This is the 4th in a series of interviews covering The Covid-19 Data Collection Projects and research. It was first published by The Magic Pills movie blog in March 2021.
I recently had the honour of talking to Dr Robbert van Haselen, MSc. He brings his scientific background to homeopathic research, pharmacy and practice. Dr. Haselen has a very distinguished career to date.
He is an independent consultant, working with different stakeholders; one of his current roles as part of Liga Medicorum Homoeopathica Internationalis (LMHI) is to consult on various projects concerning the Covid data collection projects, where he seeks to encourage the production of high-quality case reports and to improve clinical case taking. Further to our discussions, the key points will be covered in this article, with an emphasis on how practitioners can implement and apply these ideas.
Background
Dr. Robbert van Haselen has dedicated his professional career to exploring Complementary and Alternative Medicine (CAM) practices in the context of rigorous science. He has worked as Head of Clinical Research at VSM Geneesmiddelen, a Dutch homeopathic pharmaceutical company, as Deputy Director of Research at the Royal London Homoeopathic Hospital and as Head of Research for Heel, a leading German homeopathic pharmaceutical company.
He took over as Editor-in-Chief of the journal ‘Complementary Therapies in Medicine’ in 2005, and by handover in 2015 turned it into one of the world’s leading peer reviewed scientific journals dedicated to Traditional and Integrated Medicine.
In 2012 Dr van Haselen was appointed as chair of the Clinical Documentation Committee of the Homoeopathic Pharmacopoeia Convention of the United States (HPCUS). He is currently involved in a broad range of activities under the header of the “International Institute for Integrated Medicine” (INTMEDI), which he founded in 2004.
This includes founding the World Integrated Medicine Forum (www.WIMForum.org). Apart from this, he provides consultancy services that offer high quality solutions for the needs of both the private and public Traditional and Integrated Medicine sectors.
Current Focus/Objectives
With the unfortunate advent of Covid-19, homeopaths and doctors specialising in homeopathy have united to create platforms and software to record case data, with the ultimate objective of exploring how, and to what extent, we can help in the current crisis.
We are also building a community through networking, whilst tapping into some of the best minds available to help at this tragic time in history. Dr van Haselen’s involvement comes from his desire to improve the quality of case reports; to make excellence and fine detail a natural element of case taking.
One of his objectives is for homeopaths to critically analyse the content of the data collected in their case taking. He stresses the need to ‘lose the guru culture’ and enter into a deeper and more transparent clinical discussion, rooted in well-recorded cases that feature granular details so others can understand the underlying reasoning.
In all of this he stresses the need to have the intellectual humility to admit to what we do not know, and to embrace the accompanying cognitive unease. He emphasizes that growth as a homeopath can come from analyzing this discomfort, and learning from mistakes.
To this end, uniquely designed inventories and checklists (Hom-Case-Care extension and The MONARCH Causality attribution inventory, described below) are now available. These tools have been designed to cope with the added depth and detail required for rich and meaningful discussions on clinical cases.
Writing up a clinical case report in line with the latest reporting guidelines (mentioned above) is easy and hard at the same time: easy because all the aspects that need to be addressed make common-sense and are relatively easy to understand; hard because despite its apparent simplicity, it takes a lot of time and effort to properly write up a high-quality clinical case report.’ (LHMI: June, 2020.).
What came through strongly for me in talking to Dr Van Haselen, is that setting up the systems and teachings fosters a “democratizing’ of homeopathy. By making these tools available to everyone, a level playing field can be attained in which all homeopaths, irrespective of their years of experience, can participate and contribute to the discussion. Critical thinking should be further integrated in both education and practice, enhancing the practice-based nature and origins of homeopathy.
Webinar series on how to write up a high quality COVID-19 case report
In May 2020, a series of 4 webinars was launched in collaboration with the Vithoulkas Compass team to help educate more practitioners. A core theme discussed in the webinars is defining a high-quality case report. These webinars attracted a high level of interest (with 522 practitioners attending at least one webinar), and the recordings that were posted on YouTube continue to attract many viewers. I encourage you to watch the first webinar here https://youtu.be/8WeKNmHRTGY The webinars received a lot of positive feedback indicating that the training was well appreciated and beneficial to the community.
High Quality case reports
The June LMHI newsletter discusses how ‘High quality clinical case reports are the qualitative backbone of homeopathy and are an important qualitative piece in the mosaic of the various ongoing COVID-19 projects.’
It is clear however, that this is not always easy to achieve. In talking to Dr Van Haselen, he states that we often need to be prepared to move out of our comfort zones, and become more methodical, sometimes slower and more careful in our approach. Embracing the discomfort associated with the increased effort involved in accumulating high quality and comprehensive case information, is essential for learning from our cases.
By using simple tools, such as those discussed, it is possible to report the outcomes in a transparent and consistent way. Every homeopath should use critical thinking he states. In fact, Hahnemann was a critical thinker as is reflected in the following statement: “indolence, love of ease and obstinacy preclude effective service at the altar of truth”
High quality case reports are only part of the picture however, with other equally important elements required: He refers to the ‘quality value chain’ in homeopathy and a chain being only as strong as its weakest link. The most important ‘elements’ of this chain are:
- High quality homeopathic medicinal products
- High quality clinical case reports
- High quality and reliable information in the materia medica and repertories
- Optimal case taking and decision-making
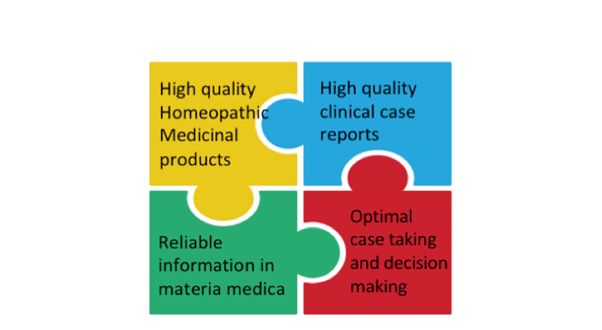
(LHMI, December 2020)
Three important tools for writing a clinical case report
The second LHMI newsletter, (Christmas 2020) highlights specific tools that have been developed to help practitioners write and identify high quality clinical case reports and allow for more transparency.
- The CARE Clinical Case Reporting Guideline (for case reports in general) statement.org/)
- The HOM-CASE CARE Extension2 (additional items to consider for homeopathic case reports) was developed, and as part of the COVID project, was converted into an ‘easy to use’ template in Word, which guides homeopaths in terms of the data that needs to be reported. https://www.dropbox.com/s/v9nv9nic0mpufbo/LMHI2020_COVID- 19_HOM-CASE_submission_form.docx?dl=0
- The MONARCH Causality attribution inventory3 (items to assess the likelihood of causal attribution of observed changes to the homeopathic remedy prescribed). A checklist in (in Word) that can be used for assessing the homeopathic treatment of COVID-19 cases is available here: https://www.dropbox.com/s/499eevc2pmt6o94/LMHI2020_COVID- 19_MONARCH_checklist.docx?dl=0
The CARE guideline and HOM-CASE CARE extension primarily address the comprehensiveness of a clinical case report.
The MONARCH Causality attribution inventory assesses the likelihood that the purported ‘remedy response’ is actually due to the remedy. In some ways, this aspect is even more important than the CARE guideline and HOM-CASE CARE extension:
The wrong attribution of a case as e.g., a ‘phosphorus’ case, inevitably invalidates the case report. The MONARCH aims to create more transparency and consistency in this process.
An important lesson learnt to date was that aspects of the ‘direction of cure’, such as the improvement of symptoms in reverse chronological order, were observed in successful COVID-19 Cases.
However, learning this lesson was only possible because the chronology of symptom development prior to, and after, the remedy administration was reported in sufficient detail. So sufficient comprehensiveness of a clinical case report is essential for this type of assessment.
In short, the ideal Clinical Case Report should be ‘comprehensive, transparent and explicitly address the likelihood of observed changes being due to the homeopathic medicine(s) prescribed. The tools above have fortunately paved the way for this to happen, together with the education and training discussed.
Dr Van Haselen, refers to heuristics, which he describes as “a mental shortcut that allows people to solve problems and make judgments quickly and efficiently.” So in medicine this is akin to the concept of ‘clinical intuition’ rooted in clinical experience.
In homeopathy it is difficult to have a straightforward, algorithmic route to follow. As a highly individualised therapeutic system, decision making is complex. So homeopaths need to be aware that their ‘clinical intuition’ may be wrong.
Hahnemann’s concept of the unprejudiced observer (aphorism 6 Organon), is virtually impossible to attain. There is inevitably bias in both case-taking and symptom/remedy selection, it is part of human nature.
We therefore need to help ourselves by being as transparent and clear as possible, which is very much in line with what Hahnemann applied and suggested. As referred to above, the suggested tools can help to achieve the ‘granularity’ required for in depth discussion on clinical cases.
How do we increase the number of cases being submitted?
Until now, the uptake on case submission continues to be disappointingly low, and whilst its use is increasing, the Hom-Case Care guideline template is not being used as frequently as it should be.
There are a variety of possible contributing factors, such as lack of time, unfamiliarity with having to comply with guidelines that are different from the usual methods of recording cases. Also, the various COVID-19 related LMHI projects were insufficiently connected and integrated.
Based on the above-mentioned analysis, it was decided to better connect all the elements by enhancing collaboration between the different stakeholders. In particular, it was decided to intensify the association with Clificol and other involved parties. This is visualized in the following diagram, which was presented at The ECH General Assembly during the session on COVID-19 on the 14th of November, 2020.
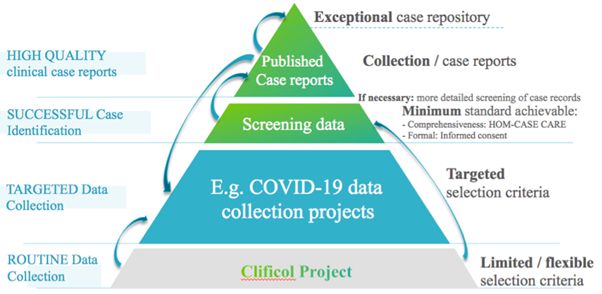
It is hoped that enhancing the connections between a case registry project such as Clificol and high-quality clinical case reports and case-series will have many benefits. The projects can complement each other more effectively, and contribute to improving the effectiveness of homeopathy.
In order to maximise these synergies, there should be close communication between the stakeholders, continuous assessment of feedback, and an efficient means of sorting out any problems, should they arise.
The RadarOpus and Clificol teams are proactive in fostering these collaborations. Information on the LMHI COVID-19 Clinical Case Report project can be accessed via the following websites:
- Clificol COVID-19: https://www.clificol.net/en/ by clicking on ‘Research Collaboration’ under the ‘The Project’ button on the home page
- RadarOpus: https://www.radaropus.com/ under the ‘Research’ button on the home page, and then you arrive at the Clificol homepage.
Conclusion
Throughout the article, the reasons why high-quality case reports are required and the tools to do so have been systematically outlined.
Dr Van Haselen, shared many of the lessons that have been learned along the way. The necessity for a richness of observation at every level, taking us back to our origins as expressed in § 6 in the 6th edition of Hahnemann’s Organon:
‘The unprejudiced observer – well aware of the futility of transcendental speculations which can receive no confirmation from experience – be his powers of penetration ever so great, takes note of nothing in every individual disease, except the changes in the health of the body and of the mind (morbid phenomena, accidents, symptoms) which can be perceived externally by means of the senses; that is to say, he notices only the deviations from the former healthy state of the now diseased individual, which are felt by the patient himself, remarked by those around him and observed by the physician. All these perceptible signs represent the disease in its whole extent, that is, together they form the true and only conceivable portrait of the disease.’
Dr Van Haselen goes on to say that this project has taught us that to achieve excellence in clinical case reports takes time. Going through the process of systematically reporting a case report can be frustrating, in particular because it also reveals the ‘gaps’ in the available information.
We must learn to embrace the discomfort associated with ‘knowing what we don’t know’, because it enables us to learn the most. This type of ‘actively open-minded thinking’ can also help reduce biases during case taking and the remedy selection process. This, in turn, will lead to improved outcomes in patients treated with homeopathy.
To me, much of what Dr Van Haselen stressed was about aligning standards, harmonising our resources and realising, that the ‘quality value chain’ as referred to above, is only as strong as the weakest link.
In the creation of resources such as this, we are also building an active and constantly improving community. He also states that every crisis, as tragic as this one is, holds the potential to be a turning point, to accelerate the further development of homeopathy as ‘practice-based medicine’, and ultimately contribute to healing, at the most productive level. I am quite sure the high standards he sets out here, will go a long way towards realising this.
‘Real knowledge is to know the extent of your own ignorance.’
– Confucius.
References
LMHI: Newsletter (June 2020) Collecting high quality COVID-19 clinical case reports
LMHI: Newsletter (December, 2020) Learning from Case Reports/Series
- Gagnier JJ, Kienle G, Altman DG, Moher D, Sox H, Riley D. The CARE guidelines: consensus-based clinical case report guideline development. Journal of Clinical Epidemiology. 2014;67(1):46-51.
- van Haselen RA. Homeopathic clinical case reports: Development of a supplement (HOM-CASE) to the CARE clinical case reporting guideline. Complementary Therapies in Medicine. 2016;25:78-85.
- Lamba CD, Gupta VK, van Haselen R, et al. Evaluation of the Modified Naranjo Criteria for Assessing Causal Attribution of Clinical Outcome to Homeopathic Intervention as Presented in Case Reports. (EFirst).
Useful Links
- The CARE Clinical Case Reporting Guideline (for case reports in general) (http://www.care-statement.org/)
- The HOM-CASE CARE Extension2 (additional items to consider for homeopathic case reports) was developed, and as part of this COVID project, this was converted into an ‘easy to use’ template in Word, which guides homeopaths in terms of the data that needs to be reported. A link to this document is given here: https://www.dropbox.com/s/v9nv9nic0mpufbo/LMHI2020_COVID- 19_HOM-CASE_submission_form.docx?dl=0
- The MONARCH Causality attribution inventory3 (items to assess the likelihood of causal attribution of observed changes to the homeopathic remedy prescribed). A link to a checklist in (in Word) that can be used for assessing the homeopathic treatment of COVID-19 cases is given here: https://www.dropbox.com/s/499eevc2pmt6o94/LMHI2020_COVID- 19_MONARCH_checklist.docx?dl=0
- Vithoulkas Compass: Improving the Quality of Covid 19 Case taking Webinar, Available at: https://kzclip.com/video/8WeKNmHRTGY/vithoulkascompass-improving-the-quality-of-covid-19-homeopathic-clinical-case-reports-webinar-1of4.html






