Abstract
Lady’s finger plants (Hibiscus escu/entus), grown in pots, were inoculated with the second-stage larvae (76:t 6) of root-knot nematodes Meloidogyne incognita, starting 7 days later they were treated with Cina 30c, Santonin 30c or Ethanol 30c by foliar spray for 10 consecutive days. The drugs in 90% ethanol were diluted with distilled water 1:1000 before application on plants. Thirty days after the last treatment the plants were uprooted. Cina 30c and Santonin 30c reduced nematode infestation of plants significantly in terms of root-gall number, root-protein content and nematode population in roots. Santonin 30c reduced root water content. Santonin 30c may have influenced the water channel proteins of root tissues thereby altering the water contents of roots. The reduced water content in roots might have adversely affected the root-knot nematodes and thus reduced nematode infestation. Ethanol 30c also has some effect on treated plants.
Keywords: Meloidogyne incognita; homeopathic drugs; root water; root gall
Introduction
Plant parasitic nematodes are ubiquitous and cause damage to field and fruit crops as well as forest trees. Meloidogyne incognita and related species produce root-knot disease of vegetables, cereals, pulses and many other crops. These are sedentary endoparasites of roots and are one the most damaging crop pathogens. 1,2 Chemical nematicides cause environmental pollution, contaminate ground water, induce resistance in nematode parasites, cause chromosome aberrations in the root tips and affect germination and growth of plants.3,4 Biological control of nematodes using fungi and other natural enemies of nematodes has not yet met with any degree of success.5-9 Development of nematode-resistant crops is not feasible because of multi species population of nematodes in the rhizospheric soil of plants.4 Nematicidal plant products are effective and easily biodegradable3,10 but procuring them from natural resources for large-scale use is a big problem. Homeopathy holds promise in controlling nematode parasites without disturbing the agroecosystem.
We have already observed that Cina 200c and Cina IOOOc reduced root-knot disease of tomato and cowpea plants.11,12 The purpose of the present study is to determine whether Gna 30e and Santonin 30c could reduce root-knot disease of lady’s finger and whether the treatment could influence the water content of root tissues.
Materials and methods
Host plants
Aseptically germinated seeds of lady’s finger plants, Hibiscus esculentlls, were sown, one seed/pot measuring 20cm in diameter and 20cm in depth. The earthen pots containing a mixture of sterile loamy soil and composted manure (2:1 v/v) were divided into five groups, each of 10 pots.
Inoculation
When the seedlings were at the 4-leaf stage they were inoculated with the second-stage larvae of M. incognita at the rate of 75 + 6 larvae/pot. Of the five groups, one group of pots remained uninoculated control.
Drugs
Flowering tops of Artemisia nilagirica (Clarke) Pamp were collected from Shillong, Meghalaya, India, dried in the shade and extracted with 90% ethanol at room temperature for 2 weeks, This mother tincture was diluted with 90% ethanol 1:100 and given 10 powerful downward strokes to prepare the first centesimal potency called Cina 1c. Cina 30c was prepared by successive dilution and succession of Cina 1c in 29 more steps. Pure Santonin, an active principle of Artemisia, was purchased from Sigma Chemical Company, USA. Santonin 30c was prepared the same way as Cina 30c. Ethanol 30c was prepared from 90% ethanol following the same procedure. All the three potencies were diluted with distilled water 1: 1000 just before application to plants.
Treatment
Seven days after inoculation three groups of plants were treated by foliar spray with Ethanol 30c, Cina 30c and Santonin 30c. The treatment continued for 10 days. Foliar spray was started when the plants were 14 days old and at 4-leaf stage and finished at 8-10 leaf stage. Each plant received 5-10ml of test agent depending on its number of leaves. Treatment was started on 21 July and continued until 3 August, 2004. The plants were kept outdoor at ambient atmospheric temperature 30+-3″C and irrigated when necessary.
Harvesting
All the plants were uprooted 30 days after the last treatment and the following parameters recorded: shoot length, shoot weight, root length, root weight, root gall number, nematode population/2.4 gm root and 200 gm soil, root and leaf protein content and water content in roots and leaves. Five samples of roots or leaves were taken at random from each group of plants and the total protein fraction in each sample was estimated by the falin-phenol method.13,14 An aliquot of the same 5 samples of roots was weighed fresh, torn into fine pieces, kept in an incubator for 12 hr at 90″‘C and weighed again. The difference in weight gave the water content in roots. Water content in the leaves was determined by the same procedure. The experiment was repeated twice and the data from the second experiment have been taken.
Results
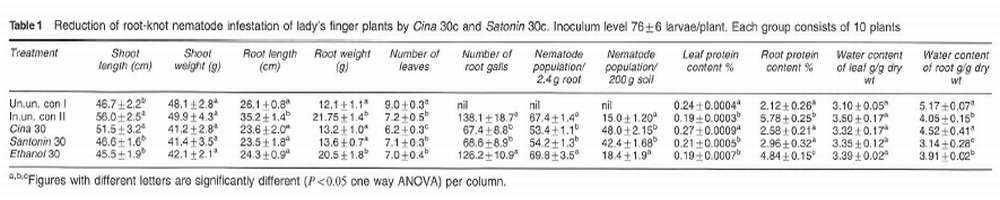
Both Cina 30e and Santonin 30c reduced root-gall number, nematode population in roots, root-protein content and increased leaf protein content significantly (P<0.05, ANOVA, Table I) as compared to the inoculated untreated group. Treatment with these two agents also resulted in a significant increase in nematode population in soil (P<O.05. ANOVA) as compared to the inoculated and untreated control (Table I). Water content in roots decreased significantly in the inoculated untreated group as compared to the uninoculated untreated control (P < 0.05. ANOV A. Table I). Santonin 30c and Ethanol 30c reduced water content in roots significantly (P < 0.05. ANOV A) as compared to the inoculated untreated group. Shoot length, root length and root weight increased significantly in the inoculated untreated group as compared to other groups (P<0.05. Table I). Ethanol 30c did not show any significant difference from the inoculated untreated group with respect to nematode infestation parameters (Table I).
Discussion
At low inoculum level, as in the present experiment, M. incoqnira does not inhibit plant growth and may even stimulate it. 15 This is evident with shoot length and root length (Table I). Cina 30c and Santonin 30c showed similar effects with respect to the reduction of nematode-induced symptoms of host plant. Ethanol 30c, the potentized vehicle control produced significant changes in shoot length, root weight, leaf-protein and root-protein content and water content of roots (Table I). Potencies like Cina 30c and Santonin 30c do not contain any drug molecules derived from their mother tinctures.
The potencies are thought to be specifically structured water, bearing a relationship with the molecules of the mother tinctures from which they have been produced, perhaps due to a hydrogen-bond-mediated structure. 16
The cells of the leaves have cell walls outside the plasma membrane. The cell wall is thick but porous allowing water and small molecules to pass readily through them.
All the cells arc interconnected by plasmodesmata which provide small openings between adjacent cells through which electric currents, ions, small molecules and water can pass. 17 Water molecules covering all the cell surfaces maintain their normal structure which, in a diseased state as with nematode infection, may assume a different structure. During foliar spray the potentized drug or a specifically structured water comes in contact with the water covering the cell membrane and brings about a change in the water structure which may influence the passage of water through the aquaporins and also the function of other integral membrane proteins. 16 This might have resulted in the altered water content in the roots of plants treated with Santonin 30c. Nematode parasites, which induce formation of giant cells in the root tissues and derive their nourishment from those cells, are likely to face an adverse condition due to change in the water content of cells. This prevented further infection of roots by the nematodes, and as a result there was reduction in root-gall number, root-protein content and number of invading nematodes. An increase in the nematode population of the rhizospheric soil of Cina and Santonin-treated plants might be due to the unfavorable condition of the root tissues. Nematode infestation decreased water absorption capacity of plants. 18
Conclusion
Cina 30c and Santonin 30c reduced root-knot disease in experimental infected lady’s finger plants. Santonin 30c and Ethanol 30c reduced water content of roots.
References
1. Semblat JP. Bongiovanni M. Wajnherg E. Dalmasso A, Abad P. Castagnone-Sereno P. Virulence and molecular diversity of parthenogenetic root-knot nematodes. Meloidogyne spp. Heredity 2000; 84: 81-89.
2. Trudgill DL. Blok VC. Apomictic, polyphagous root-knot nematodes: exceptionally successful and damaging biotrophic root pathogens. Annu Rev Phytopathol 2001; 39: 53-77.
3. Sukul NC. Plants antagonistic to plant parasitic nematodes. Indian Rev Life Sci 1992: 12: 23 52
4. Luc M, Bridge J. Sikora RA. Reflections on nematology in sub-tropical and tropical agriculture. in: Luc M. Sikora RA. Bridge J editors. Plant Parasitic Nematodes in Sub-tropical and Tropical Agriculture. Wallingford. UK: CAB International. 1993. p. xi xvii.
5. McClure MA. Biological control of nematodes. in: Baker RR. Dunn PE editors. New Directions in Biological Control: Alternatives for Suppressing Agricultural Pest and Diseases. New York: Wiley-Liss Inc. 1990. p. 169 255.
6. Atkins SD. Hidalgo-Diaz L. Kalisz H. Mauchline TH, Kerry BR. Development of a new nematode strategy for the control of root-knot nematodes (Meloidogyne spp) in organic vegetable production. Pest Manage Sci 2003: 59: 183-189.
7. Tzortzakakis EA. Petsas SE. Investigation of alternatives to methyl bromide for management of Meloidogyne javanica on green house grown tomato. Pest Manage Sci 2003: 59: 1311 1320.
8. Siddiqui IA. Shaukat SS. Trichoderma harzianum enhances the production of nematicidal compounds in Vitro and improves biocontrol of Meloidoqyne javanica by Pseudomonas fluorescence in tomato. Leu Appl Microbiol 2004; 38: 169 175.
9. Tahseen Q, Clark 1M, Atkins SD, Hirsch PR. Kerry BR. Impact of the nematophagous fungus Pochonia chlamydospooria on nematode and microbial populations. Commun Agric Appl Biol Sci 2005; 70: 81-86
10. Sukul NC. Biopesticides in nematode control. in: Dhaliwal GS. Kansal BD editors. Management of Agricultural Pollution in India. New Delhi: Commonwealth publishers. 1994. p. 248 269.
11. Sukul NC, Sukul A. Potentized Cina reduces root-knot disease of cowpeas. Environ Ecol 1999: 17: 269 273.
12. Sukul NC, Sinhababu SP. Datta SC. Nandi B, Sukul A. Nematoloxic effect of Acacia auriculiformis and Artemisia nilagirica against root-knot nematodes. Allelopathy J 2001: 8: 65 72.
13. Lowry OH, Rossebrough NJ, Farr AR, Randall RJ. Protein measurement with Folinphenol reagent. J Biol Chem 1951: 193: 265-275.
14. Chatterjee A, Sukul NC. Total protein of galled roots as an index of root-knot nematode infestation of lady’s finger plants. Phytopathology 1981; 31: 372-374.
15. Das P, Sukul NC. The influence of inoculum density of root-knot nematodes, soil type and irrigation on the growth and root gall formation of tomato plants. Visva-Bharati J Res 1984: 5: 158 192.
16. Sukul NC, Sukul A. High Dilution Effects: Physical and Biochemical Basis. Dordrecht: Kluwer Academic Publishers. 2004.
17. Nelson DL, Cox MM. Lehninger Principles of Biochemistry, 3rd edn. New York: Macmillan Worth Publishers. 2000 p 46.
18. Saxena SK. Nematode infestation in vegetables. in: Swarup G, Dasgupta DR editors. Plant Parasitic Nematodes of India: Problems and Progress. New Delhi: Indian Agricultural Research Institute. 1986. p. 312 327.
———————————-
Correspondence: NC Sukul, Department of Zoology, VisvaBharati University, Santiniketan 731235, West Bengal, India.