The cause of retention of electrical energy by water, the key to Homeopathic medicines1, has so far remained unexplained. Sometimes it is referred to as simply electrolysis. In our earlier papers we have shown such retention by applying external emf, which was above the decomposition potential of water i.e, 1.23 volts. In our later work we have shown the same result both for the human body and water, by applying external emf even well below the decomposition potential of water2. Now we provide some interesting results to show that retention of electrical energy in water is possible even without application of any external emf, which further explains the cause of retention of electrical energy by water.
Experiment 1:
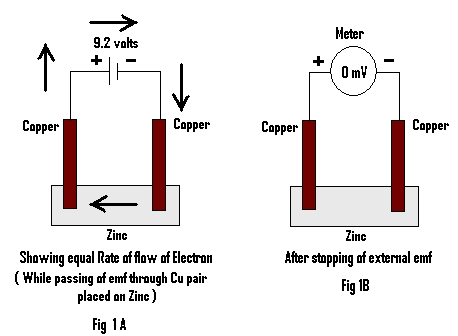
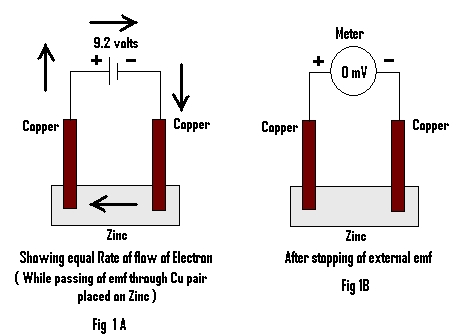
The emf of about 9 volts from an external source was allowed to pass for 3 minutes through a pair of Copper electrodes (Cu) (Cu A & Cu B) placed on a piece of metal (here Zn is used) Fig 1A.
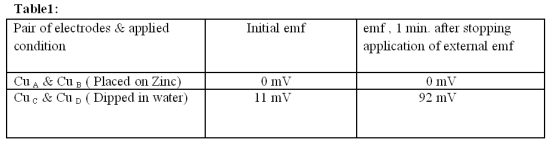
One minute after stopping application of externally applied emf, the emf generated by the metal through two Copper electrodes is measured and noted in table 1.
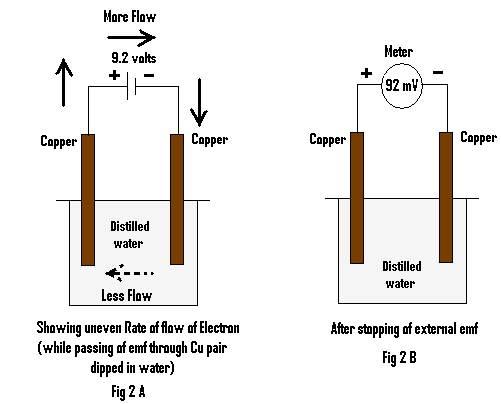
This experiment is repeated by replacing the metal with pure/distilled water i.e, a pair of Copper electrodes (Cu) (Cu C & Cu D) are dipped into pure/distilled water Fig 2A.
Emf from an external source is applied for 3 minutes and emf of the Cu pair is measured at one minute, after stopping application of externally applied emf (noted in table 1.)
Discussion:
When an external emf of about 9 volts is passed through a piece of metal (here Zn) using copper electrodes for about 3 minutes and the emf generated by the metal through two copper electrodes is measured, it is found to be Zero (Fig 1B). The metal (here Zn) is then replaced by pure/distilled water and after passing of same external emf for the same time through the pair of Cu electrodes, the emf generated by the same is measured. It is found to be about 92 milli volts (mV).
(Fig 2B) Both metal and water being conductors, the question arises, why and how did the latter retain electrical energy. Both are conductors, but yet there is a major difference between the two. The former is the very good conductor while the latter being poor. In case of metal (here Zn), actually due to high electrical conductivity, the rate of flow of electrons through the entire circuit remains almost the same at any given instant while in the case of water, which is poor conductor of electrical charges, the rate of flow of electrons through the outer circuit is more than the flow in the inner circuit (i.e: through water )
Fig 2A This uneven flow of electrons in the inner circuit results in the accumulation of excess electrons at one end, while a dearth at the other end, resulting in retention and hence generation of emf. In the case of metal due to its high conductivity, two different ends with excess and dearth of electrons hardly can exist. But for water, having poor electrical conductivity, there is every possibility for the existence of such different zones within water.
Experiment 2:
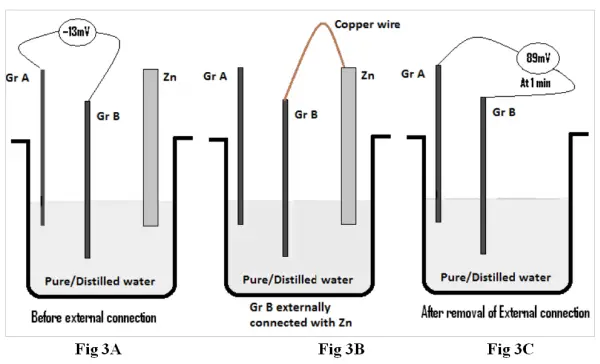
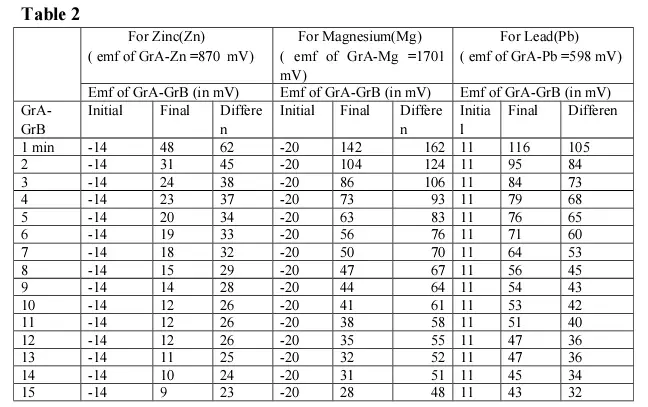
In this experiment a pair of graphite electrodes (GrA & GrB) & a Zinc (Zn) electrode are simultaneously dipped in pure /distilled water. The emf’s of Gr A – Zn and GrA – GrB is measured and recorded in table 2 Fig 3A.Thereafter the Zn electrode is connected externally with a metal wire with the graphite electrode GrB (Fig 3B).This process is allowed to continue for 3 minutes and thereafter both the external connection and Zinc electrode are removed (Fig 3C). The emf generated by the pair of graphite electrodes (after removal of external wire and Zn electrode) are recorded in table 2 at intervals from one minute up to 15 minutes and are then plotted against time.
The Zn is replaced by Mg , & Pb electrodes and the readings are recorded in table 2.
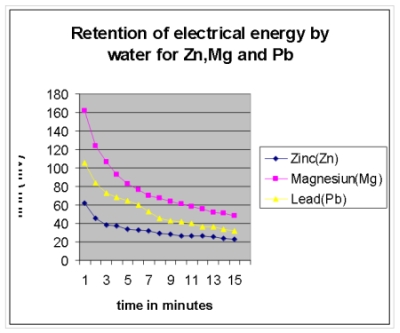
A graph is also plotted with the above result:
Discussions:
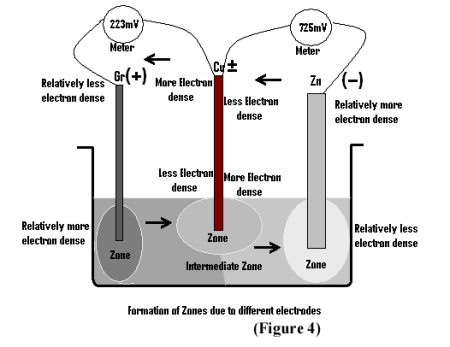
It was already stated in our earlier papers, that whenever any metal or non-metal (graphite) is dipped into pure/distilled water, a potential is generated at the non-immersed portion of the electrode due to repulsions of electrons of water molecules and that of electrodes 3,6. The difference between the electrode potential of two electrodes thus generates emf . This is also true for the case when more than two electrodes are dipped into water simultaneously and each of the upper ends (i.e; non-immersed portion) generate electrode potential even without any chemical reaction 4 . It reflects the fact that it is none but the re-arrangement of water molecules that is responsible for generating such electrode potential. For this new arrangement of water molecules some strain is invariably generated within water. Moreover, as different electrodes generate different potential, it is obvious that for each electrode the strain in the water is specific, resulting in specific arrangement of water molecules. In other words specific Zones are formed in water for different electrodes (as shown in Fig 4) while the rest of the areas are intermediate in nature. These Zones actually depends upon the density of electrons at the immersed ends of the electrodes. In order to gain stability by minimizing its strain, conduction of electrical energy between various zones starts within the water. But being a poor conductor of electrical energy, it takes different times to regain its initial orientation, for different pairs of electrodes. The graphical representation however once again confirms that the rate of drop of emf is sharp at the beginning, while the curve is almost parallel to the X-axis in the later stage in respect of all the metals i.e; Zn, Mg or Pb.
Experiment 3
a). The experiment no. 2 is repeated replacing pure/distilled water by sugar solution in the case of the Zn electrode.
b). The same experiment is repeated by salt solution (sodium chloride solution) in place of the sugar solution, in the case of the Zn electrode.
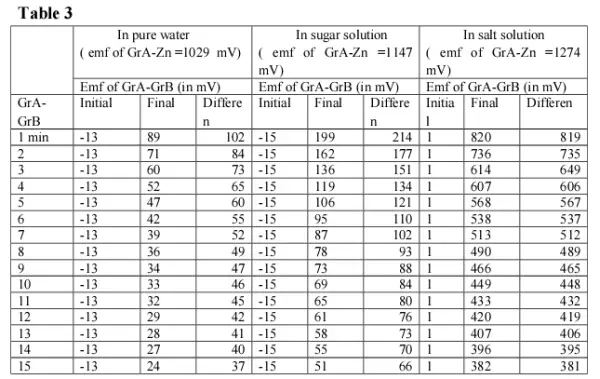
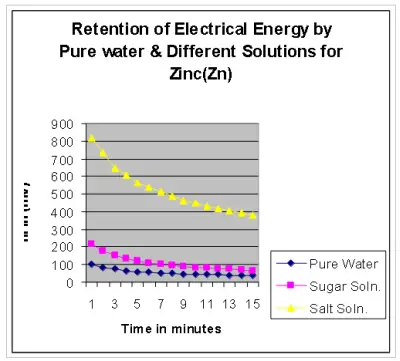
The results for Zinc in pure water, in sugar solution and in salt solution are recorded in table below(Table 3).
Discussion:
Had the ions present in salt solutions conducted electrical energy (i.e, facilitates flow of electrons ), then the emf generated in case of the salt solution due to retention would have been much lower than in pure water5. It is rather much higher than in pure water. This again raises a question whether ions conduct electrical energy in solution. Also in case of the sugar solution, which does not render any other ions in solution, the emf generated due to retention is found to be higher than in pure water. This suggest that the presence of other ions in solution is not the reason for retention of electrical energy, but there must be some force that restricts the flow of electrical energy through salt or sugar solution 5. We have already stated in our earlier paper that whenever any substance is introduced into water, a strain is developed 7 and water molecules rearrange themselves to minimize the strain. This strain results in the generation of emf . So, whenever any strain is produced in pure water, its molecules rearrange themselves to minimize the strain. But whenever any strain is produced in water containing salt or sugar molecules, the latter restricts rearrangement of the water molecules, thereby stabilizing the new orientations produced by the strain. That is why in case of salt & sugar solutions, the flow of electrical charge within water is restricted, resulting in higher retention of electrical energy.
Inferences:
i) Water retains electrical energy due to its poor conductivity.
ii) Different zones are formed within water due to the presence of
different electrodes. The relative difference in energy content of these
zones is responsible for the flow of electrical energy when connected
externally.
iii) Ions in salt solutions are poor conductors of electrical energy, but rather stabilize orientations of water molecules due to which the salt solution retains more electrical energy than pure water or sugar solution.
iv) The retention of electrical energy by water is purely due to rearrangement of water molecules, the key factor to the Homeopathic remedies.
Reference:
[1]. The Electrical Energy of Substances –The Secret of Homeopathy, Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXII, Summer Fall/ 2009 www.hanp.net)
[2] The Human Body And Water Both Retain Electrical Energy Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone Sep2012 hpathy.com
[3] Generation of electrode Potential of an electrode –An Explanation, Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXIII.2010, www.hanp.net)
[4] The Flow Of Electrical Energy Through Metal And Water-A new concept Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone, Aug 2012 hpathy.com
[5]. Solutions Ionic or Non-ionic Conduct & Retain Electrical Energy Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone, July 2012 hpathy.com)
[6]Water the Magic Liquid: Retaining Electrical Energy, Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone April 2012, hpathy.com
[7] The Homeopathic Dilution-A New Explanation, Ruhul Amin, Biplab Chakraborty & Farook Rahaman (Simillimum,vol.XXI,Summer Fall/2008. www.hanp.net