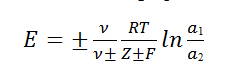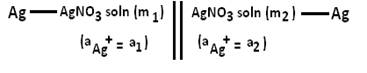Electrical potentials exist across the membranes of all cells of the body. This is believed to be due to the difference in concentration of important electrolytes and other substances in the extracellular fluid and intracellular fluid. The extracellular fluid contains a large amount of sodium ions (142mEq/L) but only a small amount of potassium ions (4mEq/L).The concentration of sodium and potassium is just opposite, the intracellular fluid being 14mEq/L and 140mEq/L respectively .In case of anions, chloride (103mEq/L) & bicarbonate(28mEq/L) dominate the extracellular region, whereas phosphates (75mEq/L) and sulphates (2mEq/l) are in excess in the intracellular fluid. Such difference of concentration of ions across the cell membrane is thought to be responsible for establishment of the normal resting membrane potential of –90millivolts, owing to the combined effects of the following factors1
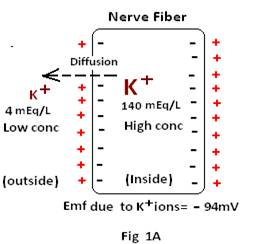
i) Potential arising due to diffusion of Potassium ions: If the resting membrane potential is solely due to the diffusion of K+ ion (Fig 1A) then the potential (emf) may be calculated from the Nernst equation, with the assumptions as shown below.
Assumptions: a) that the potential in the extracellular fluid remains at zero potential and the Nernst potential is the potential inside the membrane.
b) that the emf is said to be positive if the ion diffusing from inside to outside is a negative (–ve) ion and the emf is said to be negative if the ion is a positive (+ve) ion.
The following equation is used to calculate the Nernst potential for any monovalent ion at 370C .
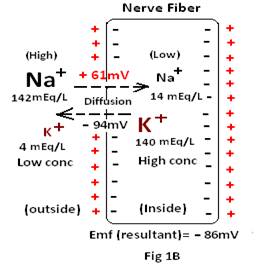
ii) Potential arising due to diffusion of Sodium ions: As the sodium ions diffuse from its region of higher concentration to lower concentration i.e; from the extracellular region to inside the cell, the calculated Nernst potential inside the fiber, for Na+ ion, would be +61mV {emf= − 61 =− 61 (1.1461− 2.1523) =− 61(−1.0062)= +61.3782 mV}. But apart from K+ & Na+ there are many others ions (cations/anions) which have different concentrations in the extracellular and intracellular regions. As the membrane is permeable to different ions, emf is calculated, using the Goldman equation , and found to be -86mV which is quite close to the potential developed due to the potassium ions.
iii) Potential due to NA+– K+ Pump: It arises due to the Electrogenic nature of the NA+– K+ pump (Fig1C). The said pump moves three Na+ ions to the outside for every two K+ ions to the inside, through a complex of two separate globular proteins called carrier proteins. Binding of two K+ ions (at extracellular region) and three Na+ ions (at intracellular region ) with the carrier protein activates the ATPase function of the protein (i.e: enzymatic action of ATPase). Subsequently, due to the activity of the ATPase, energy in the form of inorganic phosphate (iP) is liberated along with ADP by the cleaving of an ATP molecule. It is believed that the said liberated energy helps passage for Na+ ions to the outside and K+ to the inside (i.e: against the concentration gradient) through change in chemical and conformational change in the carrier protein molecule. This results in positivity outside and negativity inside the cell. The electric potential so obtained across the cell membrane by the NA+– K+ pump is therefore, termed to be electrogenic. The fact that more Na+ ions are being pumped outside than K+ ion to the inside causes continual loss of positive charges from inside the membrane creating an additional degree of negativity (about –4mV) on the inside beyond what can be attributed due to the diffusion alone.
Thus the net membrane potential (–90mV) is the sum of resultant diffusion potential of K+ ion and Na+ion (–86mV) and the electrogenic potential generated due to the NA+– K+ pump (–4mV).
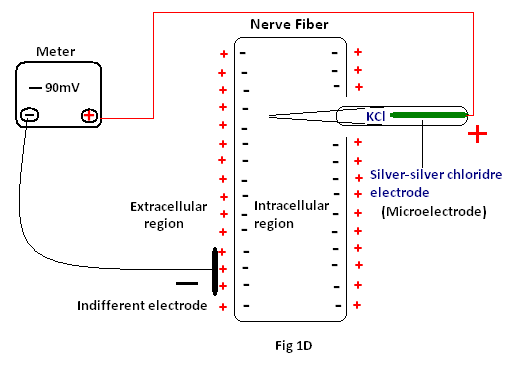
Measurement of cell membrane potential: Owing to small size of most of the fibers while measuring the membrane potential, a micro electrode consisting of a micro pipette filled with an electrolyte solution (Silver –silver chloride solution) is used. The micro pipette is inserted through the cell membrane to the interior of the fiber, then another electrode called the “indifferent electrode” is placed in the extracellular fluid , and the potential difference between the inside and outside of the fiber is measured using a sophisticated voltmeter(Fig 1D). As long as the micro electrode is outside the nerve membrane, the potential recorded is zero (i.e; emf=0), which is known as the potential of the extracellular fluid. But as the microelectrode passes through the voltage change area at the cell membrane, the potential decreases abruptly to –90mV and remains steady at the center1.
Actually, to create a negative potential inside the membrane, only enough positive ions to develop the electrical dipole layer at the membrane itself must be transported outward. An incredibly small number of ions must be transferred through the membrane to establish the normal “resting membrane potential” of –90mV inside the nerve fiber, and thus only about 1/3,000,000 to 1/100,000,000 of the total positive charges inside the fiber must be transferred.
It is said that so long as the membrane of the nerve fiber remains completely undisturbed , the membrane potential remains at –90mV (approx),which is called the resting potential. However, any factor that suddenly increases the permeability of the membrane to sodium is likely to bring a sequence of rapid changes in membrane potential lasting a minute fraction of a second ,followed immediately thereafter by return of the membrane potential to its resting value . This sequence of potential changes is called the action potential. The stimulation may be electrical, chemical, mechanical ,temperature or any factor that disturbs the normal resting state of a membrane temporarily.
Now, the question is:
i) Why is the resting membrane potential always regarded as negative around –90mV ?
ii) How does the potential develops across the membrane (i.e; emf is generated across the membrane of a cell).
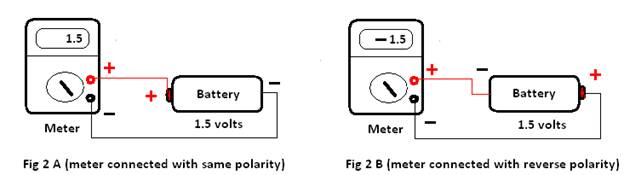
For measuring emf of a battery or electrochemical cell we always get positive emf if electrodes of the meter are properly connected to the suitable end of the battery or cell (i.e; positive electrode to the positive end of the battery and negative electrode to the negative end). In the case of a living cell, when the cell is at rest, the inner side of the membrane has a slight excess of anions (i.e negative charge) while the outer side has some excess of cations. Therefore, it should behave like a battery with its positive terminal outside the membrane and negative inside the membrane. On properly connecting a meter with its electrode (Fig 2A & 2B) it should show some positive emf. But it is stated that it shows a negative emf (Fig 1D)!
Now to see how the potential actually develops across the cell membrane, let us consider the following simple experiments:
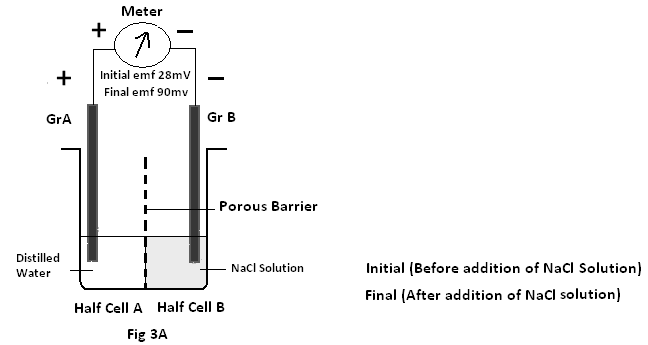
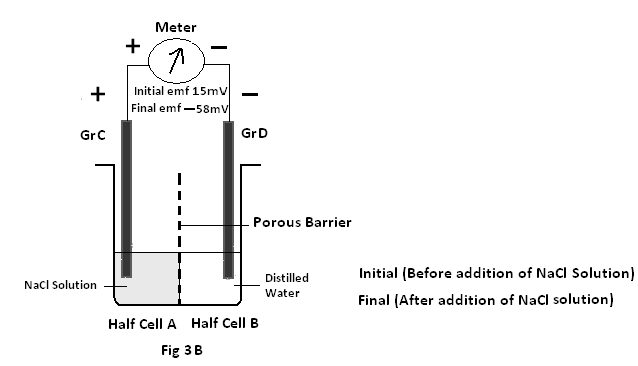
Experiment 1: A cell is prepared consisting of two half cells separated by a porous barrier with both half cells with Graphite (Gr) electrodes immersed in distilled water.
Case A: Where about 2.5 ml of NaCl solution (25% sodium chloride) is added to the negative end – i.e. half cell B. The resultant emf is found to increase from 28 mV to 90mV showing an increase in emf of about (90-28=62 mV) for GrA-GrB pair.(Fig 3A)
Case B: Where NaCl solution is added to the positive end – i.e half cell A. The resultant emf is found to decrease from 15mV to –58mV (minus 58millivolts) showing a decrease in emf of about (-58-15= – 73 mV) for GrC-GrD pair.(Fig 3B)
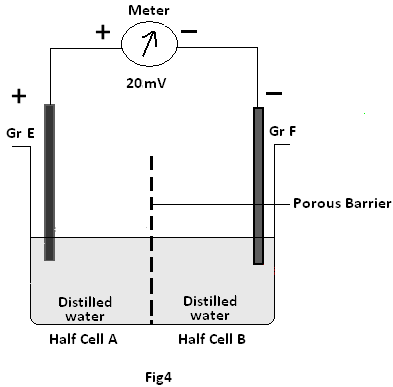
Experiment 2; In this experiment, both the half cells separated by a porous barrier contain distilled water. Yet still the Graphite pair (GrE –GrF) are found to generate emf of about 20mV.
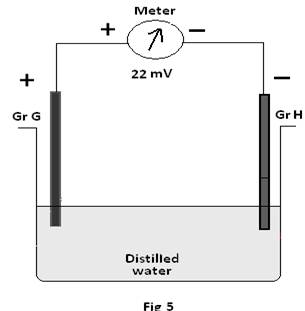
Experiment 3: Both the Graphites (G &H) are immersed in a beaker containing distilled water(i.e; without any porous barrier) and still generates some emf. The emf is found to be about 22mV.
Moreover, distilled water is found to generate different emf with different pairs of electrodes as shown below2.
| Sl no. | Pair of electrodes | Emf generated in distilled water (in mV) |
| 1 | Gr – Cu | 300 |
| 2 | Gr – Zn | 1098 |
| 3 | Gr – Au | 160 |
| 4 | Gr – Ag | 220 |
| 5 | Gr – Pb | 742 |
Discussion: From the above experiments it is found that:
i) Any pairs of electrodes (metal or non-metal such as graphite) can generate emf when partly immersed in distilled water (separated by a porous barrier or otherwise) in spite of having no reaction with water. This generation of emf is due to the rearrangement of dipoles of water molecules2,3,4.
ii) It is found in experiment 1 case A that on addition of some NaCl solution to the half cell B (i.e negative electrode) the resultant emf is increased from28mV to 90mV . But when the NaCl solution is added to the half cell A (i.e positive electrode) in experiment 1(case B),the resultant emf is decreased from15mV to –58mV . It is interesting to note in experiment 1(case B) the positive electrode (i.e GrC) placed in the half cell A started to behave as a negative electrode. So it can be said that the half cell of a cell containing more ions always acts as the negative end of a cell4.
iii) As graphite has no reaction with either distilled water or NaCl , the increase in emf on addition of NaCl solution may be attributed to the presence of extra ions (Na+ & Cl–) in distilled water.
iv) When any pair of electrodes (metal or non-metal, such as graphite) are able to generate emf even in distilled water , it is obvious that all substances which contains water generate emf only due to water, and the presence of other ions only helps to increase or decrease the measure of emf depending on the presence or absence of any porous barrier.
v) Like distilled water, Cell membrane potential will also vary depending on the electrodes used, as here also the generation of emf is due to the presence of water. Other ions (Na+,K+ Cl–, organic phosphates, suphates, etc) only help to increase or decrease the resultant emf.
vi) In experiment 2 as two graphite electrodes immersed in distilled water separated by a porous barrier generate emf like a cell membrane potential, this may be termed Porous Barrier Potential !
vii) It is well known that in an Electrolyte concentration cell without transference there is no direct transfer of electrolyte from one solution to the other .This type of cell is formed whenever two simple cells with each electrode reversible with respect to one of the ions of the electrolyte, containing solutions of different concentrations are combined .The general equation for the emf of any concentration cell without transference can be obtained from the following equation:
Where E=emf of the cell, R= Gas constant (8.3143 J), F(in coulombs) = 96500, ln=2.303log, a1 &a2 are activities of the reversible ion in the solution.
Moreover, is the total number and the number of positive or negative ions produced by the ionization of one molecule of electrolyte, and Z+ or Z- is the valence of the ion with respect to which the extreme electrodes are reversible 5,6.
Thus for the any electrolyte concentration cell, the electrodes must be dipped into solutions differing in the concentrations of the reversible ion. I.e for the cell we have Silver electrodes (Ag) dipped into Silver nitrate solution containing reversible silver ion Ag+ .
From the above, it is seen that the Nernst equation can be used to calculate the emf of a cell with electrodes reversible to one of the ions of the electrolyte. But, it is not clear how potentials due to Na+ or K+ ions present in the electrolytes (extracellular & intracellular fluids) are calculated using the Nernst equation, when there is no sodium or potassium (amalgamated) electrode used and instead one “indifferent electrode” & one micro electrode (Silver –silver chloride electrode) are used for measuring membrane potential of a cell (Fig 1D) ?
Thus, we are of the opinion that pure water, the electrical energy carrier in homeopathic remedies7, duly influenced by the presence of ions in extracellular and intracellular regions, is responsible for the generation of emf or membrane potential of a cell.
Inference: Cell membrane potential therefore, is actually the result of rearrangement of orientations of water molecules influenced by the presence of other ions (simple & complex).
References:
[1] Textbook of Medical Physiology (Twelfth Edition) (Reprinted 2012)-Guyton and Hall
[2]. Homeopathic Individualization-in the light of Electrode potential (Published in Homeopathy for Everyone June 2012 hpathy.com
[3]. Water the Magic Liquid: Retaining Electrical Energy, Ruhul Amin &
Biplab Chakraborty (Published in Homeopathy for Everyone April 2012
[4] Generation of electrode Potential of an electrode –An explanation , Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXIII.2010, www.hanp.net)
[5] Physical Chemistry, P C Rakshit ( Revised 4th Edition) 1980, Published by S C Rakshit, Kolkata , India
[6] Physical Chemistry, S Glasstone, 2nd Edition, 1946
[7]. The electrical energy of substances –the secret of homeopathy, Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXII, Summer Fall/ 2009. www.hanp.net)