Highlights
Solvatochromic dyes display changes in their spectra in the presence of homeopathic potencies. Interaction between solvatochromic dyes and potencies appears to occur through the dyes’ intramolecular electron transfer feature.
Homeopathic potencies affect the supramolecular behaviour of solvatochromic dyes. A systematic approach to the design of simple, chemical systems for investigating the nature of homeopathic medicines has led to the development of an experimental protocol in which solvatochromic dyes are used as molecular probes of serially diluted and agitated solutions. Electronic spectroscopy has been used to follow changes in the absorbance of this class of dyes across the visible spectrum in the presence of homeopathic potencies. Evidence is presented using six different solvatochromic dyes in three different solvent systems. In all cases homeopathic potencies produce consistent and reproducible changes in the spectra of the dyes. Results suggest that potencies influence the supramolecular chemistry of solvatochromic dyes, enhancing either dye aggregation or disaggregation, depending upon dye structure. Comparable dyes lacking the intramolecular charge transfer feature of solvatochromic dyes are unaffected by homeopathic potencies, suggesting potencies require the oscillating dipole of solvatochromic dyes for effective interaction. The implications of the results presented, both for an eventual understanding of the nature of homeopathic medicines and their mode of action, together with future directions for research in this area, are discussed.
Introduction
There is no doubt that a plausible and testable hypothesis for the mode of action of homeopathy and, by implication, an understanding of the physico-chemical nature of homeopathic potencies, would profoundly enhance homeopathy, both as an area of legitimate scientific study and as an effective medical approach. Research at the molecular level has the advantage over other approaches in that it can ask the kinds of searching and detailed questions necessary to arrive at fully testable hypotheses as to the modus operandi of homeopathy.
With this view in mind a programme of investigation aimed at developing well-defined chemical systems capable of detecting consistent and replicable effects of serially diluted and agitated solutions has been initiated. Specifically, a simple chemical system utilising environment sensitive solvatochromic dyes1 has been developed. Solvatochromic dyes are sensitive to, and can be used to follow, a range of solution dynamics through changes in their absorbance spectra which, conveniently, occur in the visible portion of the electromagnetic spectrum.
The system described below demonstrates not only that homeopathic potencies have in vitro effects which can be measured, but also because the system is both simple and versatile, very specific questions can be asked about what molecular effects potencies are having in solution and what their ultimate nature might be.
Whilst a range of chemical and physical systems have been employed in the past in the study of homeopathic medicines, including UV-spectroscopy,2 nuclear magnetic resonance spectroscopy,3 thermoluminescence,4high voltage plasma visualisation,5 solution conductivity6 and micro-calorimetry,7 together with theoretical studies,8 little consensus has emerged as to the nature of the homeopathic stimulus. Results suggest potencies may be electromagnetic in nature,9 or that they may involve and exploit the intrinsic ability of water to form complex hydrogen bonding networks.10 They may have their origins in quantum electrodynamics,11quantum entanglement,12 complexity theory13 or stochastic resonance.14
The present approach has grown out of a recognition that a number of criteria need to be fulfilled if homeopathic potencies are to be studied in a way that provides results that are (i) significantly above background noise and (ii) allows the development of systems that can be manipulated to reveal the effect of one variable at a time. The criteria are essentially two fold. The first involves the stability of homeopathic potencies. Whilst firm evidence is lacking, it is a commonly held belief that potencies are sensitive to ultraviolet light, X-rays and strong magnetic and electrical fields.15 For this reason it was felt that any putative detection system should avoid UV-spectroscopy, thermoluminescence, NMR and high voltage plasma visualisation. Conversely, little is known about how potencies can be effectively destroyed. Heat is commonly held to be effective, but again evidence is lacking.16 The destruction of potencies is important if one is to avoid cross-contamination where glassware and other container materials are re-used. For this reason the current study has employed disposable containers in all situations where cross-contamination is a potential problem.
The second group of criteria revolves around the issue of the control of variables. Ideally any detection system should be one in which the variables involved can be addressed individually. In this way specific questions can be asked and specific answers obtained. A well-defined and simple detection system is therefore highly desirable, especially if the system is capable of providing different types of information.
With these criteria in mind a detection system involving solvatochromic dyes has been developed. In brief, changes have been found to occur in the absorbance spectra of these dyes in the presence of homeopathic potencies. In turn it has then been possible to make certain inferences as to the specific action of potencies in solution.
A system in which homeopathic potencies are added to solutions of solvatochromic dyes is simple, versatile, and involves a very small number of components and operational steps. In addition, the system is sensitive to changes in a wide range of solution dynamics including solvent polarity, solvent-solute binding patterns, and supramolecular interactions between solute molecules.
To date six different solvatochromic dyes have been investigated, three positively solvatochromic and three negatively solvatochromic (Figure 1).
Figure 1
Structures of ET33 (2,6-Dichloro-4-(2,4,6-triphenyl-pyridinium-1-yl)-phenolate); ET30 (2,6-Diphenyl-4-(2,4,6-triphenyl-pyrinidium-1-yl)-phenolate); BM (4-[(E)-2-(1-methylpyridinium-4-yl)ethenyl]phenolate/Brooker’s merocyanine); BDN ((4-(Bis-(4-(dimethylamino)phenyl)methylene)-1(4H)-naphthalenone); NR (9-diethylamino-5H-benzo[α]phenoxazime-5-one/Nile Red); PB (N,N-dimethylindoaniline/Phenol Blue); ET33, ET30 and BM are negatively solvatochromic dyes; BDN, NR and PB are positively solvatochromic dyes. See text for an explanation of negative and positive solvatochromism.
An overview of solvatochromic dyes
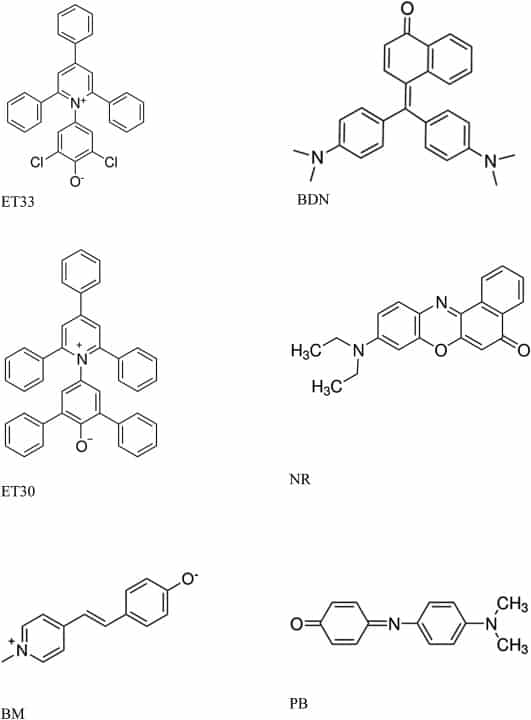
Solvatochromic dyes are characterised by possessing an electron donating group and an electron accepting group with an electron delocalised system in- between.17 For negatively solvatochromic dyes the ground or resting state is zwitterionic with a formal charge at either end of the molecule. On absorption of light an electron travels from one end of the molecule to the other to form an excited polar, but uncharged, state (Figure 2). Whilst the lifetime of the excited state is of the order of picoseconds this rapid electron oscillation occurs constantly under the influence of absorbed light. Importantly the wavelength of the absorbed light is influenced by the environment in which the dye is placed. Conversely, for positively solvatochromic dyes the ground or resting state is uncharged. On absorption of light an electron travels from one end of the molecule to the other to form an equally short-lived charged excited state (Figure 2). Again the wavelength of the light absorbed is dependent upon the nature of the environment in which the dye is placed. Negatively and positively solvatochromic dyes behave differently and complementarily to each other in a number of ways. These are important in relation to the results obtained with potencies reported below.
Figure 2
Diagram showing the structure of ground and excited states in negatively and positively solvatochromic dyes. For the positively solvatochromic dyes used in this study electron donating groups (D) are of the form =N– and electron accepting groups (A) are of the form =C=O. For the negatively solvatochromic dyes used in this study electron donating groups are of the form –O− and acceptor groups are of the form N+–. (see text for more details).
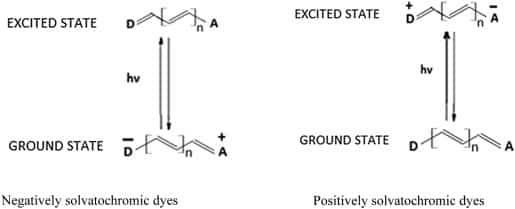
Negatively solvatochromic dyes absorb at longer and longer wavelengths (bathochromically shifted) as solvent polarity decreases. For example ET30 (Figure 1) absorbs at 450 nm in water, 550 nm in ethanol and 650 nm in tert-butyl alcohol.18 In addition these dyes tend to increasingly aggregate as solvent polarity decreases producing aggregates that are bathochromically shifted with respect to monomer (unaggregated material), a phenomenon known as aggregachromism.19 These bathochromically shifted species take the form of ‘steps’ in solution and are known as J-aggregates20 (Figure 3).
Figure 3
Schematic representation of H-aggregates (A) and J-aggregates (B) in solution.
Conversely, positively solvatochromic dyes absorb at shorter and shorter wavelengths (hypsochromically shifted) as solvent polarity decreases. For example BDN (Figure 1) absorbs at 614 nm in water, 564 nm in ethanol and 537 nm in tert-butyl alcohol.21 These dyes tend to increasingly aggregate as solvent polarity decreases producing aggregates that are hypsochromically shifted with respect to monomer,22 in contrast to that seen with negatively solvatochromic dyes. These hypsochromically shifted species take the form of ‘stacks’ in solution and are known as H-aggregates23 (Figure 3).
Dyes ET33, ET30 and BM produce J-aggregates in solution; dyes BDN, NR and PB produce H-aggregates.24
Results reported below exploit these complementary properties of positively and negatively solvatochromic dyes.
In addition, dyes ET30, ET33 and BDN bind divalent cations25 to produce optical changes which can also be utilised to demonstrate effects of homeopathic potencies.
Precautions taken in this study
The experimental protocols followed in the course of this study have focused on a number of different practical issues which need to be discussed before results are presented (for details see Materials and methodssection). The first involves the use of disposable cuvettes. Whilst quartz cuvettes have been found to produce greater differences between controls and potency assays, so too have the size of variations between assays, producing large error bars. In part this has been found to arise from the necessary re-use of quartz cuvettes. Whilst cuvettes were placed in boiling water between assays it is possible that such a procedure is insufficient to destroy all the potency present. Cross contamination is therefore a real possibility between assays. For this reason the use of single-use polystyrene (PS) and Brand UV (UV) cuvettes has been adopted. Although differences between controls and potency assays are smaller than those obtained with quartz cuvettes this has been offset by using much higher potencies (50 M) than those used originally with quartz cuvettes (200).
Choice of homeopathic potency
Another consideration has been that of the homeopathic medicines chosen for assay. It was felt that remedies derived from complex plant, mineral and animal materials should be avoided on the basis of the desire for simplicity throughout. Many remedies at different potencies have been investigated during the course of the present study and the results obtained are broadly comparable to the results presented in this paper. However, for the purposes of keeping system components to a minimum, and with an ever present desire for simplicity, potencies of glycerol have been chosen as the primary source of homeopathic remedy for assay. Glycerol is a low molecular weight compound that can be obtained at very high purity levels, is pharmacologically inactive in material doses and is fully miscible with both water and ethanol. In addition it is very closely related structurally to both water and ethanol. It was therefore considered that glycerol may be more likely than most substances to produce results that represent the potentisation process itself, free of any specific homeopathic effects that might have derived from the starting material, especially where that substance is chemically reactive or pharmacologically active.
Considerations regarding the solvent in which assays are performed
The final practical consideration in relation to experimental protocol has centred on the choice of solvent in which to carry out assays. A question from the beginning of this study has been whether bulk water is essential in order to observe any effects of homeopathic medicines. The idea that water in some way can carry the memory of substances placed in it is an enduring one,26 but has ignored the fact that pharmacies produce, and practitioners use, potencies that are in 90% ethanol. For this reason three different solvents have been chosen in which to carry out assays. These are water, ethanol and tert-butyl alcohol (see Materials and methods). These three solvents form a series in which there is decreasing hydrogen bonding capacity, increasing hydrophobicity and decreasing solvent polarity in going from water to tert-butyl alcohol. As such it was felt this series could provide valuable information with respect to how potency effects may vary in relation to these particular solvent parameters.
Results
Results with dye ET33
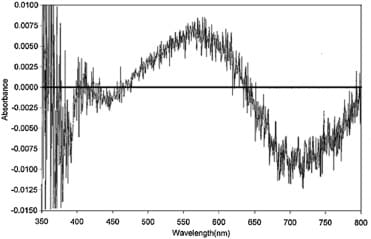
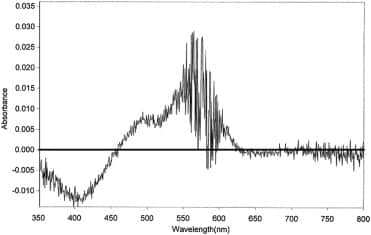
Figure 4 shows a difference spectrum obtained on adding an aliquot of glycerol 50 M (sample cuvette) and an equivalent volume of control (reference cuvette), to solutions of the negatively solvatochromic dye ET33 dissolved in ethanol. Scans were then repeatedly made from 400 nm to 800 nm over several hours (seeMaterials and methods). The scan shown was taken at 120 min after mixing. There is a relative decrease at c.542 nm and a relative increase at c.442 nm which builds up over time.
Figure 4
Difference spectrum of ET33 in ethanol with 50 μl of control added to the reference cuvette and 50 μl of glycerol 50 M added to the sample cuvette to make a total volume of 3 ml in each cuvette (see Materials and methods). ET33 is at a concentration of 245 μM. Difference spectrum consists of the sample spectrum minus the reference spectrum. Difference spectrum shown is that at t = 120 min after mixing. UV cuvettes used. Insert shows the change in ΔOD442 nm over time.
The difference is minimal at t = 0 but slowly reaches a maximum at 60–120 min (see Figure 4 insert). This difference spectrum should be considered in relation to the spectrum of ET33 in ethanol which has a broad peak at 472 nm. ET33 aggregates in solution to produce stepped aggregates (J-aggregates) that absorb at longer wavelengths than monomer (bathochromically shifted).20 Solutions of ET33 are therefore an equilibrium mixture of monomer and aggregates – hence the broad nature of the absorption spectrum of ET33. What Figure 4 appears to be showing therefore is that potency is causing ET33 to disaggregate to produce more monomer.
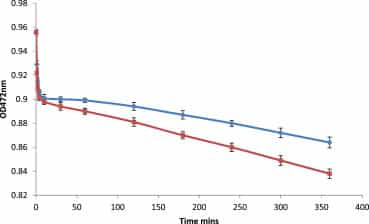
Support for this proposition comes from assays utilising divalent cations. Strontium ions interact with the phenoxide moiety of ET3325 (Figure 1) resulting in a loss of absorbance. This loss occurs in two stages: a rapid first phase due to the direct interaction of strontium ions with free dye, followed by a second phase arising from rate-limiting disaggregation of dye. If potency causes a greater level of disaggregation of dye then one might expect more rapid loss of absorbance in the presence of potency. This is indeed what is seen.Figure 5 shows assays in which either control plus strontium ions or glycerol 50 M plus strontium ions are added to solutions of ET33 in ethanol. Loss of absorbance is more rapid in the presence of potency, supporting the idea that potency enhances disaggregation of dye.
Figure 5
Absorbance loss over time at 472 nm for 245 μM ET33 in ethanol with 70 μM SrCl2 and 50 μl control (upper curve) or 70 μM SrCl2 and 50 μl glycerol 50 M (lower curve). Assays carried out in both PS and UV cuvettes. Details of assay conditions are given in Materials and methods. N = 20; error bars are to first standard deviation; p < 0.0001, indicating high statistical significance.
Difference spectra of ET33 in tert-butyl alcohol with and without potency show a decrease at c.615 nm and an increase at c.490 nm (the absorbance peak of ET33 in tert-butyl alcohol is at 548 nm). As with assays in ethanol, glycerol 50 M appears to be causing enhanced disaggregation of dye. What these results in ethanol and tert-butyl alcohol show is that bulk water is not essential to manifest a potency effect. More specifically, astert-butyl alcohol very poorly hydrogen bonds, it is likely that hydrogen bonding is not essential in order to manifest a potency effect.
Results with dye ET30
Difference spectra of ET30 (Figure 1) in water, ethanol and tert-butyl alcohol indicate enhanced disaggregation in the presence of glycerol 50 M in all three cases. The difference spectrum in tert-butyl alcohol is one of the largest seen in any of the dye/solvent combinations examined (Figure 6). As with ET33, difference spectra tend to develop over time, although on some occasions evidence of a difference spectrum is present even from t = 0. Scans in most of the dye/solvent combinations tested tend to show a return to base line with loss of any difference spectrum overnight, indicating that the potency effect is one that slowly builds up over time, reaching a maximum somewhere between 1 and 2 h and then slowly disappears overnight.
Figure 6
Difference spectrum of ET30 in tert-butyl alcohol with 50 μl of control added to the reference cuvette and 50 μl of glycerol 50 M added to the sample cuvette to make a total volume of 3 ml in each cuvette (seeMaterials and methods). ET30 is at a concentration of 245 μM. Difference spectrum consists of the sample spectrum minus the reference spectrum. Difference spectrum shown is that at t = 120 min after mixing. UV cuvettes used.
Consistent with the results seen with ET33, glycerol 50 M also increases the rate of complexation of strontium ions with ET30, through enhanced disaggregation.
Results with dye BDN
Complementary evidence to that with ET33 and ET30 is seen with the positively solvatochromic dye BDN.Figure 7 shows a difference spectrum of BDN in ethanol with and without glycerol 50 M. Conditions are exactly the same as in Figure 4 except BDN has been substituted for ET33. Scans were performed at the same time intervals as for ET33. The scan shown was performed at t = 30 min after mixing. There is a relative decrease at c.615 nm and a relative increase at c.480 nm. This difference spectrum should be seen in relation to the absorption spectrum of BDN in ethanol which shows two poorly differentiated peaks at 484 nm and 564 nm corresponding to aggregate and monomer peaks respectively. BDN in solution produces aggregates (H-aggregates) that absorb at shorter wavelengths than monomer. As with ET33, solutions of BDN are equilibrium mixtures of monomer and aggregates. However, in distinction to ET33 the aggregates in BDN are hypsochromically shifted. The difference spectrum in Figure 7 therefore appears to show that glycerol 50 M is promoting dye aggregation in BDN.
Figure 7
Difference spectrum of BDN in ethanol with 50 μl of control added to the reference cuvette and 50 μl of glycerol 50 M added to the sample cuvette to make a total volume of 3 ml in each cuvette (see Materials and methods). BDN is at a concentration of 80 μM. Difference spectrum consists of the sample spectrum minus the reference spectrum. Difference spectrum shown is that at t = 30 min after mixing. UV cuvettes used.
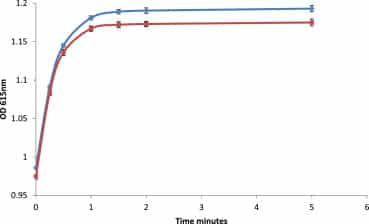
Divalent cations interact with BDN to produce a rise in absorbance at 615 nm and a decrease in absorbance at 484 nm. As with ET33 and ET30, this complexation has a second slower phase arising from rate-limiting disaggregation of dye. If potency is enhancing dye aggregation then one would expect potency to reduce the rate of increase in absorbance at 615 nm compared with that in the absence of potency. This is indeed what is seen. Figure 8 shows assays in which either control plus strontium ions or glycerol 50 M plus strontium ions are added to solutions of BDN in ethanol. Gain of absorbance is slower in the presence of potency, supporting the idea that potency protects BDN from complexation with strontium ions through enhanced dye aggregation.
Figure 8
Absorbance gain over time at 615 nm for 80 μM BDN in ethanol with 70 μM SrCl2 and 50 μl control (upper curve) or 70 μM SrCl2 and 50 μl glycerol 50 M (lower curve). Assays carried out in both PS and UV cuvettes. Details of assay conditions are given in Materials and methods. N = 20; error bars are to first standard deviation; p < 0.0001, indicating high statistical significance.
Difference spectra of BDN in water, ethanol and tert-butyl alcohol with glycerol 50 M all show increases at shorter wavelengths and decreases at longer wavelengths corresponding to enhanced dye aggregation, irrespective of solvent. Solvent does not therefore appear to be playing a direct part in the action of glycerol 50 M. Furthermore, if potency were acting on either ET33 or BDN through changes in solvent polarity or hydrogen bonding capabilities the dyes should not be affected in opposite ways. Both dyes tend to aggregate more strongly as solvent polarity decreases and disaggregate as solvent hydrogen bonding capability increases. What is a more likely explanation is that glycerol 50 M is acting directly on both ET33 and BDN. If this is the case then the charge transfer feature of both dyes may be the significant factor in determining interaction.
Results with non-solvatochromic dyes
In support of the idea that the charge transfer feature of dyes ET33, ET30 and BDN may be the significant factor in determining interaction with potencies, a number of non-solvatochromic dyes were investigated. Dyes were chosen which are zwitterionic, as structurally analogous as possible to BDN and ET33, and display aggregation in solution with monomer and aggregate spectra separated by at least 40 nm. Dyes included Patent Blue VF, Green S and Sulforhodamine 101. No evidence of any changes in their spectra on addition of glycerol 50 M were seen under conditions identical to those used for ET33 and BDN. This suggests that it is indeed the intramolecular electron transfer feature of solvatochromic dyes that is interacting with glycerol 50 M. This interaction then appears to result in changes in the supramolecular dynamics of solvatochromic dyes.
Results with other solvatochromic dyes
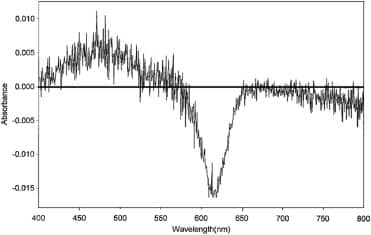
Further evidence of interactions between glycerol 50 M and solvatochromic dyes comes from experiments using the negatively solvatochromic dye BM and the positively solvatochromic dyes NR and PB (Figure 1). All these dyes produce significant difference spectra in the presence of potency. BM is particularly interesting as in all three solvents glycerol 50 M promotes aggregation. This is in contrast to that seen with ET30 and ET33. Aggregates are bathochromically shifted with respect to monomer in BM, so difference spectra show increases at longer wavelengths and decreases at shorter wavelengths. Figure 9 shows the difference spectrum in tert-butyl alcohol.
Figure 9
Difference spectrum of BM in tert-butyl alcohol with 50 μl of control added to the reference cuvette and 50 μl of glycerol 50 M added to the sample cuvette to make a total volume of 3 ml in each cuvette (see Materials and methods). BM is at a concentration of 125 μM. Difference spectrum consists of the sample spectrum minus the reference spectrum. Difference spectrum shown is that at t = 40 min after mixing. UV cuvettes used.
The difference spectra of NR and PB with glycerol 50 M in water, ethanol and tert-butyl alcohol indicate potency promotes disaggregation of both dyes in all three solvents.
Table 1 lists the effects of glycerol 50 M on the spectra of all the solvatochromic dyes examined in this study, giving the positions of maximum absorbance changes on addition of potency. It should be noted that for dyes ET30, ET33 and BM in water only single difference maxima are given. This is because all dyes absorb strongly and non-specifically below c. 400 nm, rendering accurate difference spectra beyond the capabilities of the instrument below this wavelength.
| Table 1Table showing the positions of maximum absorbance changes (difference spectra) on addition of glycerol 50 M to solvatochromic dyes used in this study in three different solvent systems. Absorbance maxima for dyes in respective solvents are given in plain type whilst the positions of maximum absorbance change on addition of potency are given in italics | ||||
| Dye | Solvent system | |||
| Water | Ethanol | Tert-butyl alcohol | ||
| Negatively solvatochromic | ||||
| ET33 | Absorbance maxima 409 nm (broad)
Decrease at c.380–420 nm with potency |
Absorbance maxima 472 nm (broad)
Increase at 442 nm and decrease at 542 nm with potency |
Absorbance maxima 548 nm (broad)
Increase at 490 nm and decrease at 615 nm with potency |
|
| ET30 | Absorbance maxima 450 nm (broad)
Decrease at c.450 nm with potency |
Absorbance maxima 550 nm (broad)
Increase at 450 nm and decrease at 580 nm with potency |
Absorbance maxima 650 nm (broad)
Increase at 580 nm and decrease at 715 nm with potency |
|
| BM | Absorbance maxima
380 nm (monomer) 442 nm (aggregate) Increase at 450 nm with potency |
Absorbance maxima
400 nm (monomer) 513 (aggregate) Decrease at 390 nm and increase at 510 nm with potency |
Absorbance maxima
402 nm (monomer) c.496 nm (minor peak) (aggregate) 576 nm (aggregate) Decrease at 404 nm and increase at 500 nm and 570 nm with potency |
|
| Positively solvatochromic | ||||
| BDN | Absorbance maxima
490 nm (aggregate) 614 nm (monomer) Increase at 460 nm and decrease at 620 nm with potency |
Absorbance maxima
484 nm (aggregate) 564 nm (overlapping aggregate and monomer peaks?) Increase at 480 nm and decrease at 615 nm with potency |
Absorbance maxima
459 nm (aggregate) 537 nm (aggregate) 609 nm (monomer) Increase at 460 nm and 535 nm and decrease at 620 nm with potency |
|
| NR | Absorbance maxima
530 nm (aggregate) 593 nm (monomer) Decrease at c.480 nm and increase at c.560 nm with potency |
Absorbance maxima 550 nm (broad) Decrease at c.475 nm and increase at c.560-570 nm with potency | Absorbance maxima 538 nm (broad) Decrease at c.465 nm and increase at c.560 nm with potency | |
| PB | Absorbance maxima 658 nm (broad) Decrease at c.520 nm and increase at c.670 nm with potency | Absorbance maxima
608 nm (broad) Decrease at c.540 nm and increase at c.680 nm with potency |
Absorbance maxima 601 nm (broad)
Decrease at c.520 nm Increase at c.620–660 nm with potency |
|
Table 2, in turn, gives a summary, derived from difference spectra, of the deduced supramolecular effects of glycerol 50 M on all dyes tested in all three solvents.
| Table 2Table showing the deduced effect of glycerol 50 M on the supramolecular chemistry (enhanced aggregation or enhanced disaggregation) of all solvatochromic dyes used in this study | ||||
| Dye | Effect of glycerol 50 M potency | |||
| Water | Ethanol | Tert-butyl alcohol | ||
| Negatively solvatochromic | ||||
| ET33 | Disaggregation | Disaggregation | Disaggregation | |
| ET30 | Disaggregation | Disaggregation | Disaggregation | |
| BM | Aggregation | Aggregation | Aggregation | |
| Positively solvatochromic | ||||
| BDN | Aggregation | Aggregation | Aggregation | |
| NR | Disaggregation | Disaggregation | Disaggregation | |
| PB | Disaggregation | Disaggregation | Disaggregation | |
Discussion
Evidence has been presented which demonstrates it is possible to develop a simple chemical system for the detection and investigation of homeopathic potencies. Solvatochromic dyes provide a versatile means of probing the nature and action of potencies in solution, providing several levels of information. Although results given are with glycerol 50 M only, a large number of homeopathic remedies in different potencies have been tested and their effects found to be similar to those presented here.
The intramolecular electron transfer feature of solvatochromic dyes appears to be essential for detecting the presence of potencies, as non-solvatochromic dyes do not display any spectroscopic changes when potencies are introduced. The interaction of potency and solvatochromic dye leads to supramolecular changes in the solution dynamics of all solvatochromic dyes tested, in the form of enhanced dye aggregation or disaggregation. Where potency causes a dye to exhibit enhanced aggregation this occurs in all three solvents tested, and similarly where a potency causes enhanced disaggregation, this occurs in all three solvents. This is in marked contrast to the situation in the absence of potency where aggregation occurs more strongly as solvent polarity decreases and disaggregation increases as solvent hydrogen bonding capability increases for all dyes tested. In addition the observation that potency causes some dyes to aggregate and others to disaggregate in the same solvent strongly suggests solvent does not mediate in the potency effects seen and that potency is interacting directly with the dyes.
Table 2 shows the aggregation/disaggregation effect of potency on all dyes tested in all three solvents. No pattern is immediately apparent. Whether a dye is positively or negatively solvatochromic does not in itself determine whether enhanced aggregation or disaggregation occurs. However, a more detailed examination of the data in Table 1 and the following observations may be relevant.
BM displays well-defined monomer and aggregate peaks in all solvents at the wavelengths shown in Table 1, indicating that dye solutions consist of two or three distinct species. The effect of potency appears to be to increase or decrease the relative proportions of these species. For instance, when glycerol 50 M is added to solutions of BM in tert-butyl alcohol difference spectra display three maxima – a decrease at c.404 nm, coincident with the absorbance maximum for BM monomer (402 nm), an increase at c.570 nm, coincident with the absorbance maximum for aggregate (576 nm) and a substantial increase at c.500 nm, coincident with a minor peak at c.496 nm, which may represent a smaller aggregate species than that at 576 nm (Figure 9). BM therefore seems to exist as distinct aggregate and monomer species in solution and this appears to be reflected in the difference spectra obtained with potencies.
In contrast other dyes such as ET33 and ET30 have spectra which are broad in all solvents, indicating a spectrum of populations from monomer through small aggregates to much larger aggregates. Despite this, when potency is added to these dyes, the difference maxima produced are distinct and, moreover, tend to lie well outside the absorbance peak of the dye. For instance, this can be clearly seen from the difference spectrum of ET30 in tert-butyl alcohol. ET30 has its absorbance peak at 650 nm, yet the difference maxima obtained on addition of potency occur at 580 nm and 715 nm (Figure 6). This would suggest that potency is causing highly aggregated dye to disaggregate to form monomer, rather than causing moderately aggregated dye to disaggregate to less moderately aggregated dye (which would lead to difference maxima that were closer together). In other words potency is acting discontinuously to cause the system to polarise, rather than simply shifting the aggregate equilibrium slightly. A slight shift in equilibrium rather than polarisation is what one might expect from an energetic point of view, so potencies appear to be doing something markedly unexpected in solution. Whilst the conversion of highly aggregated dye to monomer constitutes only some 1–2% of the total dye in the system based on absorbance values, this is a remarkable achievement given the supposedly ultra-weak nature of homeopathic potencies.
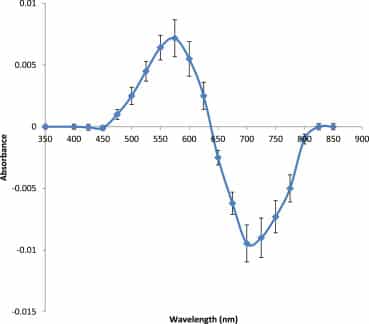
Some discussion is required at this point with regard to the reproducibility of the difference spectra given inFigure 4, Figure 6, Figure 7, Figure 9. Difference spectra for ET30 in tert-butyl alcohol have been performed in excess of 40 times over the course of several months and a composite spectra of all these runs is given inFigure 10, showing mean absorbance values at set wavelengths across the dye’s spectrum and associated error bars. This composite curve should be seen in relation to control difference spectra (50 μl of control solution added to both cuvettes; n = 20) where no discernible difference spectrum is observed (ie 0.000 ± 0.000) within the sensitivity and precision limits of the spectrophotometer used in this study (seeMaterials and methods). This difference between samples and controls is statistically highly significant (t-test, p ≤ 0.0001). Similar composite difference spectra can also be derived for ET33, BDN and BM, also giving highly significant p values of <0.0001.
Figure 10
Combined difference spectra (n = 20) of ET30 in tert-butyl alcohol with 50 μl of control added to the reference cuvette and 50 μl of glycerol 50 M added to the sample cuvette to make a total volume of 3 ml in each cuvette (see Materials and methods). ET30 is at a concentration of 245 μM. Difference spectrum consists of the sample spectrum minus the reference spectrum. UV cuvettes used. Error bars are to the first standard deviation. Control ‘difference’ spectra (n = 20) where 50 μl of control are added to both cuvettes display no discernible difference across the spectrum from 350 to 800 nm (ie 0.000 ± 0.000) giving a p value of <0.0001, indicating high statistical significance.
For dye BDN absorbance peaks are not as well separated or as distinct as in BM and it is likely that BDN exists as more than two, or even three, species in solution, but still fewer than is the case for ET30 and ET33. In the presence of potency, increases in levels of highly aggregated species are seen in all solvents, combined with a decrease at 615–620 nm, corresponding to that of monomer. As with ET30 and ET33 therefore potency seems to be acting discontinuously, in this case by preferentially converting monomer to highly aggregated dye, rather than stimulating a slight shift in overall equilibrium.
Dyes NR and PB both display enhanced disaggregation in the presence of potencies, with decreases in levels of hypsochromic species and increases in levels of bathochromic species relative to the dyes’ absorbance maxima (Table 1). These two dyes are significantly less strongly solvatochromic than ET33, ET30, BM and BDN however and consequently it is not possible to determine in any more detail solution dynamics using current instrumentation.
Returning to the issue of why some dyes display enhanced aggregation, and some disaggregation, in the presence of potencies, additional explanations may be down to particular structural features of the dyes in question and related dye dissociation constants (KD). As yet no exact figures are available for KD values but plots of absorbance peaks versus dye concentration (a standard method of determining KD) indicate both BDN and BM aggregate weakly whereas ET30, ET33, NR and PB aggregate more strongly. Examination of further solvatochromic dyes should clarify which of the above factors determine whether enhanced aggregation or disaggregation occurs in the presence of potencies.
Much work needs to be done to confirm and extend the results described in this paper. In particular the detailed effects of a range of potencies of glycerol need to be established. Preliminary results indicate that degree of effect is not smoothly related to the degree of potentisation. A similar discontinuity in the effect of potency scales has been observed by workers using other systems.27
Two of the dyes employed in this study (BM and NR) are fluorescent and fluorescence spectroscopy should add valuable information about the way in which potencies are affecting aggregation and disaggregation dynamics, particularly for BM which displays a marked dependence of fluorescence intensity on aggregation levels.
It is also hoped that the identification of the essential features of solvatochromic dyes necessary to interact with potencies will allow the synthesis of derivatives of dyes displaying ever greater spectroscopic changes. This in turn will mean more searching questions can be asked.
All solvatochromic dyes are essentially oscillating dipoles. Under the influence of light an electron travels from one end of the dye to the other and back again several hundred million times a second. It is conceivable that potencies are interacting with solvatochromic dyes because they themselves are oscillating dipoles. It is hoped that it will in time be possible to elucidate the nature of homeopathic potencies using the chemical system described herein.
Conclusions
Solvatochromic dyes appear to provide a simple and versatile method for the detection of homeopathic potencies. Six different solvatochromic dyes have been tested in three different solvents with and without potency present. In all cases the visible spectra of the dyes are affected by the presence of potency. Evidence has been presented which indicates potencies are interacting directly with the dyes themselves rather than through the solvent in which the dyes are dissolved. Potencies have no effect on non-solvatochromic dyes, suggesting it is the intramolecular electron transfer feature of solvatochromic dyes which is essential in determining dye–potency interaction. In turn this interaction results in supramolecular effects in the form of enhanced dye aggregation or disaggregation. This evidence has taken the form of difference spectra together with assays involving the interaction of dyes with strontium ions.
As solvatochromic dyes are oscillating dipoles it is possible that homeopathic potencies share some commonality with this feature. It is hoped that future research utilising solvatochromic dyes will allow specific questions to be asked regarding the physico-chemical nature and mode of action of homeopathic potencies.
Materials and methods
Materials
Solvatochromic dyes 2,6-Dichloro-4-(2,4,6-triphenylpyridinium-1-yl)-phenolate (ET33), 2,6-Diphenyl-4-(2,4,6-triphenylpyridinium-1-yl)-phenolate (ET30), N,N-dimethylindoaniline/Phenol Blue (PB), 9-diethylamino-5H-benzo[α]phenoxazime-5-one/Nile Red (NR) and 4-(Bis-(4-(dimethylamino)phenyl)methylene)-1(4H)-naphthalenone (BDN) were obtained from Sigma Aldrich UK and used as provided. 4-[(E)-2-(1-methylpyridinium-4-yl)ethenyl]phenolate/Brooker’s merocyanine (BM) was synthesised and provided by Innovapharm Ltd., Kiev, Ukraine and its structure and purity confirmed by NMR. Non-solvatochromic dyes Patent Blue VF, Green S and Sulforhodamine 101 were obtained from Sigma Aldrich or Fisher Scientific, UK. Strontium chloride, ethanol and tert-butyl alcohol were obtained from Sigma–Aldrich, UK unless specified otherwise and were of the highest purity available.
Reverse osmosis water (ROW) was used throughout this study and had a resistivity of 15 M Ω cm (checked daily).
Disposable high purity optical PS cuvettes 4.5 ml capacity/10 mm pathlength with polyethylene (PE) air-tight stoppers were obtained from Elkay Laboratory Products, UK. Disposable high purity UV 4.5 ml capacity/10 mm pathlength cuvettes (UV) with PE air-tight stoppers were obtained from Brand GMBH, Germany. High purity/low leachable trace element Nalgene PE, and fluorinated ethylenepropylene (FEP) bottles were obtained from Fisher Scientific, UK.
Solution storage
All solutions were made up and stored in FEP or PE bottles. Ethanol and tert-butyl alcohol were used from the bottles in which they were provided or transferred to PE bottles. Concentrated dye stocks in dimethylsulfoxide (DMSO) or dye solutions in ROW, ethanol or tert-butyl alcohol were stored in FEP or PE bottles at room temperature. Working dye solutions were made by dissolving dye directly into solvent (ROW, ethanol or tert-butyl alcohol) or by adding an aliquot of concentrated dye stock in DMSO to solvent. In both cases dye solutions were left to equilibrate overnight before use. Concentrations of dyes used for difference spectra and assays with strontium ions were as follows: ET33 – 245 μM in ROW, ethanol and tert-butyl alcohol; ET30 – 245 μM in ethanol and tert-butyl alcohol. ET30 is poorly soluble in ROW. Solutions were made up in 20 mM borate buffer pH 10 and centrifuged to remove any precipitate; BDN – 80 μM in ROW, ethanol and tert-butyl alcohol. BDN slowly precipitates in ROW so solutions were made up and used within an hour; BM – 125 μM in ethanol, 245 μM in ROW and 125 μM in tert-butyl alcohol; NR – 16 μM in ROW, ethanol and tert-butyl alcohol; PB – 62 μM in ROW, ethanol and tert-butyl alcohol. Dye concentrations were chosen so as to give absorbances of c.1.0 at their absorbance maxima. At this absorbance level the Unicam UV-500 spectrophotometer used in this study can comfortably handle difference spectra. No light or temperature induced degradation (determined by daily monitoring of visible spectra) of ET33, ET30, PB, BDN, BM or NR was observed over the period of this study. As a precaution solutions of BM and NR were kept in the dark as both dyes are fluorescent and potentially subject to light-induced degradation.
Homeopathic potencies
Serially diluted and succussed solutions (homeopathic potencies) of a range of compounds, including glycerol 50 M, were obtained from Helios Homeopathy Ltd Tunbridge Wells, UK or Ainsworths Homeopathic Pharmacy, London, UK and were diluted 10 fold into 90% ethanol/10% ROW to ensure consistent solvent composition. Potencies are sold as made in 90% ethanol/10% ROW, but the above step was taken as a further precaution to ensure solvent equivalence with control solutions. Glycerol 50 M is a homeopathic potency prepared by the Korsakoff method.28 Glycerol is serially diluted and succussed by hand up to the 200c potency. Thereafter potentisation is performed mechanically. At each step a portion of solution is first diluted 100 fold, and then subjected to 10 succussion strokes. 50 M means the homeopathic medicine has gone through 50,000 such cycles. A total of 500,000 succussion strokes have therefore been imparted with an effective dilution factor of 10050000. ROW is used throughout the potentisation process.
Control solutions
Controls consisted of 90% ethanol/10% ROW alone. Both potencies and controls were kept in the same 3.5 ml amber moulded glass bottles used by Helios Homeopathy Ltd (obtained from the Homeopathic Supply Company Ltd, code MPB) under the same conditions at room temperature. Any impurities leaching out of the glassware should therefore be present at the same levels in both control and potency solutions. Independent analysis by ICP-OES for levels of Aluminium, Boron, Calcium, Iron, Potassium, Magnesium, Sodium, Silicon and Titanium by LGC Health Sciences, UK confirmed the same level of each element in both control and potency bottles (kept at room temperature for 6 months prior to analysis). Furthermore, at the levels of elements detected (all <2 μg/ml) no effect on either dye difference spectra or assays involving dyes and strontium ions (see Experimental procedures below) was observed in an independent study in which salts of the above elements were deliberately added to assay solutions. Levels of other elements in control and potency solutions were below the detection limits of the ICP-OES instrumentation.
Instrumentation
Assays and spectra were recorded on a Unicam UV500 UV/VIS double-beam spectrophotometer run with preloaded Visionlite software capable of analysing curves and providing data points at set time intervals as they are generated. Manufacturer’s specifications state an accuracy of ±0.001 at an absorbance of 1.0, with a wavelength accuracy of 0.5 nm and resolution of 2 nm.
Experimental procedures
Assays were performed in single-use 4.5 ml capacity PS cuvettes (assays in ROW and ethanol) or UV cuvettes (assays in ethanol and tert-butyl alcohol) as described above. Tests showed no evaporation of contents occurred over the time periods of assays from either cuvette type using the stoppers described.
Difference spectra of dyes were performed as follows. 2.95 ml of dye solution at concentrations giving an absorbance of c.1.0 (see Solution storage above) was dispensed into sample and reference cuvettes and the spectrophotometer set to zero across the range of wavelengths to be assessed (generally 350 or 400 nm to 700 or 800 nm depending upon the dye being investigated). 50 μl of control solution (90% ethanol/10% ROW) was then added to the reference cuvette and 50 μl of potency solution (90% ethanol/10% ROW) added to the sample cuvette. Solutions in both cuvettes are therefore materially identical. Solutions were scanned at time intervals up to 3 h to give difference spectra of dye with potency minus dye with control. Scans of solutions left overnight were also performed.
‘Difference’ spectra in which 50 μl of control solution was added to both cuvettes demonstrated no effect over comparable time scales compared with dye solutions to which potency had been added.
Assays with strontium ions were carried out as follows. To 2.95 ml of dye solution in PS or UV cuvettes was added 50 μl of either control solution or potency solution together with, in both cases, 1 μl of a 210 mM solution of strontium chloride. Absorbance was then followed at a set wavelength to monitor changes in the interaction of dye with strontium ions. The spectrophotometer was zeroed with solvent alone in the respective cuvette type prior to assay. All presented data show average values of at least 20 assays under any one set of conditions.
PS or UV cuvettes have been used throughout this study. Primarily this is because they are single-use and therefore remove any possibility for cross-contamination by residual potency potentially encountered with reusable quartz cuvettes (see text). Laboratory temperature was maintained at 21 ± 1°C for assays involving ROW and ethanol and 24 ± 1°C for assays involving tert-butyl alcohol, using a combination of background heating and air conditioning.
The highest purity ROW, ethanol and tert-butyl alcohol were used in all assays. It is perhaps worth noting however that on addition of 50 μl of either control or potency solutions to 2.95 ml of dye solution the relative levels of host solvent drop slightly because of the introduction of 45 μl of ethanol and 5 μl of ROW. Bulk solvent is therefore at a level of 98.5% in the case of those assays performed in ROW, 99.8% in the case of those assays performed in ethanol and 98.33% in the case of those assays performed in tert-butyl alcohol. As these small changes occur in both control and sample solutions they have not been taken into account, especially as potency effects appear to be solvent-independent (see text).
Conflict of interest statement
No source of funding had any influence on the design, analysis, interpretation or outcome of the research contained within this manuscript, nor on the writing of the manuscript.
Acknowledgements
Primary funding for this work is gratefully acknowledged from The Homeopathy Research Institute.
Additional funding from the following individuals, institutions and companies in alphabetical order is also gratefully acknowledged – Alliance of Registered Homeopaths, European Central Council of Homeopaths, Helios Homeopathy Ltd., Jane Milburn, The Tanner Trust and Charles Wansbrough.
Grateful acknowledgement also to Alexander Tournier for helpful discussions and comments during the compilation of this manuscript.
References
1. Reichardt, C. and Welton, T. Solvent effects on the absorption spectra of organic compounds. in:Solvents and Solvent Effects in Organic Chemistry. 4th edn. Weinheim.Wiley-VCH, ; 2011: 359–424
2. Wolf, U., Wolf, M., Heusser, P., Thurneysen, A., and Baumgartner, S. Homeopathic preparations of quartz, sulphur and copper sulphate assessed by UV-spectroscopy. Evid Based Complement Altern Medorrhinum 2011; : 644–654
3. Aabel, S., Fossheim, S., and Rise, F. Nuclear magnetic resonance (NMR) studies of homeopathic solutions. Br Homeopath J. 2001; 90: 14–20
4. Rey, L. Thermoluminescence of ultra-high dilutions of lithium chloride and sodium chloride. Phys A Stat Mech Appl. 2003; 323: 67–74
5. Assumpcao, R. Electrical impedance and HV plasma images of high dilutions of sodium chloride.Homeopathy. 2008; 97: 129–133
6. Elia, V., Baiano, S., Duro, I., Napoli, E., Niccoli, M., and Nonatelli, L. Permanent physico-chemical properties of extremely diluted aqueous solutions of homeopathic medicines. Homeopathy. 2004; 93:144–150
7. Belon, P., Elia, V., Elia, L., Montanino, M., Napoli, E., and Niccoli, M. Conductometric and calorimetric studies of the serially diluted and agitated solutions. On the combined anomalous effect of time and volume parameters. J Therm Anal Calorim. 2008; 93: 459–469
8. Bell, I.R. The evolution of homeopathic theory-driven research and methodological toolbox. Am Homeopath. 2008; 14: 56
9. Montagnier, L., Aissa, J., Ferris, S., Montagnier, J.-L., and Lavallee, C. Electromagnetic signals are produced by aqueous nanostructures derived from bacterial DNA sequences. Interdiscip Sci Comput Life Sci. 2009; 1: 81–90
10. Yin Lo, S., Geng, X., and Gann, D. Evidence for the existence of stable-water-clusters at room temperature and normal pressure. Phys Letts A. 2009; 373: 3872–3876
11. Marchettini, N., Del Giudice, E., Voeikov, V., and Tiezzi, E. Water: a medium where dissipative structures are produced by a coherent dynamics. J Theor Biol. 2010; 265: 511–516
12. Milgrom, L.R. Entanglement, knowledge, and their possible effects on the outcomes of blinded trials of homeopathic provings. J Altern Complement Medorrhinum 2006; 12: 271–279
13. Bellavite, P. Complexity science and homeopathy: a synthetic overview. Homeopathy. 2003; 92:203–212
14. Torres, J.L. and Ruiz, M.A.G. Stochastic resonance and the homeopathic effect. Br Homoeopath J.1996; 85: 134–140
15. Speight, P. Homoeopathy: a home prescriber. C W Daniel Co Ltd, UK; 1992
16. Hahnemann S. Organon of medicine. 5th and 6th edns.
17. Reichardt, C. Pyridinium N-phenolate betaine dyes as empirical indicators of solvent polarity: some new findings. Pure Appl Chem. 2008; 80: 1415–1432
18. Reichardt, C. Solvatochromic dyes as solvent polarity indicators. Chem Rev. 1994; 94: 2319–2358
19. Banfield, P. and 20. Wurthner, F., Yao, S., Debaerdemaeker, T., and Wortmann, R. Dimerization of merocyanine dyes. Structural and energetic characterization of dipolar dye aggregates and implications for nonlinear optical materials. J Am Chem Soc. 2002; 124: 9431–9447
21. Cartwright SJ. Unpublished results 2015.
22. Eisfeld, A. and Briggs, J.S. The J- and H- bands of organic dye aggregates. Chem Phys. 2006; 324:376–384
23. Mishra, A., Behera, R.K., Behera, P.K., Mishra, B.M., and Behera, G.B. Cyanines during the 1990’s.Chem Rev. 2000; 100: 1973–2012
24. Balzani, V., Ceroni, P., and Juris, A. Photochemistry and photophysics: concepts, research, applications. Wiley-VCH Weinheim, Germany; 2014
25. Reichardt, C. Solvatochromism, thermochromism, piezochromism, halochromism and chiro-solvatochromism of pyridinium N-phenoxide betaine dyes. Chem Soc Rev. 1992; 21: 147–153
26. Chaplin, M. The memory of water: an overview. Homeopathy. 2007; 96: 143–150
27. Davenas, E., Beauvais, F., Amara, J. et al. Human basophil degranulation triggered by very dilute antiserum against IgE. Nature. 1988; 333: 816–818
28. http://www.helios.co.uk/remedies/remedy-preparation.
Reprinted from:
http://www.homeopathyjournal.net/article/S1475-4916(15)00062-4/references



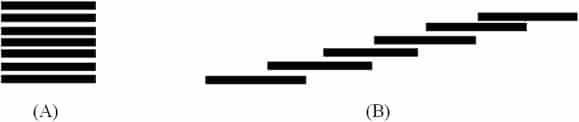


Thank you Dr. Cartwright. For the first time in history we have a simple method to detect homeopathic potencies. This is a huge boon to homeopathy on many levels. It has more uses than we can imagine at the moment. I hope the test is made widely available so its potential to serve homeopathy can be realized.
Congratulations for this research. With it we have one more proof of the existence of something in homeoremedies.
Now, from my Programmatic Medicine approach (https://hpathy.com/homeopathy-papers/homeopathy-a-functional-programmatic-medicine/ ) the molecular way homeoremedies act is not the most important aspect of homeopathy, but what they produce at the end, and that is triggering to work healing programs through a nervous system reaction of the information homeoremedies have. The question is now: how this molecular aspect of homeoremedies infers in nerves producing the CNS- brain healing reaction? Let’s remember Master Hahnemann knew the homeoremedies influence in nerves: Organon; paragraphs 16, 272 and 247. The Chronic Diseases; Pages 45 and 49, an aspect, as far as I know, only I have being studying.