Orientation of Water Molecules Explains Conduction of Electrical Energy in Water or in Aqueous Solutions
The principle behind the efficacy of homeopathic dilution is the conveyance of electrical charge possessed by matter through different orientations of water 1,2. It remained a question as to how the electrical charge is conveyed through water. Ions as the answer cannot explain why even non-ionic solutions conduct and retain electrical energy3. The magic of Homeopathy lies with orientation of water molecules, so we herein performed the following experiments to confirm that electrical energy in water and in aqueous solution is conveyed not through ions, but orientation of water molecules.
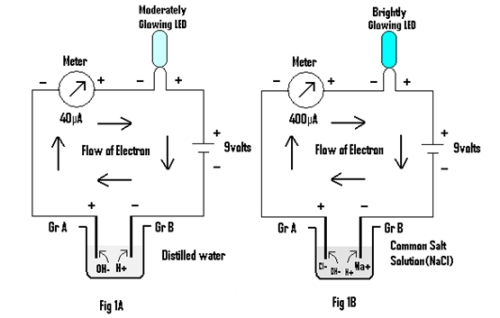
Experiment 1: In this experiment a meter, LED and a 9 volt battery is connected in series through a pair of Graphite electrodes placed in distilled water & salt solution as shown in Fig 1A & 1B respectively.
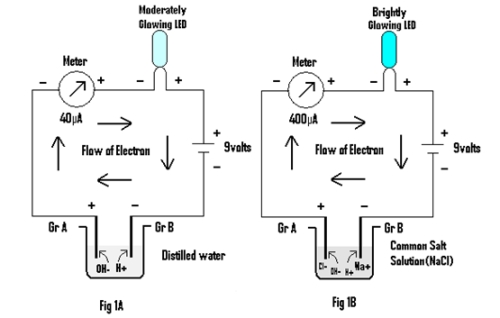
Result & Discussion:
In both the cases LED is found to glow, but the brightness of the LED is greater in the latter than in the former. Also the value of the current in the latter (400 µA) is greater than in the former(40 µA). The conduction of electrical energy being attributed to the presence of ions in both distilled water and in salt solution .The H+ ions (cations ) & OH– ions(anions) in the former move towards the Cathode (Gr B,i.e negative electrode) & Anode (Gr A,i.e positive electrode) respectively, while in the latter, Na+ & Cl– ions move toward the Cathode and Anode along with H+ and OH– ions of water itself during conduction of electrical energy. Thus it seems that ions conduct electrical energy by getting neutralized at the oppositely charged electrode.
In order to see whether the ions actually move to their oppositely charged electrodes while conducting electrical energy, the following experiment is performed:
Experiment 2:
Case 1:
A circuit is prepared as shown in Fig 2A (i.e through distilled water) and two Graphite electrodes (Gr C & Gr D) are placed just opposite to Graphites A & B (i.e Gr A & Gr B). The Gr C & Gr D generates emf of about 3.35 volts. A LED is also found to glow, showing flow of current of about 14µA as in Fig2B.
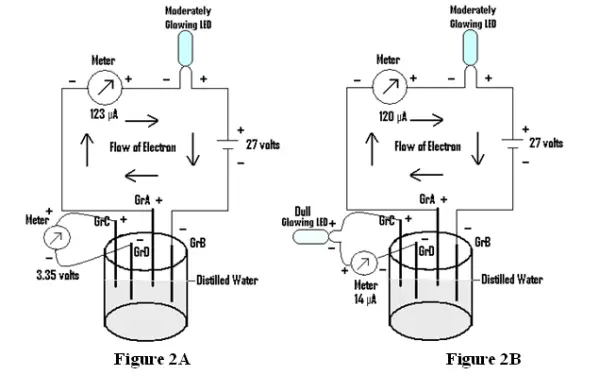
Case 2:
A circuit is prepared as shown in Fig 2C (i.e through distilled water) and two Graphite electrodes (Gr C & Gr D) are placed just opposite to Graphites A & B (i.e Gr A & GrB). The emf of about 9volts is applied at Gr A & Gr B and the emf shown by Gr C &Gr D is found to be 1.07volts. Now the application of emf at Gr A & Gr B is stopped and Gr C & Gr D is connected to a source of battery( here water battery is used) generating emf of about 1.02 volts. The emf shown by Gr C & Gr D is measured, as Initial emf, and found to be 0.94volts(Fig 2D). Next the emf of Gr C & Gr D is measured, as Final emf on application of 9 volts at Gr A & Gr B and is found to be 1.06 volts. But on reversing the polarity of applied emf at Gr C & Gr D(Fig 2E) , the final emf shown by the Gr C & Gr D is found to be –0.84 volts.
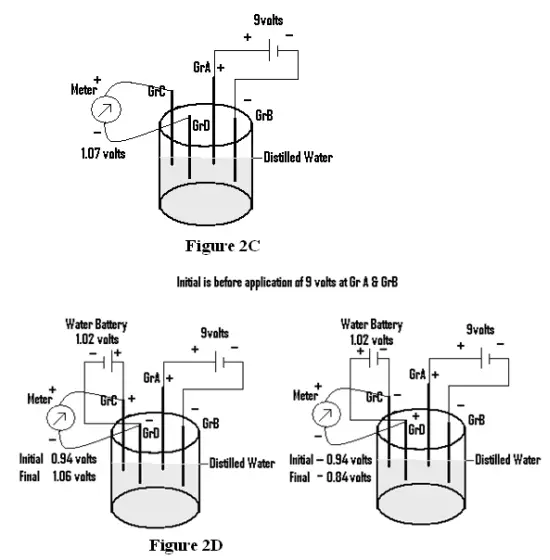
(Figure 2C, Figure 2D & Figure 2E)
Discussion:
How and why the ions preferably go to a particular Graphite electrode rendering it some particular charge ( i.e Gr C ( against Gr A) is positively charged while Gr D(against Gr B is negatively charged), is not explained so far for the cases shown in Fig 2A,2B &2C.
Moreover the final emf generated by Gr C & Gr D pair in respect of figures 2D &2E for case 2 are found to be different when polarity of Gr C & Gr D is reversed. If the resulting flow of electrical energy through water would be only due to approach of ions to their oppositely charged electrodes (i.e cations to cathode & anions to the anode),to get themselves neutralized, the resultant emf of Gr C & Gr D would have been the same irrespective of the polarity of applied emf at Gr C & Gr D. So it again reflects that the approach of ions is not the actual cause of conduction of electrical energy through water.
It was already shown by us earlier that electrical energy can flow through water simultaneously in the same or opposite direction.4 Herein we shall be considering only the case where electrical energy flows through water in the opposite direction.
Experiment 3:
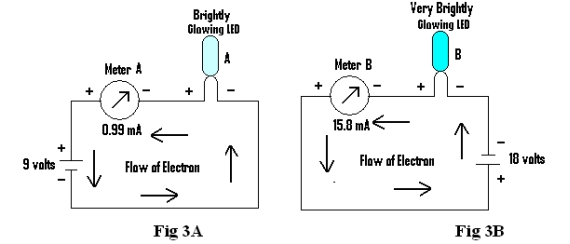
Two different circuits are prepared with a battery, LED and a meter for measuring current connected in series as shown in Fig 3A & 3B.The batteries being 9 volts and 18 volts respectively. The circuits are subjected to different conditions and readings of Meter A, Meter B, and type of brightness of LED A & LED B is recorded in table below.
Condition 1: When the circuits are separate (Fig 3A & 3B)
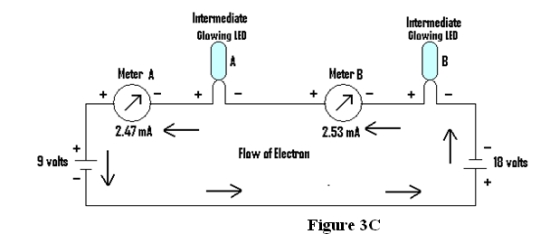
Condition 2: When the circuits are amalgamated (Fig 3C).
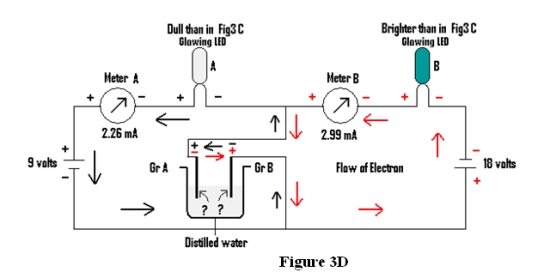
Condition 3: When the amalgamated circuit is connected by Graphites through distilled water (Fig 3D).
| Conditions | Brightness of LED | Meter readings (in mA) | ||
| LED A | LED B | Meter A | Meter B | |
| Condition 1 | Brightly | Very Brightly | 0.99mA | 15.8mA |
| Condition 2 | Intermediate | Intermediate | 2.47mA | 2.53mA |
| Condition 3 | Dull than in 3C | Brighter than in 3C | 2.26mA | 2.99mA |
Discussion:
In this case(Fig 3D) it is found that for the circuit containing LED “A” the flow of electrons is from Graphite B to Graphite A, while for the other circuit containing LED B, it is from Graphite A to Graphite B. If the conduction of electrical energy would be due to approach of ions to their opposite electrodes, for LED “A” H+ ions would have gone to Graphite B(Cathode) & OH- ions to Graphite A(Anode) while for LED “B” the H+ ions would have gone to Graphite A(Cathode) & OH- ions to Graphite B(Anode) for simultaneous conduction of electrical energy in opposite direction for glowing of both LED “A” & LED “B” . Moreover, when Distilled water is replaced by salt solution almost the same result is obtained as for condition 3 above, with meter readings of 1.52mA & 4.26mA respectively. So it reflects that both the Graphites are behaving as Cathode & Anode simultaneously and some Cations &Anions are preferring Graphite A while some others Cations &Anions are preferring Graphite B!!
Now we present a most interesting experiment wherein we have shown that even two different negative electrodes in distilled water show a sufficient emf and are able to light a LED.
Experiment 4:
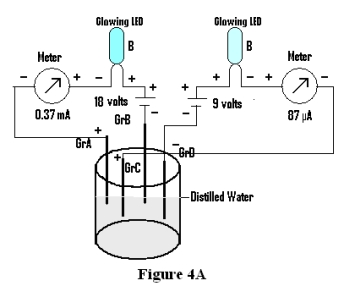
In this experiment four Graphite electrodes are placed in a beaker containing distilled water and emf of each pair is measured and recorded as Initial emf in table below. Now a circuit containing a battery of 18 volts , a meter for measuring current and an LED is connected in series through Graphite A & Graphite B placed in the distilled water. Another circuit is prepared through Graphite C &Graphite D connected through meter and a battery of 9 volts . Both LED’s are found to glow (Figure 4A).
Now emf of all pairs of Graphite are measured and recorded as Final emf in table …..
| Graphite Pairs | Gr A-GrB | Gr A-GrC | Gr A-GrD | Gr B-GrC | Gr D-GrB | Gr C-GrD |
| Initial emf | 11 mV | -13mV | 12mV | -22mV | -1mV | 10mV |
| Final emf | 12.24 volts | 3.26volts | 7.72volts | -10.68volts | 5.83volts | 4.71volts |
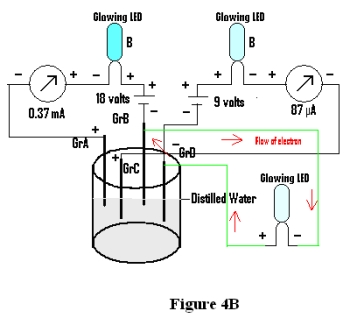
Now a LED is connected to the two negatively charged graphites ( Gr D & Gr B), as shown in Fig 4B, and the same is found to glow.
Discussion:
It is found that initial emfs shown by all the pairs of Graphite electrodes changes when they are connected to circuits. Moreover the emf shown by two “negative” electrodes definitely suggests that generation of emf is due to relative difference in charges between the two electrodes 5. The difference in charges of the electrode in turn is due to different orientations of water molecules in the vicinity of the particular electrode.
The glowing of LED when connected with Gr D & Gr B cannot be explained by the theory of conduction of electrical energy by ions in solution, as both being negatively charged, it is hardly possible that cations will prefer one and anions the other. It is actually due to the fact that different zones 6 are formed within water due to the presence of different electrodes along with their charge, if any, due to applied emf. Therefore, the relative difference in energy content of these zones are responsible for flow of electrical energy between any two zones when connected externally.
Inference:
It is not ions but the different orientations of water molecules that conducts electrical energy in water or in aqueous solution by the formation of zones.
That is the key to homeopathic dilution.
Reference:
[1]. The electrical energy of substances –the secret of homeopathy, Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXII, Summer Fall/ 2009 www.hanp.net)
[2] The Homeopathic Dilution-A New Explanation, Ruhul Amin, Biplab Chakraborty & Farook Rahaman (Simillimum,vol.XXI, Summer Fall/2008). www.hanp.net
[3] Solutions Ionic or Non-ionic Conducts & Retain Electrical Energy Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone July 2012 hpathy.com)
[4] The flow of electrical energy through metal and water-A new concept Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone Aug 2012 hpathy.com
[5]. Generation of electrode Potential of an electrode –an explanation, Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXIII.2010, www.hanp.net)
[6] The science behind retention of electrical energy by water, Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone Sep 2012 hpathy.com)




I find this study as quite informative. as engineers we always worked with quantitative approach in electro chemistry. But in homeopathy the approach is on the property of material in water when homeopathic process of dilution is employed. It is indeed a good observation in proving the homeopathic philosophy of treatment scientifically. This has enhanced my faith in homeopathy.
Sir,
Thanks for your inspiring comment.
Can the authors explain the relevance of all this to homeopathic remedies presented as dry tablets?
Also, would any homeopath like to take 6 remedies in identical bottles, but unlabelled, and correctly differentiate between them by whatever means they like?
Consider figure 3D.
Have you tried replacing the distilled water / salt solution with a selection of resistors?
Do you get the same effect?
Have you tried putting a volt meter across the distilled water? What does the +/- value of that number tell you about the direction of the current in the water?
Sir,
Water being the important constituent of Homeopathic medicines we have concentrated our work strictly on water itself.
For the second query please refer to our previous works specially on
1) “Generation of electrode potential of an electrode –an explanation” published in SIMILLIMUM (www.hanp.net) also available in the website of National Research Council of Canada(NRC CISTI)
2) “The electrical energy of substances the secret of homeopathy” published in SIMILLIMUM(www.hanp.net) also available in the website of National Research Council of Canada(NRC CISTI) and British Library Direct.
Thanks
Well water is not a constituent of the overwhelming majority of homeopathic remedies offered for sale. Are you saying they have a different mode of action from aqueous presentations?
Would you care to accept the challenge to differentiate between 6 unlabelled remedies? No? I thought not.
Les Rose doesn’t seem to understand homeopathy or its pharmacology, and instead, he prefers to ignore the above research as well as the hundreds of basic science trials and controlled clinical trials that show biological activity and clinical efficacy from homeopathic medicine. As for Les’ idea for a study, he might benefit from reading this article and review of research in one of the most respected material science journals in which the authors review a study they conducted using 3 different types of spectroscopy to detect nanoparticles remaining in homeopathic solutions at the 6C, 30C and 200C potencies. Therefore, people who say that there is “nothing” remaining in homeopathic medicines in the high potencies are simply wrong or un-informed (or both).
Reference:
http://www.ncbi.nlm.nih.gov/pubmed/23083226