Overview: This is the seventh paper of the series on a scientific view of Homeopathy. In our previous works we demonstrated that the responsible factor in homeopathic remedies is the electrical energy content of the remedies, which is different in every remedy (produced by different substances) and also different in every potency of each remedy 4,5. That the difference of energy content is governed by the orientation of dipoles of water molecules, has already been proven by various experiments .The main force responsible for this phenomenon, repulsions of electrons, is further proved by our latest work
Abstract: The electrochemical series, as defined in science, is obtained by arranging the elements in order of their standard electrode potentials. The standard electrode potential of an electrode (metal/non-metal) immersed in a solution containing its own ions is obtained by introducing the Hydrogen electrode in the solution and measuring the difference in potential thereof. [ In hydrogen electrode a Platinized Platinum electrode is saturated with hydrogen at one atmosphere pressure and immersed in a solution of H+ at unit activity. The potential developed is arbitrarily fixed at Zero]. The chemical reaction at electrode being represented by Mn+ + ne = M
Herein we also obtained the same result with the available metals, even with graphite as a reference electrode, without any chemical reactions. Here we show that the repulsion of electrons is the main factor that allows these results.
We also demonstrate that the mechanism of generation of emf of an electrochemical cell, with or without chemical reactions, is the result of repulsion of electrons.
In an electrochemical cell, with chemical reactions, the chemical reactions only provide more electrons to flow through a conductor in unit time.
Experiment 1: An experiment is performed by immersing the following electrodes in a beaker containing distilled water. The emf of different electrode pairs is measured separately and readings recorded below. The same experiment is done by holding the electrodes in the fingers (i.e human body) and noting the emf thereof in the table below:
Table:1
| Sl No. | Electrode pairs | Observed emf (in mV)In Distilled water | Observed emf (in mV)In human body | |
| (A) | (B) | |||
| 1 | Gr – Mg (Graphite – Magnesium) | 1970 | 1180 | 1500 |
| 2 | Gr – Zn (Graphite – Zinc) | 1150 | 740 | 900 |
| 3 | Gr – Pb (Graphite – Lead) | 778 | 534 | 600 |
| 4 | Gr – Cu (Graphite – Copper) | 240 | 250 | 270 |
| 5 | Gr – Ag (Graphite – Silver) | 230 | 240 | 262 |
| 6 | Gr – Au (Graphite – Gold) | 101 | 210 | 220 |
| 7 | Au – Zn (Gold – Zinc) | 920 | 500 | 700 |
| 8 | Ag – Zn (Silver – Zinc) | 960 | 548 | 735 |
| 9 | Au – Ag (Gold – Silver) | 75 | 30 | 50 |
| 10 | Ag – Cu (Silver – Copper) | 25 | 44 | 85 |
| 11 | Au – Cu (Gold – Copper) | 65 | 77 | 90 |
| 12 | Cu – Zn (Copper – Zinc) | 850 | 490 | 596 |
Case 1: Let us consider a irreversible cell Zn | H2SO4 | Cu wherein Zn and Cu metals are dipped in sulphuric acid thereby generating emf of about 1 volt. In this cell “1” Faraday of electricity is produced when 1 gm-equivalent of zinc goes into solution (at –ve electrode) and 1 gm-equivalent hydrogen is liberated at the positive electrode, Cu (copper) i.e: emf is produced due to a chemical reaction at the cost of the zinc electrode. This cell made up of Cu & Zn electrodes still generates sufficient emf when Sulphuric acid is replaced by distilled water and the human body as shown at sl no. 12 of Table: 1 above. The question now arises, how does this cell generate emf with distilled water and contact with the human body – whether Zn still goes into distilled water and the human body for such generation!
Moreover generation of emf through chemical reactions cannot explain the other results as obtained at sl. No. 1 to 11, as there is hardly any possibility for Graphite, Zinc, Copper, Silver, Lead and Gold etc. to go into distilled water and the human body for generation of emf.
Case 2: Consider the Leclanche Cell, wherein the Zn plate is the –ve electrode and graphite is the +ve electrode . The electrolyte being a paste of MnO2, NH4Cl and ZnCl2.
Here, also the emf of about 1.5 volts is generated at the cost of the Zn electode as shown by the following reactions:
At the –ve electrode :Zn+ 2MnO2+ H2O→ Zn++ +Mn2O3 + 2OH –
At the +ve electrode : 2MnO2 + H2O +2e → Mn2O3+ 2OH–
Subsequently, secondary chemical reactions occur which consume the OH– and Zn++ through NH4Cl as :
2NH4Cl+ 2OH– = 2NH3+2Cl– +2H2O
Zn++ + 2NH3+2Cl– = Zn(NH3) 2Cl2
This cell made up of Carbon (Gr) & Zn electrodes still generates sufficient emf when the electrolyte is replaced by distilled water & the human body as shown at sl no. 2 of Table: 1
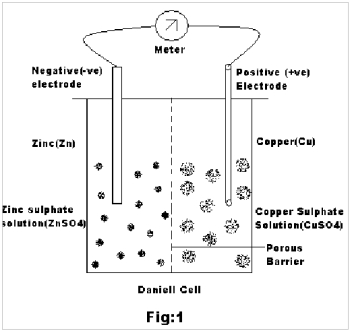
Case 3: From the above experiment it is also found that the emf of the Cu-Zn pair is about 850 mV(i.e 0.850 volts). Now let us consider the Daniell Cell2,3 wherein a zinc rod is partially immersed in ZnSO4 solution and a copper rod in CuSO4 solution as the two electrodes. The two solutions are separated by a porous barrier that allows the current to pass through but prevents mass diffusion (Fig.1).
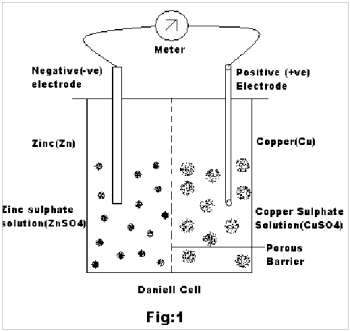
During the production of current, Zn atoms pass into solution as Zn++ ions leaving behind two electrons each in the metal electrode. On the other hand, copper ions from the solution are deposited on the copper electrode by accepting two electrons each from the metal. Thus there is an excess of electrons in the Zn electrode and deficit of the same at the Cu electrode. When the circuit is closed, the electrons from Zn flow to the copper through the wire to feed the copper ions in the process of its discharge.
The Zinc electrode, where zinc gives up its electrons, i.e Oxidation occurs, is the negative electrode of the cell. The copper electrode , where copper ions take up electrons, i.e reduction occurs, is the positive electrode.
The cell being represented as:
– +
Zn | ZnSO4 : CuSO4 | Cu
The reaction at – ve electrode is: Zn= Zn++ + 2e (i.e: Loss of electrons or oxidation occurs)
The reaction at + ve electrode is :Cu++ + 2e= Cu (i.e: Gain of electrons or reduction occurs)
The total cell reaction being: Zn +Cu++ =Zn+++Cu
So when 2 coulombs of electricity are produced, 65/96500 gm of zinc goes into solution at the –ve electrode and 63.5/96500 gm of copper is deposited at the +ve electrode of the cell. In other words, for production of 1 Faraday of electricity, 1 gm-equivalent of Zn goes into solution and 1gm-equivalent of Cu is deposited on Cu electrode. The emf of the cell is about 1.10volts. From the above it is clear that for generation of emf some amount of zinc goes into solution and copper ions are deposited on copper electrode, i.e Zn electrode becomes thin while the copper becomes thicker. Now when Cu& Zn produces a remarkable emf of about 850 mV(0.850 volts) even in distilled water, it means at least some amount of Zn must have gone into distilled water and some amount of copper ions from distilled water have got deposited on copper electrode. But as per prevailing scientific norms, neither is it possible for zinc to get dissolved in distilled water nor distilled water to supply copper ions to be deposited on the copper electrode.
Similarly, though generation of emf by Gr-Zn Pair, Gr-Cu , Gr-Ag, Gr-Au, etc. when dipped in distilled water and contacting the human body are quite appreciable than Gr-Gr, Cu-Cu etc6 ,yet none of them can be explained from the existing theories of generation of emf i.e:
- chemical reactions ( since none of them react with distilled water)
- transfer of matter from one half cell to the other half cell, due to concentration difference ( as here no question of transfer of matter arises as both the electrodes are dipped in the same distilled water without any porous barrier).
Now, the question arises regarding the source of generation of emf. We have already shown previously that the emf generated in distilled water can run a calculator or LED1 .
A calculator rating with power, P=0.0004W & 3 volts was run using electrodes dipped in distilled water (i.e: a case without chemical reaction).
Power, P=4X10-4 watts
=4X10-4 joules/sec [ Since we know 1watt=1joule/sec]……………….. (1)
Also, P=V.I [ where P=Power, V= potential difference in volts, I=current in ampere]
∴ I= P/V =(4X10-4)/3
= 1.3333 X10-4 A (ampere)
Now 1ampere=1coulomb/sec and charge of electron, e=1.602X10-19 coulomb
i.e; 1.602X10-19 coulomb =1e (electron)
∴ 1coulomb=1/(1.602X10-19 )e = 6.25X1018 e
[ The current in any part of a circuit is one ampere if one coulomb of charge passes through it in one second.]
So , For1ampere=6.25X1018 electrons/sec
∴ 1.3333 X10-4 ampere=6.25X1018 X 1.3333 X10-4 electrons/sec =8.333 X1014 electrons/sec……………. ………………(2)
From eqn. 1 & 2 above it is obvious that in a second 8.333 X1014 electrons conveys 4X10-4 joules of energy.
∴ 1 electron conveys energy = (4X10-4)/ (8.333 X1014)joules = 0.48 X10-18 joules or 4.8 X10-19 joules
Next, the electron volt being defined as the kinetic energy gained by an electron initially at rest, in moving through a potential difference of 1volt i.e;1eV= 1e X 1volt =(1.602X10-19 coulomb) X (1volt)
=1.602X10-19 joules
As the calculator being rated with 0.0004 watt & 3volts the electron having energy of 1.602X10-19 coulomb X 3volt = 4.806 X10-19 joules is required to make it run (i.e: when calculator is connected to a dry cell of “3 volts”, a case of chemical reaction.)
From the above calculations it is well understood that the energy conveyed by the electrons for the cases with chemical reaction are almost the same for the cases without chemical reaction. In this case the calculator was run by making a series connection with few cells ( prepared with electrodes dipped in distilled water) generating emf of about 3volts.
This, further confirms that each electron has some specific energy which depends on the emf generated by a given pair of electrodes i.e;
- for a potential difference of 1volt, energy of electron=1.602X10-19 joules
- for a potential difference of 1.5volt, energy of electron=1.602X10-19 X1.5 joules
=2.403 X10-19 joules
- for a potential difference of 0.5volt, energy of electron=1.602X10-19 X0.5 joules
=0.801 X10-19 joules etc.
Now it is already explained that generation of emf is not due to any chemical reaction, but is due to the difference in repulsion of electrons of electrodes with that of available electrons of water. Here by means of mathematical calculations we find the minimum distance between two electrons which is sufficient to produce such energy.
Due to repulsion, the electron gains Kinetic energy that is equal to work done on it.
Say, for an electron moving under potential difference of 1volt having energy of 1eV=1.602X10-19 joules and this energy being the kinetic energy of the electron, we have ½ mv2 =1.602X10-19 joules
v2=2X1.602X10-19/9.1X10-31 =0.352X1012 [ since mass of electron =9.1X10-31 K.g]
v = 5.93X 105m/s
Moreover, work done on a single electron, W= q X v
=1.602X10-19 C X 1volts
=1.602X10-19 joules
Now. Force X Distance =Work done=1.602X10-19 joules
Now the Force (due to repulsion) due to which the electron moves =Work done/distance ( length of the electrode)
length of the electrode is about 0.1 metre.
=1.602X10-19/0.1= 1.602X10-18 N.
Moreover, if Q1 and Q2 are the two point charges placed at a separation d, then according to Coulomb’s Law, the force F between them is given by the relation:
F=Q1.Q2/4пεd2, where ε=K.ε 0 ,is the permittivity of that medium and K is the relative permittivity of that medium.Here, K for water is 80.1 & ε 0= 8.85X 10-12 Coulomb2/NewtonX metre2
Or d2=Q1.Q2/4 п K. ε 0 F
=(1.602X10-19X1.602X10-19 )/ (4X3.1428X80.1X 8.85X 10-12X1.602X10-18)
=2.566404X10-38/14276.28 X10-30
=2.566404X10-38/1.427628 X10-26
=1.80145X10-12
Hence d =1.3421X10-6 m
i.e distance between the two approaching electrons (between that of electrodes and water) is about 1.3421X10-6 m for electrode pairs generating emf of 1 volt. Similarly, for emf of 0.5 volts the said distance will be 1.8981 X10-6 m & for emf of 1.5 volt it is 1.0947X10-6 m. The approach of electron is quite viable as Radius of atom is in order of10-10 m & radius of nucleus being of 10-14 m to 10-15 m.
Experiment 2: Now, let us consider an experiment wherein Gr,Cu & Zn electrodes are dipped in distilled water simultaneously (Fig. 2) and emf of the three pairs are recorded as below:
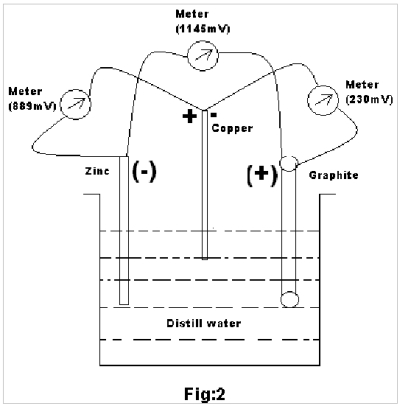
Table: 2
-
Sl. No. Pairs of electrodes Observed emf (mV) 1 Gr – Cu 230 2 Gr – Zn 1145 3 Cu – Zn 889
(1) If the generation of emf is solely due to the movement of electrons from the –ve electrode to the + ve electrode, the emf of Gr – Zn & Cu – Zn should have been the same or nearly same. But they are actually 1145 mV & 889 mV respectively showing a difference of about 256 mV, thereby suggesting that the generation of emf doesn’t solely depend on the –ve electrode Zn but also on the + ve electrodes.
(2) On measuring emf of Gr – Cu pair (in distilled water ) its emf is found to be about 230 mV thereby suggesting that here Cu acts as –ve electrode whereas with respect to Zn it acts as +ve electrode.
So it can now be said that Cu electrode behaves as +ve & -ve electrode simultaneously for Zn & Gr respectively. This suggests that electrons at Cu end should have some effect while generation of emf with Zn electrode.
Experiment 3: To confirm the effect of +ve electrode one experiment is performed by taking two Graphites marked A & B in a beaker containing distilled water. A third electrode is then introduced in shape of Zn and emf’s of all combination are measured and recorded below:
Table:3
-
Sl. No. Electrode Pairs Observed emf (in mV) 1 GrA– Zn 1175 2 GrB– Zn 1169 3 GrA– GrB 06
The same metal Zn generating different emf with different Graphites definitely suggest that generation of emf depends on the +ve electrode as well.
Discussion: Each electrode being made up of either metal or non-metal has conducting electrons (free electrons) / п electrons. When any electrode is dipped in water its conducting electrons/ п electrons are repelled by available electrons of water with different velocities and hence different energies. Now when two different electrodes are joined by a conductor, electrons from both the electrodes try to go to the opposite end. Thus by doing so the electrons moving from the +ve end to –ve end somewhat hinders/restricts the motion of electrons from –ve electrode to + electrode. As a result the energy obtained in shape of emf is the Resultant energy of electrons of both the electrodes. The generation of different emf by Zn with different Graphites is not only dependent on repulsion of electrons but also on the resistance offered by the conductor here, the electrodes. Since resistance offered by the conductor is
- directly proportional to its length &
- inversely proportional to its area of cross section, so despite being made up of the same material there is every possibility to have the electrode being of different length and cross sectional area, thereby producing different energized electrons and hence emf.
Inference:
- Generation of emf is the result of repulsion of electrons.
- Chemical reaction is not directly responsible for generation of emf but it only provides electrons more rapidly, thus increasing the rate of flow of available electrons and hence current.
- It once again shows that whenever any substance is introduced in water it produces a specific strain in water and water molecules rearranges themselves accordingly to minimize the strain. Thus each homeopathic medicine prepared from different substances is different and every potency of a specific medicine is different.
References:
[1] Generation of electrode Potential of an electrode –an explanation , Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXIII.2010, www.hanp.net)
[2] Physical Chemistry, P C Rakshit ( Revised 4th Edition) 1980, Published by S C Rakshit, Kolkata , India
[3] Physical Chemistry, S Glasstone, 2nd Edition, 1946
[4]. The Homeopathic Dilution-A New Explanation, Ruhul Amin ,Biplab Chakraborty & Farook Rahaman(Simillimum,vol.XXI,Summer Fall/2008. www.hanp.net)
[5]. The electrical energy of substances –the secret of homeopathy, Ruhul Amin & Biplab Chakraborty(Simillimum,vol.XXII, Summer Fall/ 2009. www.hanp.net)
[6].Water the Magic Liquid: Retaining Electrical Energy, Ruhul Amin & Biplab Chakraborty (Published in homeopathy4everyone April 2012)