conduction of electrical energy takes place through distilled water even below the decomposition potential of water (i.e. 1.23volts)
Now, according to the Arrhenius theory of electrolytic dissociation:
- A portion of the electrolyte in solution is dissociated into positively charged and negatively charged particles. These charged particles are called ions, positively charged and negatively charged particles being cations and anions respectively. The electrical energy is conducted in water or an aqueous solution of salt through migration and subsequent neutralization of ions at the oppositely charged electrodes. Therefore, it is not clear
- Why and how ions approach the electrically neutral Graphite Rods GrC & GrD (in experiments 1& 2)?
- What makes ions approach Gr3 & Gr4 , in experiment 4, for both the conditions 1 &2 irrespective of the electrical nature of Gr3 & Gr4 (i.e. how do anions move to both Gr 4 & Gr3 by reversing the placement of Gr1 & Gr). In other words, if Gr4 acted as the anode to receive electrons from Gr2 in condition1, then how did the same graphite Gr4 act as cathode to release electrons in condition2?
Under a potential gradient, cations move toward the negative electrode or cathode and anions move toward the positive electrode or anode. When the cations reach the cathode, they get neutralized and neutral molecules are formed there. Similarly, when the anions reach the anode they get neutralized by giving up their negative charge and become neutral. This shows that electrolysis occurs only at the electrodes.
- As per existing theory, electrical conduction between GrA & GrB may be regarded as owing to the migration and neutralization of ions ,as ions have sufficient opportunity for migrating to the oppositely charged electrodes (cations towards cathode GrB and anions towards anode GrA) and getting neutralized by accepting or releasing electrons, but approach of any ions to the electrically neutral rods GrC & GrD and its subsequent neutralization by accepting or releasing electrons is quite astonishing. Thus, it is not clear how such rods GrC & GrD give up or accept electrons having no charge, i.e. in contradiction to Arrhenius theory wherein it is stated that electrolysis occurs only at the electrodes ( i.e. at the cathode and anode where negative and positive terminals of a battery are connected respectively) with neutralization of ions by acceptance of electrons by cations at cathode and release of electrons by anions at the anode.
- The current in the micro ampere range suggests that about 1012 electrons flow through the circuit per second and in doing so 1012 H+ ions are to be neutralized at GrD (behaving as cathode for lighting the LED) and 1012 OH– ions are to be neutralized at GrC (behaving as anode for lighting the LED) i.e. 0.5×1012 molecules of hydrogen will be formed at electrically neutral rod GrD and 0.25×1012 molecules of oxygen will be formed at electrically neutral rod GrC. It means that ions can be neutralized and hydrogen gas and oxygen gas can be obtained even at electrically neutral graphite rods, other than actual cathode and anode where emf is applied from external source, in contradiction to the Arrhenius theory.
- Why and how Gr D, in experiments 1 &2, happen to be cathodes and supply electrons to the LED, i.e. how ions prefer electrically neutral graphite rods to make them behave as cathodes or anodes remains undecided.
- It appears that in experiment 3, for conduction of electrical energy between GrE and GrF, cations move to the cathode (GrF, negative electrode) and get neutralized there while anions get neutralized at anode (GrE, positive electrode).It further suggest that H2 and O2 are obtained at the cathode and anode respectively, i.e. electrolysis of water is possible even at any potential below its decomposition potential of 1.23volts!
- The electrolytic solution at any instant remains neutral even if the number of positively and negatively charged particles are unequal in the solution. This is due to the fact that the total positive charge carried by the cations is equal to the net negative charge carried by the anions.It is again not clear that when it is not possible for ions to approach and get neutralized at the electrically neutral graphite rods (as shown in experiments 1,2 &4) ,while conducting electrical current, how then the total positively charge carried by the cations is equal to the net negative charge carried by the anions.
Explanations for conduction of electrical current in water:
It is clear that although the current is carried through the metallic part of the circuit by the flow of electrons, for a flow of current through the electrolyte some process must occur at the surface of the cathode in the electrolyte which removes electrons, while at the anode surface electrons must be supplied. It is believed that cations receive electrons from the cathode and the anions give up their electrons to the anode, i.e. the electrons pass from the cathode to the solution and from the solution to the anode. But how then does the conduction of electrons takes place through the solution in between the electrodes (i.e. cathode to anode) ?
Now, to help resolve mysteries of electrical conduction in water we suggest the
following plausible mechanism:
At the cathode (–ve electrode):
Step 1: Self-ionization of water
Step 2: Reduction (i.e. gain of electrons) of hydronium ion at cathode
2H3O+ + 2e → 2H2O + H2 ↑
As the water undergoes self ionization 4 the inflow of electrons from the external source of emf helps neutralize the positive charge on the hydronium ion (H3O+) at the cathode and formation of water and hydrogen gas. But the presence of OH– ions formed at step 1 indicate the presence of some definite excess negative charge there. This excess negative charge is retained at the zone near the cathode, due to inflow of electrons from the external source, may be regarded as hydrated electrons.
At the anode (+ve electrode):
Step 1: Self-ionization of water
Step 2: Oxidation (i.e. loss of electrons) of hydroxyl ion at anode
4OH–– 4e →4OH → 2H2O + O2 ↑
The loss of electrons to the external source of emf by hydroxyl ion (OH–) at the anode results in neutralization of the negative charge on the hydroxyl ion and helps formation of water and oxygen gas. But the presence of H3O+ ions formed at step 1 indicates the presence of some definite excess positive charge there. This excess positive charge at the zone near the anode, due to outflow of electrons to the external source, may be regarded as hydrated protons .
Thus, in water negative charges on OH– ion that remained counterbalanced by the presence of positive charges on hydronium ions (H3O+), water being electrically neutral, becomes bare due to reduction of hydronium ions (H3O+) by the electrons from the external source at the cathode. The bare OH– ions, stabilized by water molecules, act as hydrated electrons. Thus, conduction of electrical energy between two electrodes (cathode & anode ) in distilled water is due to delocalization of hydrated electrons through molecular orientations of water.
Moreover, as water at any instant is electrically neutral, the number of electrons gained by water at the negative electrode zone remains equal to the electrons lost by water at the positive electrode zone.
Explanations for conduction of electrical current in dilute aqueous salt solution:
On passing of electrical current through a dilute aqueous solution of sodium chloride (NaCl) it is seen that neither Na metal is found at the cathode nor Chlorine gas is obtained at the anode but still it’s presence in solution increases the conductivity of the salt solution (as found in experiments 1& 2). This shows that though Na+ ions or Cl– ions do not get neutralized at the cathode or anode during electrical conduction they yet help in extensive electrical conduction.
Now, to explain such extensive electrical conduction in the presence of salt, we
suggest a similar mechanism for electrical conduction in aqueous salt solutions.
At the cathode (–ve electrode):
Step 1: Self-ionization of water
Step 2: Ionization of water due to Salt
The addition of NaCl helps more ionization of water molecules as evident from more electrolysis of water molecules in the presence of a small quantity of acid, base or salts ( i.e. increase in rate of evolution of hydrogen and oxygen gas in aq NaCl than in distilled water)
Step 3 : Reduction (i.e. addition of electrons) of hydronium ion at cathode
2H3O+ + 2e → 2H2O + H2 ↑
At the anode (+ve electrode):
Step 1: Self-ionization of water
Step 2: Ionization of water due to Salt
(The addition of NaCl helps more ionization of water molecules as stated above)
Step 3: Oxidation (i.e. loss of electrons) of the hydroxyl ion at the anode
4OH—– 4e →4OH → 2H2O + O2 ↑
The presence of ions in solutions helps conduction of more electrical energy as explained below:
- The presence of ions shifts the equilibrium of self ionization of water towards the right: The presence of Na+ ions and Cl– ions in solution shifts the equilibrium of self ionization of water (i.e. step 1) to the right thereby causing more ionization of water. As more hydronium ions are made available by the salt, more hydronium ions get neutralized by the electrons from the external source, definitely helping increase the number of bare OH– ions and in other words hydrated electrons that help more conduction of electrical energy.
- The effect of solvent on the degree of dissociation of a given substance :
As the nature of solvent plays an important role in determining the degree of dissociation of a given substance, it hence plays an important role in deciding whether the solution shall behave as a strong or weak electrolyte. The higher the dielectric constant of the medium ,the smaller is the electrostatic attraction between the ions and greater is the probability of their existence in the free state5 This means that the solvent plays a very important role both in formation and stabilization of ions of the solute in solution. Thus, in dilute sodium chloride solution, water not only helps formation of ions of sodium chloride but its stabilization through hydration. The stabilization of ions by hydration, i.e. involvement/engagement of water molecules, in turn stabilizes molecular orientations of water by restricting random orientations of water molecules and this helps more conduction of electrical energy through the stabilized orientations of water.
It therefore implies that:
- Self-ionization of water is only the dissociation of water into hydrated protons (H3O+) & hydrated electrons (OH–), water being electrically neutral.
- Accumulation of hydrated electrons near the cathode makes it negative zone while hydrated protons near the anode make it behave as positive zone. In the absence of GrC &GrD the hydrated electrons are delocalized through molecular orientations of water from its zone of higher concentration (near Cathode, GrB) to the zone of lower concentration (near Anode GrA) thereby neutralizing hydrated protons. But in the presence of GrC &GrD , the resistance to flow of electrons in graphite being lower than in water. the hydrated electrons prefer to flow from its Zone of higher concentration (near Cathode GrB) to the zone of lower concentration(near Anode GrA) through graphite rods GrC & GrD lighting the LED as found in experiments 1 & 2. Thus, it is now clear that electrical energy between two electrodes is not carried by ions, as formerly believed, but hydrated electrons having resemblance to conduction of electrical energy through delocalization of valence electrons in metals and pi-electrons in graphite (non-metal).
Inference:
Delocalization of hydrated electrons through molecular orientations of water conduct electrical energy in water or in aqueous salt solutions.
Ref:
[1] Avogadro’s Number Does Not Limit the Efficacy of Homeopathic Remedies Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, Feb2015hpathy.com
[2] Delocalization of Electrons Through Orientations of Water Molecules Helps Electrical Conduction In Water,Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, March 2014hpathy.com
[3] Untold Facts of Science –The Science Behind Homeopathy,Ruhul Amin &BiplabChakraborty(Published in Homeopathy for Everyone, May2012hpathy.com
[4] A New Concise Inorganic Chemistry ( Third Edition) -JD LEE
[5] An Introduction to Electrochemistry- Samuel Glasstone