Homeopathy stands out as an experimental discipline, as is evidenced by the vast amount of homeopathic data collected over more than 2 centuries. However, homeopathy remains controversial from the perspective of Western medicine because of the apparent inadequacy or inconsistency of data to support use of the ultralow concentrations in which homeopathic medications are given. Consequently, the prevailing attitude does not consider the small amount of ingredients used in homeopathy as being capable of inducing significant changes in classic pharmacologic models. 1
Homeopathic research has evolved over the past 20 years with the advent of comprehensive biomedical research methodologies that make subtle effects more amenable to detection (i.e., clinical trials, observational studies, statistical evaluations, computerized storage programs, and instrumental or laboratory testing). Some innovative models have been used to contribute to the debate about the efficacy of homeopathic medications, including demonstration of the efficacy of ultrahigh dilutions of IgE on basophil degranulation, inhibition of basophil degranulation by ultrahigh dilutions of histamine, and other models that incorporate the measurement of cell viability and cytokine induction, which may be diagnostic laboratory techniques for determining the immunologic potency of various homeopathic preparations and may help clarify the clinical benefit of therapies with these substances.2-5
For the past 10 years, homeopathic research has ventured into the evaluation of combination formulas used to treat a variety of clinical conditions. 6-9 In many respects, this is a considerable challenge, given the controversies surrounding research on single-component homeopathic preparations. However, by focusing on well-established endpoints and using conventional research methodologies, it is possible to elicite meaningful data from well-designed preclinical studies.
The field of allergy and immunology has been viewed as a bridge into homeopathy since both disciplines share the concept that modulations in biologic systems are sensitive to very low concentrations of natural or endogenous substances. 1 Therefore, appropriate models for evaluating multicomponent homeopathic preparations and determining their effects on immune function or mediators of immunity are essential. Traumeel ® S (Heel GmbH, Baden-Baden, Germany ), an extensively evaluated commercial homeopathic preparation with a combination of ingredients, has been shown to modulate the secretion of interleukin (IL)-1β, tumor necrosis factor (TNF)-α and IL-8 in immune cells. 10 Thus, modulation of mediators of immunity may represent an opportunity to assess appropriate homeopathic mixtures using in vitro models. In the present study we attempted to further validate this hypothesis by exploring the effects of 2 multicomponent homeopathic formulas, Flu Terminator ® and Phase 6, on a panel of pro- and anti-inflammatory cytokines in human leukocyte cultures.

MATERIALS AND METHODS
Preparations and reagents
Phase 6 and Flu Terminator are homeopathic preparations that were obtained from the manufacturer. Roswell Park Memorial Institute (RPMI) cell culture medium, fetal bovine serum (FBS), phosphate-buffered saline (PBS), and antibiotics were purchased from Invitrogen, Inc., Corporation, Carlsbad, CA. Enzyme-linked immunosorbent assay (ELISA) reagent kits for human-specific cytokines (IL-1, IL-4, IL-10, and interferon [IFN]-γ and TNF-α were purchased from BD Biosciences, San Diego, CA. The constituent ingredients of Flu Terminator and Phase 6 are listed in Table 1.
Isolation of human leukocytes and incubation with Phase 6 and Flu Terminator
Leukocytes were isolated from human blood samples collected from healthy volunteers using Histopaque 1066 (Sigma Chemical Co., St. Louis, MO) gradient centrifugation.9 Cells were washed with PBS and 106 cells/mL were incubated for 24 hours with varying doses of Phase 6 or Flu Terminator (0-200 µL/mL) in RPMI medium supplemented with 10% FBS and antibiotics at 37°C in a 5% humidified CO 2 incubator. Human leukocytes treated with 0-200 µL of 20% ethanol (the carrier) served as controls in order to determine the carrier effects of Phase 6 and Flu Terminator.
Analysis of cytokine synthesis
After 24 hours of drug incubation, cell-culture medium was removed by centrifugation at 400 g for 10 min. Cytokines (IL-1, IL-4, IL-10, IFN-γ, and TNF-α) synthesized and released into the medium by leukocytes were estimated by ELISA, using reagent kits from BD Biosciences. Briefly, 100 µL of medium was used for a multiwell ELISA protocol according to the manufacturer’s recommendations, and the synthesis of cytokines (in concentrations of pg/mL) by the leukocytes was quantified. 11 The experiment was repeated thrice for estimation of standard deviation values, and the estimates of cytokine concentrations were analyzed statistically through analysis of variance (ANOVA).
RESULTS AND DISCUSSION
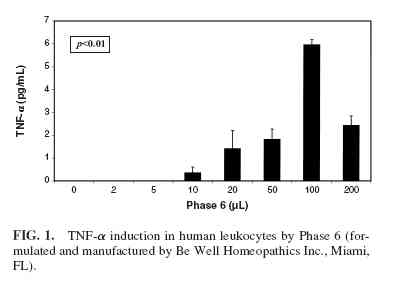
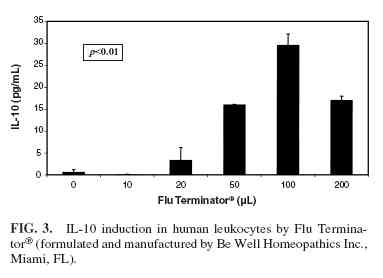
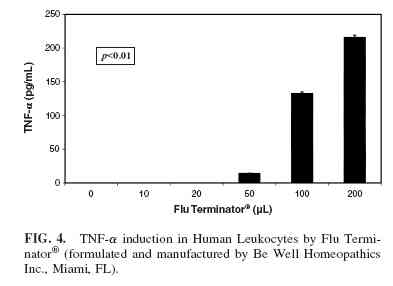
Phase 6 induced the production of TNF-α (Fig. 1) and IFNγ(Fig. 2) at doses of Flu Terminator and Phase 6 ranging from 10 µL to 100 µL, with a decrease in production at 200 µL ( p <0.01). However IL-1, IL-4, and IL-10 were not induced by Phase 6. The cells treated with increasing quantities of 20% ethanol failed to show any cytokine production, indicating that the effect of the two homeopathic preparations was not due to the solvent carrier. Flu Terminator induced production of IL-10 and TNF-α (Figs. 3 and 4), apparently in a dose-dependent manner ( p < 0.01). However, Flu Terminator failed to induce synthesis of IL-4 and IFN-γ. The cells treated with increasing quantities of 20% ethanol failed to show any cytokine production, indicating that the effect was not due to the solvent carrier.
Although a growing body of clinical research supports the existence of biologic effects of homeopathic treatments, the principles governing these interventions remain elusive despite abundant theories. The principles of homeopathy will not achieve mainstream medical acceptance without homeopathic preparations being subjected to stringent evidence- based research using conventional methods. However, it is equally essential to recognize that bias by some scientists who dismiss outright the effects of homeopathic preparations as incredible or owing to placebo because the dilutions are so great is not an evidence-based approach to their efficacy.
Both Phase 6 and Flu Terminator are mixtures of highly diluted extracts and minerals that range from 1/10 of the starting material to high dilutions with concentrations beyond (smaller than) Avogadro’s number (6.23 * 1023), which corresponds roughly to a 24X dilution. Both preparations are recommended for general immune defense and for the treatment of viral illnesses, respectively. They were therefore appropriate preparations for evaluating their effects on mediators of immunity, which in our study consisted of pro- and anti-inflammatory cytokines.
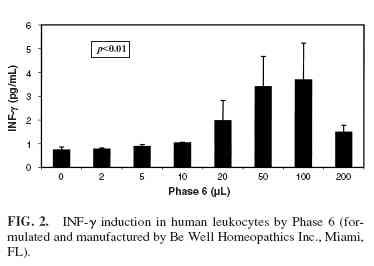
The results obtained in our study were reproducible and statistically significant. While it is not possible to draw a specific conclusion other than to note the presence of a biological effect, several points are noteworthy. In the case of Phase 6, two important proinflammatory cytokines associated with the T-helper 1 (Th2) pathway showed a “U”- shaped response pattern, and not in the dose-response manner typical for a pharmaceutical agent. None of the cytokines associated with the Th2 pathway that were studied were elevated. This is an important observation, suggesting that the effects of the two homeopathic preparations that we studied are not random. Rather they seem to reflect a selective effect on specific biologic pathways in the activation of proinflammatory cytokines.
In the case Flu Terminator, the concentrations of TNF-α, a proinflammatory cytokine, and IL-10, an anti-inflammatory cytokine, were both elevated again, with a “U”-shaped response pattern. While this may appear contradictory, it is important to be aware that the Th2 and Th2 pathways of immune response act in a complementary manner in response to various infections, thus providing a mechanism for immune modulation. The data in our study in no way address the timing of these responses, but do suggest that Flu Terminator may play a role in activation and modulation of the immune response associated with infection.
In either case, skepticism associated with the possibility that ultralow concentrations of homeopathic medications can modulate the expression of these cytokines, irrespective of the theoretical mechanism by which they do this, must be reconsidered. Examination of principles of immunology may provide some support for these observations. Studies of the effects of ultralow concentrations of various sub-stances have clearly demonstrated reproducible biologic effects in vitro and in vivo . 2-10,12,13
The concentrations of cytokines required to induce effects in vivo have been found to be in the femtomolar range (10-15M), clearly within the range of homeopathic preparations. 14-16 Such concentrations are at the lower limits of detection by standard laboratory assays, and whether these preparations can demonstrate biologic effects unequivocally without inferring a concentration (theoretical determination) is therefore unclear.
However, there are examples of concentrations of cytokines that demonstrate biologic effects clearly, which have been indirectly determined.
These include the inhibition of basophil deregulation by ultralow concentrations of histamine, with confirmation of receptor specificity, and other effects on immune function in vitro of other bioactive compounds at ultralow concentrations. 4,5 It is also noteworthy that antigens that have immunostimulatory effects may elicit these effects at femtomolar or even subfemtomolar concentrations.17-19 Clearly, at least in the case of immune function, it does appear that cells in vitro normally respond to signals associated with ultralow concentrations of bioactive molecules, thus lending some support to the principles of homeopathy.
CONCLUSIONS
The impact on cytokine expression of Phase 6 and Flu Terminator in our study lends additional support to the growing body of evidence that dilutions of extracts and minerals do affect biologic systems. Furthermore, such studies also support the possibility that homeopathic agents, as in the case of pharmaceuticals, may affect specific molecular targets, thus defining potential medical applications for these agents. The controversy over the efficacy of low concentrations of homeopathic agents will, however, probably remain until more adequate techniques become available to assess molecular interactions at such concentrations; only then will more reliable models for evaluating effects of homeopathic dilutions be established and expand our knowledge of the physical and biological basis of these interactions.
REFERENCES
1. Bellavite P, Conforti A, Piasere V, Ortolani R. Immunology and homeopathy: 1. Historical background. eCAM 2005;2: 441-452.
2. Brizzi M, Lazzarato L, Nani D, et al. Biostatistical insight into the As
2 O 3 high dilutions effects on the rate and variability of wheat seedling growth. MACROBUTTON HtmlResAnchor Forsch Komplementarmed Klass Naturheilkd. 2005;12:277-283.
3. Elsasser-Beile U, Voss M, Schuhle R, Wetterauer U. Biological. Effects of natural and recombinant mistletoe lectin and an aqueous mistletoe extract on human monocytes and lymphocytes in vitro . J Clin Lab Anal 2000;14:255-259.
4. Bellavite P, Conforti A, Pontarollo F, Ortolani R. Immunology and homeopathy: 2. Cells of the immune system and inflammation. Evidence-Based Complement Alternat Med 2006; 3:13-24.
5. Belon P, Cumps J, Ennis M, et al. Histamine dilutions modulate basophil activation. Inflamm Res 2004;53:181-188.
6. Wiesenauer M, Gaus W, Bohnacker U, Haussler S. Efficiency of homeopathic preparation combinations in sinusitis: Results of a randomized double blind study with general practitioners. Arzneimittelforschung 1989:39:620-625.
7. Maiwald VL, Weinfurtner T, Mau J, Connert WD. Therapy of common cold with a homeopathic combination preparation in comparison with acetylsalicylic acid: A controlled, randomized double-blind study. Arzneimittelforschung 1988;38: 578-582.
8. Jacobs J, Guthrie BL, Montes GA, et al. Homeopathic combination remedy in the treatment of acute childhood diarrhea in borders. J Altern Complement Med 2006;12:723-732.
9. Samsonavae EI, Smirnova TN, Poliakov VE. Effective complex homeopathic outpatient treatment of a child with allergic rhinitis and neutropenia. Vestn Otorinolaringol 2006;2:57-59.
10. Porozov S, Cahalon L, Weiser M, et al. Inhibition of IL-1β and TNF-α secretion from resting and activated human lymphocytes by the homeopathic medication Traumeel ® S. Dev Immunol 2004;11:143-149.
11. Nair PK, Rodriguez S, Ramachandran R, et al. Immune stimulating properties of a novel polysaccharide from the medicinal plant Tinospora cordifolia . Int Immunopharmacol 2004;4:1645-1659.
12. Bellavite P, Ortolani R, Conforti A. Immunology and homeopathy: 3. Experimental studies on animal models. Evidence- Based Complement Alternat Med 2006;3:171-186.
13. Walchli C, Baumgartner S, Bastide M. Effects of low doses and high homeopathic potencies in normal and cancerous human lymphocytes:
An in vitro study. J Altern Complement Med 2006;12:421-427.
14. Collins DP, Luebering BJ, Shaut DM. T-lymphocyte functionality assessed by analysis of cytokine receptor expression, intracellular cytokine expression, and femtomolar detection of cytokine secretion by quantitative flow cytometry. Cytometry 1998;33:249-255.
15. Bank U, Reinhold D, Kunz D, et al. Effects of interleukin-6 (IL-6) and transforming growth factor- β (TGF-β ) on neutrophil elastase release. Inflammation 1995;19:83-99.
16. Curfs JHAJ, Meis JFGM, Hoogkamp-Korstanje JAA. A primer on cytokines: Sources, receptors, effects and inducers. Clin Microbiol Rev 1997;10:742-780.
17. Klinman NR. The secondary immune response to a haptogen in vitro . J Exp Med 1971;133:963-973.
18. Dejda A, Sokolowska P. Nowak JZ. Neuroprotective potential of three neuropeptides PACAP, VIP and PHI. Pharmacol Rep 2005;57:307-320.
19. Lichtenberger III FJ, Montague CR, Hunter M, et al. NAC and DTT promote TGF-β 1 monomer formation: Demonstration of competitive binding. Journal of Inflammation 2006;3:7-15.