2017 Independent Research Paper – Canadian College of Homeopathic Medicine
Table of Contents
- INTRODUCTION………………………………………………………………………………….2
- UNDERSTANDING SLEEP APNEA
CHAPTER 1: UNDERSTANDING SLEEP APNEA
- Definition & Types……………………………………………………………………….3
- Incidence & Prevalence…………………………………………………………………6
- Predisposing Factors…………………..………………………………………………..7
- Conventional Treatment of OSA and CSA in adults………………………………….9
- Conventional Treatment of OSA in children…………………………………………….11
CHAPTER 2: CAUSATION or ETIOLOGY IN CSA
- Freeze Response………………………………………………………………………13
- Post-Traumatic Stress…………………………………………………………………14
- The PTSD-Limbic System-Breathing Connection………………………………….15
- Breathing-Related Trauma……………………………………………………………16
- Head Trauma…………………………………………………………………………..19
- HOMEOPATHIC TREATMENT OF SLEEP APNEA
CHAPTER 3: SIGNS &SYMPTOMS & BEYOND
- Symptoms of OSA and CSA…………………………………………………….……22
- Symptoms specific tochildren………………………………………………………..23
- Beyond signs & symptoms………………………………………..…………………..30
- Flow-chart………………………………………………………………………………31
CHAPTER 4: RUBRICS & REPERTORIZATION
- Rubrics for Symptoms…………………………………………………………………33
- Rubrics and Remedies for Causation……………………………………………….36
CHAPTER 5: REMEDIES & DIFFERENTIAL DIAGNOSIS
- Remedies……………………………………………………………….………………38
- Differential diagnosis…………………………………………………………………..53
- SURVEY RESULTS……………………………………………………………………………54
- ..………………………………………………………………………………..56
- REFERENCES………………………………………………………………………………….58
- ONLINE RESOURCES………………………………………………………………………..66
- APPENDICES…………………………………………………………………………………..67
INTRODUCTION
Understanding the difference between obstructive sleep apnea (OSA) and central nervous system sleep apnea (CSA) can help homeopaths differentiate among the remedies that may be indicated. This is because the two types of sleep apnea are very different in origin. OSA occurs when the airway is narrowed or partially blocked e.g. when tissues at the back of the throat or the posterior soft palate over-relax or collapse during sleep or when enlarged tonsils or adenoids partially block the airway. CSA can occur for various reasons, one of which is related primarily to the breathing centre in the brainstem, and is not well understood by scientists despite considerable research.
Pioneering work by psychologist, Dr Damaris Drewry, has revealed that there is a type of CSA that is much more common than previously thought, and that it is often based on one or more traumatizing events. The effects of the trauma are imprinted in the autonomic nervous system as part of the fight/flight/freeze response and may lie dormant for varying periods of time until triggered by life stresses, resulting in the appearance of sleep apnea symptoms. By addressing these traumatizing events, Dr Drewry has been able to successfully resolve many cases of CSA.
While various techniques are used in homeopathic case analysis (e.g. totality of symptoms, keynotes, essence, organ specificity, or a combination of these), there are instances where causation is the only key that can unlock the puzzle of the case. Knowing the underlying cause of our patients’ symptoms can be invaluable in helping to guide us towards the correct group of remedies, especially when the seemingly well-indicated remedy (based solely on symptoms but not taking causation into consideration) fails to work.
While CPAP machines and intra-oral appliances may work for some patients, these are only symptomatic treatment options that do nothing to address the underlying root causes of the problem. In homeopathic terms, they are merely palliative as opposed to being curative. Knowing the cause of the problem may allow the homeopath to cure the patient who could otherwise be at increased risk of developing serious health complications such as hypertension and consequent cardiovascular morbidity.
As the diagnosis of sleep apnea has become more prevalent in recent years, research into this topic would be of increasing value as more people seek alternative or complementary help in the coming years, especially when Dr Drewry’s work* becomes more widely known and the general public comes to realize that there is the possibility of cure in many cases of trauma-basedCSA.
*Information about Dr Drewry’s work can be found at BeyondTalkTherapy.com and SleepApneaSolution.org.
CHAPTER 1: UNDERSTANDING SLEEP APNEA
Definition of Sleep Apnea
The word “apnea” derives from Greek etymology and means “without breath“. Sleep apnea is the temporary cessation or suspension of breathing that occurs involuntarily and repeatedly during sleep.
Types of Sleep Apnea
There are 4 types of sleep apnea:
- obstructive sleep apnea (OSA),
- central sleep apnea (CSA),
- mixed sleep apnea (patient presenting with symptoms of both OSA and CSA), and
- complex sleep apnea (patient ending up with symptoms of both OSA and CSA following treatment)1,2.
Obstructive sleep apnea (OSA)
A disorder, first described in 1965, characterized by recurrent interruptions of breathing during sleep due to temporary obstruction of the airway by lax, excessively bulky, or malformed pharyngeal tissues (soft palate, uvula, and sometimes tonsils), with resultant hypoxemia and chronic lethargy. Sleep in the supine position predisposes apneic episodes.
Stedman’s Medical Dictionary © Wolters Kluwer Health.
OSA always involves blockage of the airway but there is continued respiratory effort3. The blockage is due to soft tissue sat the back of the throat collapsing and closing off the airway intermittently during sleep, or when enlarged tonsils and/or adenoids partially block the airway.The patient snores loudly or there are breathing pauses and, depending on the severity of the condition, either awakens partially or completely with a snort or gasping, smothering or choking.
Central sleep apnea (CSA)
Central sleep apnea (CSA) is characterized by a lack of drive to breathe during sleep, resulting in repetitive periods of insufficient ventilation and compromised gas exchange.4It comprises a heterogeneous group of conditions characterized by repeatedly suspended respiratory effort that is involuntary. In CSA, the problem arises primarily in the respiratory centre in the brainstem that is responsible for normal breathing or secondary to other conditions such as diseases, drugs (esp. chronic opioid use5,6), or high altitude.
The American Academy of Sleep Medicine’s International Classification of Sleep Disorders, 3rd edition (ICSD-3), distinguishes 5 subtypes of central sleep apnea (CSA) syndromes in adults:
- primary CSA,
- Cheyne-Stokes breathing-CSA pattern (e.g. patients with heart failure or stroke),
- CSA due to medical conditions other than Cheyne-Stokes,
- CSA due to drugs (narcotics/opioids), and
- high altitude-induced periodic breathing.
Pathophysiology7,8.9
Normal ventilation is regulated to keep arterial oxygen (PaO2) and carbon dioxide (PaCO2) levels within narrow ranges. This is achieved by feedback loops that involve chemoreceptors, intrapulmonary vagal receptors, respiratory control centers in the brain stem, and respiratory muscles. During wakeful periods, signals from the cerebral cortex influence respiration. This is known as behavioral control and involves nonchemical stimuli such as pulmonary mechanoreceptors. During sleep, behavioral control is lost and chemical control is the major mechanism regulating ventilation, with PaCO2 being the major stimulus. Reduction of PaCO2of just a few mmHg below the apneic threshold can result in apneas.
Two types of pathophysiological phenomena can cause central sleep apnea syndromes:
- ventilatory instability (either decreased or increased ventilator drive), or
- depression of the brainstem respiratory centers or chemoreceptors.
CSA patients fall into 2 groups based onPaCO2 level and ventilatory drive.
Group 1: presents with hypercapnia (abnormally elevated PaCO2 levels) with decreased ventilatory drive. Known causes include hypothyroidism and central lesions (e.g. brainstem infarctions, encephalitis).
Group 2: presents with eucapnia (a state in which the PaCO2 is optimal) or hypocapnia (less than the normal level of PaCO2) with increased ventilatory drive, but with episodes of apnea, periodic breathing, or both. This may be secondary to an existing medical condition such as stroke or heart failure, or due to the use of opioids, or resulting from a recent ascent to high altitudes.
While unstable ventilatory control during sleep is the hallmark of CSA, the pathophysiology and the prevalence of the various forms of CSA vary greatly.4Recent advances in scientific research have shown that the mechanisms responsible for CSA and OSA overlap, and patients with central apneas often have obstructive events, with the pharynx narrowing considerably during a central apneic event.10,11 The significance of this is that CSA can be mistaken for OSA and, in fact, is often treated as such.
Mixed vs Complex Sleep Apnea
Central and obstructive sleep apneas can occur together in one patient. When this happens spontaneously, prior to positive airway pressure therapy, it is termed mixed SA. When it happens subsequent to positive airway pressure therapy, it is termed complex SA. In the first instance, the patient presents with symptoms of both types of sleep apnea. In the second, the patient develops CSA or a Cheyne-Stokes breathing pattern while/after being treated for OSA using positive airway pressure therapy. In other words, a worse condition has been induced iatrogenically.
In 2006, Mayo Clinic researchers described a phenomenon12 in which a significant number of patients diagnosed with severe OSA(15 percent of 223 subjects) failed to breathe normally after undergoing positive airway pressure therapy. Instead, their sleep apnea assumed the characteristics of CSA. In other words, the therapy had induced CSA in OSA patients, culminating in iatrogenic complex SA. In homeopathic terms, the clinicians have suppressed their patients’ symptoms, resulting in a worsening of the patients’ condition.
As far back as 1991, researchers had published an article titled Occurrence of breathing disorders during CPAP administration in obstructive sleep apnoea (Homeopathy Treatment for Sleep Apnoea) syndrome13 in The European Respiratory Journal, in which they had noted an alarming trend:
We analysed the breathing pattern during sleep in forty patients while CPAP was administered. A regular breathing pattern throughout the sleep study was observed in 15 patients. In the remaining 25 subjects, one or more of the following events was observed: central apnoeas, hypopnoeas, periodic breathing, prolonged oxyhaemoglobin desaturations.
(Marrone et al., 1991)
Despite this and similar research, researchers keep coming up with ever more complicated methods to “treat” iatrogenic complex SA, often advocating the addition of drugs to CPAP use or alternative ventilatory assist modalities14,15. For instance, as recently as 2014, researchers Khan and Franco acknowledge the iatrogenic nature of complex sleep apnea in their article, Complex sleep apnea syndrome16, but then go on to advocate more complex “treatment” possibilities:
Complex sleep apnea syndrome is the diagnostic term for the form of central sleep apnea that persists or develops upon treatment of primarily obstructive sleep apnea with CPAP. The pathogenesis likely is related to a combination of the impact of CPAP therapy on ventilation, disturbed ventilatory control related to sleep and host response, and other medical comorbidities.
For those who are poor candidates for CPAP therapy and those who are CPAP therapy failures, more advanced respiratory assist devices including bilevel PAP-ST or adaptive servoventilation therapy can be effective. Other therapeutic medications such as acetazolamide or theophylline may offer an alternative when positive pressure devices of any type are ineffective or poorly tolerated.
(Khan and Franco, 2014)
In a similar vein, patients with upper airway resistance syndrome17(a condition in which patients snore, wake frequently during the night, and have excessive daytime sleepiness) have no breathing abnormalities that characterize sleep apnea and do not show a reduction in PaO2 levels. However, these patients are treated by conventional medicine as if they had sleep apnea, with CPAP devices. While this could result in “significant improvements in the excessive daytime sleepiness”, this is a typical short-sighted allopathic intervention that can have serious consequences on the patient’s overall health, conceivably giving rise to iatrogenic sleep apnea.
Incidence& Prevalence of OSA
- OSA affectsabout 4% of menand 2% of women18, with the maximum prevalence in middle-aged men (45 to 64 years of age)19.
- Sleep-disordered breathing is more severe in postmenopausal than in premenopausal women20.
- OSA affects approximately 3% of children, with snoring being the most common presenting complaint, adenotonsillar hypertrophy being the most common cause, with obesity, hypotonic neuromuscular diseases, and craniofacial anomalies being other major risk factors21.
- Obese children are 4-6 times more likely to have OSA, compared with lean subjects.22
- The concurrent presence of adenotonsillar hypertrophy and obesity appears to facilitate the emergence of OSA in children.23
- The prevalence of sleep apnea in non-obese and otherwise healthy children younger than 8 years old is estimated as 2-5%. OSA may develop in children of all ages, even in infants. Prematurely born infants are at a higher risk to develop sleep apnea.
- SDB (sleep-disordered breathing) was 4 to 6 times more likely in black children compared with white children and almost 3 to 5 times more likely in former preterm compared with term children. The estimated population prevalence of SDB was 2.2%.24
- SDB is a relatively common condition in 8- to 11-year-old children. Potentially vulnerable subgroups, black children, and former preterm infants, are at increased risk.24
- Children with tonsillar hypertrophy had a 3.7 times higher risk of suffering SDB than those with normal size tonsils after adjustment for age, sex and body fat percentage.25
- Abnormal craniofacial morphology (cross bite or convex facial profile), but not excess body fat, is associated with an increased risk of having SDB in 6-8-year-old Finnish children.25
Incidence & Prevalence of CSA
CSA is generally considered to be less prevalent in the general population than OSA.26This is because researchers are mainly looking at one particular type of CSA—secondary CSA—found mostly in elderly males27who have certain co-morbid conditions, especially heart failure. In the next chapter, this idea—that CSA is rare and a disease of elderly men with failing hearts—will be refuted when the causation of a common type of CSAis revealed, showing that even young and otherwise healthy men and women can exhibit both signs and symptoms of CSA.
Predisposing/Risk factors
Predisposing factors for sleep apnea include those that contribute directly or indirectly to airway narrowing (e.g. obesity, craniofacial morphology, enlarged tonsils or adenoids), co-morbidities such as neuromuscular diseases, and habits such as smoking, alcohol and drug use.
The strongest risk factor is central obesity, reflected by BMI (best body mass index BMI calculator online), neck circumference (>17 inches for men, >15 inches for women), and waist-hip ratio. [Obesity contributes directly to upper airway narrowing by increased volume of pharyngeal fat deposits, under the mandible and within the tongue, soft palate, or uvula while excess fat-free muscular tissue increases the size of many upper airway structures, compressing lateral airway walls. Obesity contributes indirectly to upper airway narrowing, esp. during sleep, as lung volumes are markedly reduced by a combination of increased abdominal fat mass and recumbent posture.]
- Obesity (more significant in adults than in children).
- Aging (up to age 65).
- Male gender.
- Menopause20.
- (Increased risk in African Americans appears to be independent of the effects of obesity or respiratory problems.28)
- Abnormal craniofacial morphology (e.g. receding chin or mandible, or narrow or receding maxilla).
- Large tonsils or adenoids (especially in children).
- Upper and lower respiratory tract problems in children and adolescents.28
- Allergies29 in children (Allergy is frequently present in pediatric patients with habitual snoring; presence of allergy is associated with an increased risk of OSA in this population.)
- Neuromuscular disorders that affect the muscles in the airways.
- Smoking30 (Cigarette smoking causes increased upper airway inflammation; even exposure to second-hand smoke is associated with habitual snoring).
- Alcohol use30 (Ingestion of alcohol before sleep has been shown to increase upper airway collapsibility and to precipitate obstructive apneas and hypopneas during sleep.)
- Drug use, esp. sedatives, sleeping pills, opioids, or heart medications.
- Heart failure and stroke.
- Family history of sleep apnea. (Genetic factors associated with craniofacial structure, body fat distribution and neural control of the upper airway muscles interact to produce the OSA phenotype.31)
Significance
People with untreated sleep apnea stop breathing repeatedly during sleep, sometimes hundreds of times during the night and often for a minute or longer each time. In most cases the sleeper is unaware of these breath stoppages because they don’t trigger a full awakening, but with each apnea event, the brain rouses the sleeper, albeit only partially, to signal breathing to resume. As a result, the patient’s sleep is extremely fragmented and of poor quality, never dropping into the deep stage of sleep (stage 4) that is required for detoxification of major organs and healing purposes. Patients with disrupted sleep stay within the shallower stages of sleep, with major repercussions on their physical, mental, and emotional well-being.
Complications
It is believed that many who have OSA do not seek treatment or even diagnostic confirmation of their symptoms. Reasons include lack of insurance, not wishing to increase insurance premiums, not wishing to use an oral appliance or CPAP machine or be told that they need surgery. Left untreated, OSA may predispose to serious health conditions, including hypertension and consequent cardiovascular morbidity32. It may also be associated with insulin resistance33, late-onset epilepsy34, cognitive impairment35 (e.g. difficulty in concentrating, increased forgetfulness, inability to make decisions), and peri-operative complications36. Sleep disorders in general have been associated with motor vehicle accidents36, workplace accidents, impaired cognition (reduced ability or inability to concentrate, think, or remember), mood changes, and decreased quality of life.
Effects in Infants and Children
Consequences of OSA in children include failure to thrive (young children failing to gain weight or grow at a normal rate; parents of infants with significant OSA may report difficulty with feeding37), enuresis, behavioural issues such as attention-deficit disorder and hyperactivity, poor academic performance, and cardiopulmonary disease.38 In fact, children with undiagnosed sleep apnea may be misdiagnosed with attention-deficit and hyperactivity39 disorder (ADHD).
Confounding Factors
While some articles have linked OSA with increased risk of cardiovascular events and of stroke, some researchers, e.g. Hermann and Bassetti40, urge caution and further research due to confounding factors such as hypertension, hyperlipidemia, diabetes mellitus, and smoking:
Despite considerable progress regarding our understanding about the role of SDB in vascular disease, further studies are needed to determine whether cerebrovascular morbidity is related to SDB independent of associated vascular risk factors, whether SDB has indeed a negative impact on stroke progression and whether CPAP treatment ameliorates stroke outcome.
(Hermann and Bassetti, 2003)
Ethical Considerations
While many researchers recommend weight loss as first line treatment of OSA in the obese patient, researchers sponsored by manufacturers of CPAP machines(e.g. Respironics and Resmed) and those who hold patents for similar equipment are the ones who, perhaps unsurprisingly, come up with rather dire conclusions41 and highly recommend CPAP usage:
Sleep-disordered breathing is associated with all-cause mortality and specifically that due to coronary artery disease, particularly in men aged 40–70 with severe sleep-disordered breathing.
(Punjabi et al., 2003)
Conventional Diagnosis
Diagnosis rests on the basis of history and clinical findings, confirmed by polysomnography, which is completed via overnight observation in a sleep laboratory. Testing may not be done if CSA causes no symptoms or is clearly related to an identifiable disorder (e.g. heart failure, chronic opioid use). To diagnose causes of CSA, brain or brain stem imaging may be indicated.
Conventional Understanding
Conventional medicine considers all types of sleep apnea to be incurable, so treatment is “supportive”. In homeopathic terms, it is palliative as opposed to being curative. Patients with moderate to severe sleep apnea are often traumatized by the dire prognosis given to them by sleep doctors—that they will “get a stroke in 5 years without a CPAP machine”, and that it is their only option for the rest of their lives.
Conventional Treatment for OSA& CSA in Adults
Conventional treatment in OSA includes non-invasive therapies as well as surgery. Where obesity is an issue, it should be, but often is not, addressed. Cases of mild to moderate OSA are sometimes treated using oral appliances. These arecustom-made by dentists who are trained, often in one short course, in “dental sleep medicine”, and are intended to improve breathing by holding the tongue in place or by forward positioning of the lower jaw using a mandibular advancement device to, theoretically, improve the patency of the upper airway during sleep.
Recent research by Park and colleagues42 (2016) has shown that the mandibular advancement device (MAD) has been successful in the treatment of OSA, but only in a subset of patients:
The OSA patients who had lower body mass index and upper airway narrowing at the level of palate and tongue base showed relatively higher rates of a satisfactory outcome even in cases of moderate or severe OSA. These results suggest that the use of a MAD may be an alternative treatment option in OSA patients with retropalatal and retroglossal area narrowing regardless of disease severity. Additionally, MADs can be recommended as an initial treatment modality, and the effectiveness of MADs in achieving success may not be inferior to CPAP.
(Park et al., 2016)
Patient compliance with oral appliances is not high, as might be expected, and tooth movement such as changes in overbite and overjet, as well as TMJ pain and dysfunction, can result.
Almost all cases of moderate to severe OSA are told that a CPAP (Continuous Positive Air Pressure) machine is their only option for life, despite the fact that both CPAP and BiPAP devices have been shown to cause CSA in OSA patients.43
In secondary CSA, first line of treatment is management of underlying conditions and avoidance of opioids and other sedatives if relevant. Second line of treatment is CPAP or ASV and oxygen or, if none of these work, BiPAP in cases if CHF, or drugs if PAP therapy is not tolerated. Most concerning is the treatment outlined for idiopathic primary CSA: not just PAP therapy but dangerous drugs that have “limited supportive evidence”44.
Surgery for adultsshould be considered only as a last resort for patients who cannot tolerate CPAP use, as it is fraught with complications, especially in the case of soft tissue surgery such as uvulopalatopharyngoplasty (UPPP), the most widely performed surgery in adultswith OSA, despite research by Boyd and colleagues who concluded in their article, Comparative Effectiveness of Maxillomandibular Advancement and Uvulopalatopharyngoplasty for the Treatment of Moderate to Severe Obstructive Sleep Apnea45, that:
MMA (maxillomandibular advancement) should be the surgical treatment option of choice for most patients with moderate to severe OSA who are unable to adequately adhere to CPAP.
(Boyd et al., 2013)
The extremely poor prognosis of UPPP is often attributed to the patient gaining weight and “the natural aging of the body and increase in tissue collapsibility”46.
Compliance with CPAP devices is poor and other conventional treatment options often ineffective, as pointed out by Dempsey and colleagues in the article Physiology in Medicine: Obstructive sleep apnea pathogenesis and treatment—considerations beyond airway anatomy.47
Given that several non-anatomical determinants of cyclical airway obstruction are prevalent in many patients with OSA, that appropriate CPAP therapy is not acceptable to about one-half of patients diagnosed with OSA, and that oral appliances and surgical therapies are not always effective, alternative therapies or combination of therapies are needed, preferably tailored to specific risk factors of a patient.
(Dempsey et al., 2014)
The American Academy of Sleep Medicine updated their clinical guidelines48 in 2015 with statements that appear excessive, e.g.: “We recommend sleep physicians prescribe oral appliances, rather than no therapy, for adult patients who request treatment of primary snoring (without obstructive sleep apnea)”, especially when we consider that a recent (2015) well-designed Swedish study49 by Marklund and colleagues has concluded that while a custom-made, adjustable oral appliance may reduce OSA and snoring, it does so without any positive effect on daytime sleepiness, headaches, insomnia, or quality of life.
However, while less effective than CPAP, oral appliances are more effective than surgery such as UPPP, as noted by Hoffstein in his 2007 meta-analysis, Review of oral appliances for treatment of sleep-disordered breathing.50
ConventionalTreatment for OSA in Children
In a recent (2016) article, Non-surgical management of obstructive sleep apnoea: a review51, published in the journal Pediatrics and International Child Health, researchers examined recent literature on non-surgical management of paediatric obstructive sleep apnoea (OSA) and concluded that interventions all have their limitations.
In mild cases of OSA, intra-nasal steroids and other anti-inflammatory medications may give relief, but the long-term safety of these treatments has not been established. Weight loss in obese children has been shown to be effective in selected patients but is limited in practice. Non-invasive ventilation may be effective but compliance can be a major obstacle. Oral appliances are effective by stenting the pharyngeal airway, but research in this area is limited.
CONCLUSION:There are number of potential, if not proven, alternative management strategies for children with OSA, which could be considered in the absence of early surgical intervention.
(Whitla and Lennon, 2016)
Children with swollen tonsils and/or adenoids often have these surgically removed as a first line of treatment52 even though this does not work for every child .In their well-researched article, Pediatric Obstructive Sleep Apnea. Complications, Management, and Long-term Outcomes39, Capdevila and fellow researchers note that surgery do not always improve outcome in certain groups of children:
The recommended initial treatment, even in obese children, consists of surgical removal of the adenoids and tonsils. However, not all children who undergo adenotonsillectomy (T&A) for OSA are cured. In a meta-analysis of the published literature, the success rate for T&A in the context of OSA was approximately 85%. This figure may actually be lower, particularly among obese children with OSA, or among children with severe OSA
(Capdevila et al., 2008)
In the article, Risk factors and treatment for obstructive sleep apnea amongst obese children and adults53, researcher Kohler points out that obesity as a predisposing factor of OSA is less significant in children than in adults, and that conventional treatment (including surgical removal of tonsils and adenoids) may not give rise to optimal outcome unless the obesity is addressed.
In view of recent findings, a direct association between body mass and upper airway obstruction should be viewed with caution. Obesity may play a more significant role in the predisposition to OSA amongst particular subgroups of the population, such as adults, and those with particular craniofacial and upper airway morphology…Further, commonly used treatment methods for OSA (such as adenotonsillectomy for children and continuous positive airway pressure for adults) may be less effective for obese individuals. Weight-reduction strategies appear important for an optimal outcome…
(Kohler, 2009)
SUMMARY of Conventional Treatment
| NON-INVASIVE | INVASIVE/SURGERY |
| § Obesity management
§ Oral appliances: o in adults, e.g. MAD o in children, rapid maxillary expansion (orthodontic procedure to widen palate); functional appliances § Drugs/Medications § Ventilators (e.g. CPAP, BiPAP, APAP, ASV) |
§ Adenotonsillectomy in children § Orthognathic/craniofacial surgery, e.g. MMA § Nasal surgery (e.g. deviated septum, turbinates, sinus disease) § Surgery of nasopharynx, e.g. UPPP § Tongue surgery § Bariatric surgery (weight loss) § Tracheostomy (in COPD, CHF) |
- APAP: adjustable positive airway pressure
- ASV: adaptive servo-ventilation
- BiPAP: bi-level positive airway pressure
- CHF: congestive heart failure
- COPD: chronic obstructive pulmonary disease
- CPAP: continuous positive airway pressure
- MAD: mandibular advancement device
- MMA: maxillomandibular advancement
- UPPP: uvulopalatopharyngoplast
Alternative/Complementary/Preventative Treatment of OSA in Children
While obesity as a risk factor for sleep apnea is one issue that must be addressed, abnormal cranial morphology is an even more significant risk factor that needs to be understood. Narrow maxillae and receding mandibles are not inevitable genetic expressions but are amenable to epigenetic influences. Dr. Weston Price’s pioneering work in the 1930s shows how a poor diet can affect the size and shape of the oral cavity, which not only results in crowded teeth but affects tongue placement that can lead to poor breathing habits such as mouth-breathing as well as dysfunctional swallowing patterns. Poor nutrition and lack of breastfeeding over several generations can lead to sub-optimal orofacial growth, as breastfeeding guides and promotes the development of an infant’s palate, helping create adequate width and patency of the upper airway. Children with a diet high in processed foods simply do not chew enough, and masticatory muscles that pull on muscle attachments become under-utilized, resulting in sub-optimal jaw development.
Also, “tongue-tie” or a short lingual frenulum54 can predispose to abnormal tongue function which in turn can impact orofacial growth that can lead to disordered breathing during sleep. Dentists, pediatricians and otolaryngologists should assess the lingual frenulum in a child with difficulties such as mouth-breathing, snoring, tooth grinding and clenching, speech impediments, and feeding problems. While frenulum removal can help, it may not resolve all abnormal breathing patterns, in which case orofacial myofunctional therapy, both pre- and post-surgery, is recommended to restore normal breathing through the nose. Alternatively, the Buteyko Methodcan help both children and adults learn how to breathe properly through the nose. This method has helped both asthma and sleep apnea patients of all ages.
CHAPTER 2: CAUSATION IN CSA
Freeze Response
Coined by Cannon in the ’20s, the phrase “fight or flight” in the context of human response to perceived threat has been influential in subsequent work on anxiety and anxiety disorders. In their article Exploring Human Freeze Responses to a Threat Stressor55, Schmidt and fellow researchers refer to Barlow’s treatise, Anxiety and Its Disorders: The Nature and Treatment of Anxiety and Panic,and elaborate on the “freeze response”.
Part of Barlow’s (2002) description of an adaptive alarm model suggests that a freeze response may occur in some threatening situations. Specifically, freezing — or tonic immobility — may overwhelm other competing action tendencies. For example, when fleeing or aggressive responses are likely to be ineffective, a freeze response may take place. Similar to the flight/fight response, a freeze response is believed to have adaptive value. In the context of predatory attack, some animals will freeze or “play dead.” This response, often referred to as tonic immobility (Gallup, 1977), includes motor and vocal inhibition with an abrupt initiation and cessation.
Freezing in the context of an attack seems counterintuitive. However, tonic immobility may be the best option when the animal perceives little immediate chance of escaping or winning a fight (Arduino & Gould, 1984; Korte, Koolhaas, Wingfield, & McEwen, 2005). For example, tonic immobility may be useful when additional attacks are provoked by movement or when immobility may increase the chance of escaping, such as when a predator believes its prey to be dead and releases it.
(Schmidt et al., 2008)
Not only mammalian wildlife have been observed to exhibit the freeze response. Human beings undergoing extraordinary stressful situations, in which neither “fight” nor “flight” are possibilities, are known to exhibit the “freeze” response.
Despite evidence suggesting that tonic immobility may be a key facet of alarm reactions, freezing has received relatively little scientific attention in humans. One exception is the PTSD/rape literature wherein several studies have described a rape-induced paralysis that appears to share many of the features of tonic immobility. This literature suggests that a relatively high percentage of rape victims feel paralyzed and unable to act despite no loss of consciousness during the assault (Burgess & Holmstrom, 1976; Heidt, Marx, & Forsyth, 2005). Since fear, predation, contact, and restraint are common to both rape and the induction of tonic immobility in animals, it has been concluded that these phenomena are essentially isomorphic (Suarez & Gallup, 1979).
(Schmidt et al., 2008)
If a freeze response is not discharged (as when a wild animal shudders back to life after playing dead when the predator has left), and it often is not in human beings who have been conditioned not to show their emotions even under extreme duress, then PTSD can and often does result. This conditioned suppression of instinctive emotional (e.g. showing feelings in the form of weeping) and physical response (e.g. shaking, shivering, shuddering) is culturally encouraged in males more than in females, which may explain the higher proportion of men diagnosed with PTSD.
POST-TRAUMATIC STRESS
Several sleep disorders—nightmares, insomnia, sleep apnoea and periodic limb movements—are highly prevalent in PTSD, and several studies found disturbed sleep to be a risk factor for the subsequent development of PTSD. Moreover, sleep disturbances are a frequent residual complaint after successful PTSD treatment: a finding that applies both to psychological and pharmacological treatment. In contrast, treatment focusing on sleep does alleviate both sleep disturbances and PTSD symptom severity. A growing body of evidence shows that disturbed sleep is more than a secondary symptom of PTSD—it seems to be a core feature.
(Spoormaker and Montgomery, 2008)
The PTSD and Sleep Apnea Connection
Studies have shown a very high incidence of sleep apnea in anxiety disorders, including PTSD. Sleep apnea is known to be a co-morbid condition in PTSD in combat veterans56,57 and in women who have suffered sexual trauma58,59,60. Because researchers have no idea how these disorders trigger sleep apnea, they point to the fact that treating the sleep disorder results in significant improvement in the anxiety disorder as proof that the sleep apnea was pre-existing, just undiagnosed. However, Dr Damaris Drewry’s seminal work on patients with idiopathic CSA, which she has termed “psycho-physiological CSA”, has shown that this supposition is patently false.
In her article, Central Nervous System Apnea Can Be Caused by Traumatizing Events and It Can Be Resolved61, published in the peer-reviewed journal, The International Journal of Healing and Caring, Dr Drewry explains her theory of the connection between psycho-physiological CSA and post-traumatic stress.
The author postulates that the false perception of imminent death during a traumatizing event is interpreted as fact by the subconscious mind, which then gives the directive to create a stop-breathing program in the autonomic nervous system, possibly via the activity of the neuropeptide Orexin. The “stop-breathing program” is a faulty survival mechanism in direct opposition to the body’s innate directive to breathe continuously during sleep, and results from archived traumatic memories that run subconsciously all the time.
The “stop-breathing program” is a survival mechanism in direct opposition to the body’s innate directive to breathe continuously, and results from archived traumatic memories. People in crisis may stop breathing in order to avoid inhaling water, toxins, or amniotic fluid, or to make themselves unobtrusive in order to avoid a threat.
(Drewry, 2017)
Drawing on a body of work that encompasses 90 men and women over 8 years (between 2008 and 2016), Dr Drewry makes a compelling case for the PTSD/CSA connection. Of her 90 sleep apnea subjects, 64 (i.e. 71%) had reported symptoms of anxiety and panic while 33 (i.e. 24%) had been diagnosed with PTSD.
In a traumatic event, the experience, along with meaning given to the experience, is stored in memory. Decisions made during specific types of breathing-related traumatizing events are re-lived as Post-Traumatic Stress (PTS) which sets up a subconscious program that interferes with sleep. PTS continues to run that subconscious stop-breathing program until the brain can be made to realize that those events are no longer happening and are no longer a threat to survival.
(Drewry, 2017)
The PTSD – Limbic System – Breathing Connection
The limbic system is a set of structures located on top of the brainstem. The amygdalae are a pair of limbic system structures involved in many of our emotions and motivations, particularly those related to survival. It is involved in the processing of emotions such as fear, anger, and pleasure. The amygdala is also responsible for determining what memories are stored and it is thought that this determination is based on the intensity of the emotional response evoked. There are neurons in the amygdala that are responsible for fear conditioning, an associative learning process by which we learn through repeated experiences to fear something. The hippocampus is responsible for long-term memory storage and retrieval.
Our experiences can cause changes in brain circuitry, physiology, and even anatomy. For instance, hyperactivity of the amygdalae or having one amygdala that is smaller than the other has been associated with fear and anxiety disorders, including PTSD. Fearful situations can overwhelm normal brain functioning by sensitizing the amygdalae to the point where they over-react to situations that are not life-threatening. This is the essence of PTSD, where the fight/flight/freeze response (an activity mediated by the amygdalae) is activated inappropriately.
The limbic system influences both the sympathetic part of the autonomic peripheral nervous system and the endocrine system, which means that stress hormones are released with the activation of the fight/flight/freeze response. The presence of increased levels of stress hormones and an over-active sympathetic nervous system impairs our immune function and our vital force, making us less able to deal with life’s stresses. This, essentially, is what happens physiologically when severe or prolonged psychological trauma culminates in PTSD.
In Amygdala and Emotional Breathing in Humans62, authors Masaoka and Homma point out that respiratory psychophysiology studies have found that respiratory patterns are affected by fear and anxiety in humans. They go on to make the link between the limbic system and the respiratory centre in the brainstem.
The brainstem regulates respiration to adjust for a metabolic requirement but final respiratory output appears to be from interactions between maintaining homeostasis and input from many types of sensory information and emotions from the higher cortical and limbic structures.
(Masaoka and Homma, 2004)
If PTSD has to do with an over-active amygdala, and breathing patterns are subject to influence from the limbic system, does it then follow that any kind of psychological trauma that results in PTSD can result in breathing disorders? And would the odds increase if the trauma is breathing-related?
Breathing-Related Trauma
It is Dr Drewry’s contention that trauma that results in a freeze response (the tendency towards tonic immobility in the context of threat) may give rise to psycho-physiologial CSA. In her work with people with CSA, Dr Drewry has discovered the following common themes, many of them breathing-related. All have shown up as causation in cases of sleep apnea that have partially or completely resolved following her specific therapy.
- Asthmatic attacks.
- Anxiety or panic attacks.
- Near-drowning experience.
- Waking during general anesthesia.
- Losing consciousness unexpectedly/suddenly/unintentionally, e.g.
- ether mask for tonsillectomy as a child and being frightened/panicky/struggling against mask;
- knocked out in a fight or accident or while playing sport.
- Losing consciousness intentionally, e.g. stopping breathing to experience a “high”
- Birth trauma e.g. “blue baby” (born with umbilical cord wrapped around neck), or born premature and kept in an incubator.
- Intra-uterine trauma e.g. mother smoked63 or drank alcohol or did drugs while pregnant.
In their article, Prenatal exposure to cigarettes in infants with obstructive sleep apneas63, Kahn and fellow researchers noted the correlation between parental smoking and obstructive apnea in infants over 20 years ago, even though they did not comprehend how one led to the other:
CONCLUSION: Prenatal smoking by mothers correlated with an increase in frequency and length of obstructive apneas and a decrease in birth weight of their infants. The infants were under greater risk for obstructive apnea if both parents smoked. Explanations for our results are unknown to us, but these findings may be of interest in the study of infant breathing behavior and epidemiological characteristics of sudden infant death syndrome.
(Kahn et al., 1994)
The following groups of people appear to be most at risk:
- combat veterans,
- road traffic accident survivors,
- victims of bullying and abuse,
- witnesses of violence.
It is easy to understand how combat veterans and road traffic accident survivors would end up with PTSD. As for victims of bullying and abuse, they often develop coping or survival mechanisms such as staying very still and breathing very shallowly in order to avoid the notice of an alcoholic or abusive parent or spouse. It is not difficult to imagine children who witness a parent or sibling being abused becoming frozen in shock and horror.
Approximately 25% of Dr Drewry’s 90 clients had a combination of OSA and CSA, and while they felt emotional relief after therapy, airway obstruction still interfered with their breathing. The conclusion she drew from this was that CSA induced by trauma (and maintained by PTSD) is amenable to psychotherapy but the obstructive component of these patients’ sleep apnea was secondary to other factors (usually food allergies) and thus not amenable to psychotherapy.
For those whose CSA completely resolved following therapy, trauma was the one commonality. Dr Drewry intuitively understood in 2008 that a subconscious “stop-breathing program” was created during her client’s near-drowning incident. She speculated that a neurotransmitter must be involved and sought a scientific basis that would explain the startling efficacy of her therapy from a neurological perspective64,65, and found it in neurologist Dr Ralph Rynes, MD, PhD, FDTI, FCNS, who made the following observations in a personal communication (2013):
“I always teach [4th year Psychiatry Residents] about trauma, and to identify sleep apnea as a potential symptom of trauma. I think as physicians we focus too much on obstructive sleep apnea and are too quick to recommend a sleep specialist who automatically puts them on a CPAP. My personal inclination is that the orexin (or hypocretin) pathway is involved. The hypocretins are involved in the sleep/wake cycle and how glucose regulation is affected during sleep, as well as the GABA and glutamate pathways.”.
The Science behind the Theory: The Orexin – CO2– Sleep Apnea Connection
Recent research has shown that orexin neurons are part of a powerful, multitasking control system of the brain, active in the coordination of vital behaviours such as wakefulness and breathing. In his 2008 article, Orchestrating Brain Activity66, Dr Denis Burdakov explains how, with frequent apneic episodes, carbon dioxide accumulates and brain acidosis results. This pH imbalance releases an electrical ‘brake’ in orexin neurons, allowing them to fire faster. The underlying physiological process that his team discovered is that this ‘brake’ consists of potassium pores in the orexin cell membrane, the pores closing with rising acidity, making the orexin neurons fire rapidly. Awakening and increased breathing result, leading to dissipation of excess CO2 and restoration of pH balance in the brain. This means that orexin neurons provide an excitatory stimulus to key brainstem areas involved in ventilation. This is especially important when breathing needs to be increased to get rid of excess CO2, as in sleep apnea.
Panic attacks, Phobias, PTSD
The pathophysiology of panic attacks is well represented in the scientific literature. One of the most well-studied panic provocation challenges is CO2 inhalation67,68.
It is now clear that patients with a wide variety of anxiety disorders hyperventilate both chronically and acutely. Therefore, we present an explanation of the physiological changes produced by hyperventilation. In order to further study ventilatory physiology in patients with anxiety disorder, our group and others have used the carbon dioxide challenge test. The data from these experiments suggest that patients with panic disorder are hypersensitive to carbon dioxide and that carbon dioxide inhalation induces panic attacks in susceptible patients. Hyperventilation appears to be a secondary, but pathophysiologically important, event in the generation of acute panic.
(Gorman and Uy,1987)
In their 2008 article, Exploring Human Freeze Responses to a Threat Stressor55, Schmidt et al. describe how they used a CO2 challenge to successfully provoke both freeze and flight responses in different subjects. In their article, Acids in the brain: a factor in panic?69, Esquivel et al. muse on the pathophysiology of hyperventilation disrupting brain homeostasis to the point where panic (a fear response that may involve fight/flight/or freeze response depending on circumstances) can arise.In their article, Respiration and autonomic regulation and orexin70, researchers Nattie and Li explain the connection between Orexin and breathing and stress.
Orexin, a small neuropeptide released from neurons in the hypothalamus with widespread projections throughout the central nervous system, has broad biological roles including the modulation of breathing and autonomic function.
Orexin is also essential in physiological responses to stress. Orexin-mediated processes may operate at two levels: (1) in sleep-wake and circadian states and (2) in stress, for example, the defense or “fight-or-flight” response and panic-anxiety syndrome.
Nattie and Li (2012)
In the same year (2012), an article titled Orexin, Stress and Anxiety/Panic states71was published in the same journal (Progress in Brain Research) byJohnson et al.
Orexin’s role in post-traumatic stress disorder (PTSD) and phobias: Orexin’s ability to excite amygdala nuclei (Bisetti et al., 2006) suggests Orexin may regulate fear conditioning which plays a role in phobias and PTSD. The amygdala is strongly linked to conditioned fear (Tye et al., 2011), and pathology associated with PTSD [see review (Mahan and Ressler, 2012)].
(Johnson et al., 2012)
Researchers Ohno and Sakurai in their 2008 article, Orexin neuronal circuitry: role in the regulation of sleep and wakefulness72, make the connection between orexin and vigilance that is needed for survival.
A link between the limbic system and orexin neurons might be important for increasing vigilance during emotional stimuli. Orexin neurons are also regulated by peripheral metabolic cues, including ghrelin, leptin, and glucose, suggesting that they might have important roles as a link between energy homeostasis and vigilance states. Recent research has also implicated orexins in reward systems and the mechanisms of drug addiction. These observations suggest that orexin neurons sense the outer and inner environment of the body, and maintain proper wakefulness of animals for survival.
(Ohno and Sakurai,2008)
We already know that PTSD sufferers are hyper-vigilant during the day. This hyper-vigilance conceivably carries on into the night, even during sleep, disallowing normal slumber by rousing them from one breath to the next through orexin-mediated physiological mechanisms, all orchestrated to ensure that they survive (at least in the short-term).
HEAD TRAUMA
In the introduction to their 2009 article,Treatment of Sleep Disorders after Traumatic Brain Injury73, Castriotta and fellow researchers elaborated on the various causes of traumatic brain injury (TBI) and the types of sleep disorders that can result.
Traumatic brain injury (TBI) has been increasingly recognized as a major health problem in both the civilian and military population as a consequence of motor vehicle accidents and explosive devices. Every year, over 124,000 civilians in the U.S. who sustain a TBI develop a long-term disability. Presently, there are over 3.3 million Americans living with a TBI-related long-term disability. Recent studies have revealed a high prevalence of sleep disorders in the TBI population, including narcolepsy, posttraumatic hypersomnia (PTH), periodic limb movements in sleep (PLMS), and especially obstructive sleep apnea (OSA), with serious consequences.
(Castriotta et al.,2009)
In their 2007 article, Prevalence and consequences of sleep disorders in traumatic brain injury74. Castriotta and fellow researchers concluded that there is a high prevalence of sleep disorders (46%) and of excessive daytime sleepiness in subjects with traumatic brain injury (TBI). The prevalence of OSA in this population was 23%. Back in 2001, Castriotta and Lai had set out to investigate the frequency of sleep disorders in TBI patients with hypersomnia and to discern the relationship between post-traumatic sleep disorders and pre-traumatic sleep symptoms. In their article, Sleep disorders associated with traumatic brain75injury, they described how a diagnosis of treatable sleep disorder was made in all 10 subjects and how hypersomnia can be pre-existing (and contribute to the likelihood of a motor vehicle accident) or post-traumatic.
Sleep-disordered breathing was found in 7 subjects: overt obstructive sleep apnea (OSA) was diagnosed in 5 subjects, rapid eye movement–related OSA in 1, and upper airway resistance syndrome (UARS) in 1. Narcolepsy was diagnosed in 2 subjects, and the diagnosis of posttraumatic hypersomnia was made in 1 subject. Three subjects had symptoms of hypersomnia before their injury (1 each with narcolepsy, OSA, UARS), and 2 of these were driving a car at the time of injury. Conclusion: Treatable sleep disorders appear to be common in the sleepy TBI population, but may be largely undiagnosed and untreated.
(Castriotta and Lai,2001)
In their 2007 article, Sleep disorders in chronic traumatic brain injury76, researchers Verma and colleagues found that the rate of sleep apnea in their study population (60 adult patients with TBIwho presented with sleep-related complaints 3 months to 2 years following TBI, none of whom had sleep complaints prior to the TBI) was significantly higher than population norms.
In their 2012 article, Sleep disturbances among soldiers with combat-related traumatic brain injury77, Collen and fellow researchers report finding obstructive sleep apnea syndrome (OSAS) in 34.5% of 116 soldiers with combat-related TBI, of whom 55.2% had insomnia. Interestingly, the authors observed that those with blast injuries developed more anxiety and insomnia while those who suffered from blunt trauma had significantly more OSAS.
In their 2012 article, Traumatic Brain Injury and Sleep Disorders78, researchers Viola-Saltzman, and Watson acknowledge that both central and obstructive sleep apnea can result from TBI.
Sleep disturbance is common following traumatic brain injury (TBI), affecting 30–70% of individuals, many occurring after mild injuries. Insomnia, fatigue and sleepiness are the most frequent post-TBI sleep complaints with narcolepsy (with or without cataplexy), sleep apnea (obstructive and/or central), periodic limb movement disorder, and parasomnias occurring less commonly. In addition, depression, anxiety and pain are common TBI co-morbidities with substantial influence on sleep quality.
(Viola-Saltzman, andWatson, 2012)
Viola-Saltzman and Watson make a point of including sports-related head injury (concussion) in their discussion, together with other civilian TBI such as falls, motor vehicle accidents, assaults, etc., with such injuries often occurring in the context of construction or industrial accidents, domestic and child abuse, as opposed to military or combat TBI. They explain the mechanisms of TBI in different areas of the brain resulting in different types of sleep disorder.
The type of sleep disturbance resulting from a closed head injury depends on the location of injury within sleep-regulating brain regions. Posttraumatic hypersomnia is seen when areas involving the maintenance of wakefulness are damaged. These regions include the brainstem reticular formation, posterior hypothalamus and the area surrounding the third ventricle. High cervical cord lesions have also been known to cause sleepiness and obstructive sleep apnea (OSA).In addition, whiplash may cause hypersomnia by precipitating sleep-disordered breathing.
Coup-contrecoup brain injury following head trauma occurs most frequently at the base of the skull in areas of bony irregularities (especially the sphenoid ridges), with consequent damage to the inferior frontal and anterior temporal regions, including the basal forebrain (an area involved in sleep initiation). As a result, insomnia is a common symptom following injuries of this mechanism. Closed head injury can involve the suprachiasmatic nucleus and/or its output tracts leading to disturbance of circadian rhythmicity with concomitant hypersomnia and insomnia.
(Viola-Saltzman, and Watson, 2012)
It is not surprising that offensive and defensive linesmen accounted for 85 percent of the cases of sleep-disordered breathing in 52 professional football players(Prevalence of Sleep-Disordered Breathing in Professional Football Players79). After all, as the article points out, linesmen also had the largest neck circumference and highest body-mass index, as well as significantly higher systolic and diastolic blood pressure.
Certainly, high BMI (from upper body muscular hypertrophy and sheer size) and thick muscular necks are known predisposing factors. But being predisposed to sleep apnea is one thing; acquiring sleep apnea is something else altogether. And what of the 15% who do not have the predisposing factor of high BMI? What causes these athletes to go from having no sleep apnea to having it? Common sense tells us that athletes in contact sports take a lot of high velocity hits and spills which can cause concussion, with or without loss of consciousness.
Recent data (2011) from helmet sensors show that running backs and quarterbacks suffer the hardest hits to the head, while linemen and linebackers are hit on the head most often.79Data on head acceleration and hit direction are used to calculate a composite score of exposure that researchers believe might be a good predictor of concussion.80 On average, running backs had the highest score, followed by quarterbacks and then linebackers. Offensive and defensive linemen had the lowest scores, but along with linebackers, they were hit on the head most often.79 Both hit severity and hit frequency need to be taken into account because repeated head impacts may cause sub-concussive neurological damage over time.
In their 2007 article, Cognitive impairment in patients with traumatic brain injury and obstructive sleep apnea81, Wilde and fellow researchers compared19 patients with TBI and OSA with 16 TBI patients without OSA.They found that the TBI patients with OSA performed significantly worse than the non-sleep disordered TBI patients on verbal and visual delayed-recall measures, made more attention lapses, but performed comparably on motor, visual construction, and attention tests. They concluded that OSA is associated with more impairment of sustained attention and memory in TBI patients and that early identification and treatment of OSA may improve cognitive and potentially functional outcomes in these patients.
In his 2013 article, Recognizing Obstructive Sleep Apnea in Patients With Traumatic Brain Injury82, Dr Christopher Lettieri came to the following conclusions:
- TBI is a growing societal concern that is increasingly recognized among athletes, elderly persons, and military personnel.
- TBI is commonly associated with psychiatric conditions (anxiety, depression, PTSD), neuromuscular and neuro-cognitive impairments (chronic pain, cognitive difficulties), and sleep-related disorders (sleep apnea, posttraumatic hypersomnia, periodic limb movement disorder, insomnia, circadian rhythm sleep disturbances).
- Sleep disorders in patients with TBI are often underdiagnosed and undertreated, can complicate TBI and worsen outcomes, increase disability, or impair rehabilitation.
- Patients with TBI should be evaluated for sleep disorders or referred for formal sleep evaluations, because recognizing and treating these conditions may improve outcomes.
- Because of the inherent cognitive limitations in TBI patients as reporters of their symptoms, all TBI patients with suspected sleep disturbances should undergo a comprehensive, objective evaluation.
CHAPTER 3: SIGNS &SYMPTOMS & BEYOND
SYMPTOMS COMMON TO BOTH OSA AND CSA
Night-time symptoms include:
- loud snoring
- pauses in breathing (i.e. apneic episodes) followed by gasping for air or choking or snorting, with/without awakening
- disrupted sleep (waking multiple times a night)
- restless sleep (tossing and turning)
- insomnia
Daytime symptoms (as a result of insufficient sleep at night) include:
- daytime sleepiness
- fatigue due to non restorative sleep
- frequent day time naps
- dry mouth or sore throat on waking (from mouth-breathing during sleep)
- headache on waking
- shallow breathing during the day
- cognitive difficulties (e.g. difficulty concentrating, confusion, poor memory)
- mood and personality changes (e.g. irritability, anxiety, depression)
SIGNS AND SYMPTOMS SPECIFIC TO OSA
- enlarged tonsils and/or adenoids
- allergies
- post-nasal drip
- chronic rhinitis or sinusitis
- craniofacial anomalies, e.g. receding chin or mandible, narrow palate or maxilla
- abnormalities or weakness in the soft tissues and muscles that surround the airway
- increased overbite
- large tongue
- large neck circumference
- obesity
SIGNS AND SYMPTOMS SPECIFIC TO CSA
CSA can occur in non-obese individuals with little or no snoring, and may be asymptomatic, in which case the only signs noticed by observant caretakers or bed partners may include long respiratory pauses, shallow breaths, or restless sleep. Patients with hypercapnic forms may experience symptoms such as daytime somnolence, lethargy, and morning headache, but may experience less sleepiness during the day than people with obstructive sleep apnea. In cases of CSA secondary to diseases, Cheyne-Stokes respiration and/or hyperventilation may be observed.
SIGNS AND SYMPTOMSSPECIFIC TO CHILDREN
The prevalence of pediatric OSA is approximately 3% in children21. Adenotonsillar hypertrophy is the most common cause, and obesity, hypotonic neuromuscular diseases, and craniofacial anomalies are other major risk factors21. Apart from snoring (the most common presenting complaint), children may have symptoms that differ from adults. Compared with adults, fewer children with OSA report excessive daytime sleepiness, with the notable exception of obese children83,84.Clinical presentation varies according to age.
In their article, Obstructive sleep apnea syndrome in children: Epidemiology, pathophysiology, diagnosis and sequelae21, researchers Chang and Chae noted the following symptoms:
Agitated sleep with frequent postural changes, excessive sweating, or abnormal sleep positions such as hyperextension of neck or abnormal prone position may suggest a sleep-disordered breathing. Night terror, sleepwalking, and enuresis are frequently associated, during slow-wave sleep, with sleep-disordered breathing. Excessive daytime sleepiness becomes apparent in older children, whereas hyperactivity or inattention is usually predominant in younger children. Morning headache and poor appetite may also be present.
(Chang & Chae, 2010)
In their well-researched article, Obstructive Sleep Apnea in Children38, Drs. Chan, Edman, and Koltai also noted different symptoms for children of different age groups:
The presenting problem in children with sleep-disordered breathing depends on the child’s age. In children younger than five years, snoring is the most common complaint. Other nighttime symptoms frequently reported by parents include mouth breathing, diaphoresis, paradoxic rib-cage movement, restlessness, frequent awakenings, and witnessed apneic episodes.Children five years and older commonly exhibit enuresis, behavior problems, deficient attention span, and failure to thrive, in addition to snoring. Compared with adults, fewer children with OSA report excessive daytime somnolence, with the notable exception of obese children.
(Chan, Edman & Koltai, 2004)
In the same article, the medical researchers went on to describe other symptoms that are associated with sleep-disordered breathing in children, e.g. failure to thrive and nocturnal enuresis, that may be due to endocrine dysregulation arising from poor quality sleep.
Poor growth and failure to thrive are more common in children with sleep-disordered breathing. Growth velocity increases after adenotonsillectomy. The hypothesized etiology for failure to thrive is increased work of breathing, with subsequent increase in baseline caloric expenditure. Decreased production of growth hormone during fragmented sleep may contribute further to poor growth.
Enuresis associated with OSA often resolves after successful treatment of sleep-disordered breathing. Increased urine production results from hormonal disregulation. These alterations are accompanied by increased levels of catecholamines and frequent arousal that further contribute to enuresis.
(Chan, Edman & Koltai, 2004)
They then go on to note that cognitive and behavioural difficulties in children with OSA result from poor quality sleep and can lead to poor academic performance in their teens if snoring persists, and that successful treatment of OSA results in improved academic performance, suggesting causality.
As far back as 20 years ago, Chervin and fellow researchers85 had already discovered a possible link between snoring and ADD/ADHD:
The association of snoring with inattention and hyperactivity suggests that SRBDs (sleep related breathing disorders) and perhaps other sleep disorders could be a cause of inattention and hyperactivity in some children.
(Chervin et al., 1997)
More recently, research into the connection between ADHD and sleep disorders in children was done in Australia, where ADHD“is the most common mental health disorder in childhood, affecting up to 11% of Australians aged 6 to17 years”. Sleep problems were found by Sung and fellow researchers86to be extremely common, affecting almost three-quarters of all participants.
Up to 50% of parents of children with ADHD report difficulties with their children’s sleep, including difficulties initiating sleep (delayed onset sleep or bedtime resistance) and maintaining sleep (frequent nocturnal awakenings or restlessness).Daytime sleepiness, tiredness on waking, and nightmares are also more common in children with ADHD than controls. Objective studies using polysomnography, an actigraph, and video monitoring have shown that children with ADHD have increased sleep latency, decreased rapid eye movement sleep percentages, and increased nocturnal activity. Other sleep problems identified in children with ADHD include sleep-disordered breathing (≤ 25%) and restless legs syndrome or periodic limb movement disorder (≤ 36%).
(Sung et al., 2008)
New research by Yürümez and Kılıç87came up with an even higher frequency of sleep problems in ADHD boys (84.8%) compared to controls:
In conclusion, these results underscore the importance of screening all children who have a symptom constellation suggestive of ADHD for sleep problems that may either play a causative role or exacerbate the clinical appearance of ADHD in a given child. Correct evaluation and treatment of sleep problems increase the life quality of family and child and also decrease the severity of ADHD symptoms.
(Yürümez & Kılıç, 2016)
In 2007, avery well-designed study by Huang and fellow researchers used polysomnography performed after ADHD diagnosis to show the presence of mild OSA. After otolaryngological evaluation, three treatment options were offered: no treatment, treatment of OSA with adenotonsillectomy, or methylphenidate (MPH), a commonly used ADHD drug. Of the 66 subjects, 14 had no treatment, 25 had adenotonsillectomy, and 27 were treated with MPH.
Both the surgical and MPH groups improved more than the non-treatment group. Symptoms including attention span, impulse control, response time and total ADHD score improved more in the surgical group than the MPH group. The surgical group also had a TOVA ADHD score close to that obtained in normal controls. The researchers concluded in their article, Attention-deficit/hyperactivity disorder with obstructive sleep apnea: a treatment outcome study88:
Recognition and surgical treatment of underlying mild sleep-disordered breathing (SDB) in children with ADHD may prevent unnecessary long-term MPH usage and the potential side effects associated with drug intake.
(Huang et al., 2007)
In 2009, Silvestri and fellow researchers89 sought to confirm specific sleep disturbances in 55 children diagnosed with ADHDby means of nocturnal video-polysomnography (video-PSG), other than the ones already described in ADHD literature, e.g. periodic leg movement disorder (PLMD), restless legs syndrome (RLS) and sleep related breathing disorder (SRBD).
Most children/parents reported disturbed, fragmentary sleep at night; complaints were motor restlessness (50%), sleep walking (47.6%), night terrors (38%), confusional arousals (28.5%), snoring (21.4%), and leg discomfort at night associated with RLS (11.9%). Different sleep disorders seem to address specific ADHD phenotypes and correlate with severity of symptoms as in sleep related movement disorders occurring in Hyperactive/Impulsive and Combined ADHD subtypes. Besides, an abnormality of the arousal process in slow wave sleep with consequent abnormal prevalence of disorders of arousal possibly enhanced by SRBD has also been detected in 52% of our sample.
(Silvestri et al., 2009)
Silvestri and fellow researchers concluded that the use of sleep studies, e.g. video-PSG, should become part of the diagnostic screening for ADHD.
A meta-analysis by Sadeh and fellow researchers90 in 2006 revealed that children with ADHD are more likely than controls to suffer from PLMS (periodic limb movements in sleep).In their article, Periodic limb movement disorder of sleep in children91, Crabtree and fellow researchers concluded that while a relationship between ADHD and PLMD (periodic limb movement disorder) was observed, it is not so much a direct relationship as a connection mediated by sleep fragmentation associated with PLM-induced arousals.
As many as 1 in 2 of patients investigated for OSA has PLMS92.Significant predictors of PLMS were predisposing medical conditions (esp. depression, fibromyalgia, and diabetes mellitus), age (not uncommon in older men and women), medications, obesity, and OSA91. PLMS can occur with and without arousals. In children, existing PLMS can become more common after adenotonsillectomy93.Periodic limb movements in sleep (PLMS) are related to arousal, sympathetic activation, and increases in blood pressure, but it is not clear if they are causative or simply a part of the arousal process.
Recent research (2017) found that alpha and beta brain activity, systolic blood pressure, and heart rate were significantly increased following PLMS94.Beta activity is closely linked to motor behavior and is generally attenuated during active movements. It is the dominant rhythm in patients who are alert or anxious or who have their eyes open.
Tooth grinding during sleep, i.e. sleep bruxism (SB), can occur at any age. In their article, Associations of sleep bruxism with age, sleep apnea, and daytime problematic behaviors in children95, Tachibana and fellow researchers reported the results of a sleep questionnaire answered by guardians of 6023 Japanese children aged 2-12 years. Sleep bruxism was reported in 21% pf the children, with the prevalence highest in the age group of 5-7 years. There were significant correlations with this particular age group and the following features: ‘moves a lot during sleep’, ‘sleeps with mouth open’, and ‘snores loudly’. There was also a significant correlation with obstructive sleep apnea, which had adirect effect on daytime problematic behavior.
A similar large-scale questionnaire survey of 6389 primary schoolchildren in Hong Kong by Lam and fellow researchers96 resulted in the following conclusions: almost 6% of Hong Kong primary schoolchildren suffered from frequent sleep bruxism, with the condition being most prevalent among young boys. It was found to be associated with a variety of medical conditions, including upper respiratory tract infection, comorbid sleep conditions or parasomnias, especially sleep talking and sleep related breathing problems, with neuropsychiatric sequelae such as hyperactivity, irritability and deterioration in their academic performance.
A statistical association was also found between sleep bruxism and obstructive sleep apnea by Ferreira and fellow researchers97 in Brazilian pre-schoolers (3-5 year olds).Drumond and fellow researchers98 found that rhinitis and sinusitis were associated with sleepbruxism. In their article, Sleep bruxism associated with obstructive sleep apnoea syndrome – A pilot study using a new portable device99, Winck and fellow researchers assert that it is clear that sleepbruxismand obstructive sleep apnoea syndrome share common pathophysiologic pathways.
Researchers Oksenberg and Arons point out in their 2002 article100that sleep bruxism rarely occurs in isolation and that, among associated sleep symptoms and disorders,OSA was the highest risk factor for sleep bruxism. In a patient with both severe OSA and sleep bruxism, polysomnographic recordings pre-CPAP and during CPAP therapy were carried out. The former showed multiple events of sounded tooth grinding, most of them appearing as an arousal response at the end of apnea/hypopnea events, all of them plus most breathing abnormalities eliminated in the latter. This suggests that when sleepbruxism is related to apnea/hypopneas, the successful treatment of these breathing abnormalities may eliminate bruxism during sleep.
In their article, Frequency of obstructive sleep apnea syndrome in dental patients with tooth wear101, Durán-Cantolla and fellow researchers found a statistically significant association between AHI (apnea-hypopnea index)severity and tooth wear severity.
They concluded that “tooth wear could be a tool to identify those patients at risk of having OSAS.” In other words, excessive tooth wear as a marker for OSA is something that dental professionals need to be aware of.
In their article, Sleepwalking and night terrors: psychopathological and psychophysiological correlates102, Szelenberger and colleagues point out that sleepwalking and night terrors are considered to be manifestations of the same nosologic continuum, citing predisposing factors such as a genetic tendency for deep sleep and “triggering factors which increase sleep fragmentation, such as stress, environmental or endogenous stimuli, and stimulants”.
In 2003, Guilleminault and fellow researchers evaluated the clinical presentation and polysomnography of prepubertal children with sleep terrors and sleepwalking, and compared them with a control group. In their article, Sleepwalking and sleep terrors in prepubertal children: what triggers them?103, they described their study of 84prepubertal children (5 with sleep terrors, 79 with both sleep terrors and sleepwalking, and 36 normal controls). 51 of the 84 children had a diagnosis of an additional sleep disorder: 49 with sleep-disordered breathing (SDB) and 2 with restless leg syndrome (RLS). Of the 51 children, 45 were treated. The results were clear, prompt, and highly suggestive of a causative link between SDB and parasomnias:
Forty-three of 49 children with SDB were treated with tonsillectomy, adenoidectomy, and/or turbinate revision, and 2 of 2 children with RLS were treated with Pramipexole, a dopamine agonist, at bedtime. Treatment of the precipitating sleep disorder eliminated parasomnias in all 45 children. In all 43 children who received surgery, polysomnography performed 3 to 4 months later indicated the disappearance of SDB. The recordings also showed an absence of confusional arousals. In all surgically treated cases, parents also reported subsequent absence of the parasomnia. The 2 symptomatic children who were treated with Pramipexole had a complete absence of confusional arousals on the follow-up recording and reported no parasomnia since treatment. The periodic limb movement syndrome arousal index (number of EEG arousals associated with periodic limb movement/hour) decreased from 11 and 16 to 0 and 0.2, respectively. Parasomnia persisted in the 6 children who were untreated for SDB. Surgeons had refused to perform surgery on these children because of lack of data on the relationship between parasomnia and SDB-related tonsil and adenoid enlargement.
(Guilleminault et al., 2003)
Several interesting facts came out of this study:
- Apneas are rare in children, so the SDB may be subtle and not always detectable on polysomnograms, which means more sensitive equipment should be used.
- The high incidence of SDB in family members of children with parasomnias suggest that SDB may manifest as parasomnias in children.
- A familial presence of parasomnias was noted.
The fact that sleep terrors, sleepwalking, RLS and SDB all appear to have familial tendencies led Guilleminault and his colleagues to note: “It remains to be investigated whether a genetic factor directly influences sleep terror and sleepwalking or instead influences other disorders that fragment sleep and lead to confusional arousals.”
Putting It All Together
By now, it would seem obvious that snoring, sleep-disordered breathing (subtle or otherwise, but without apneic episodes to distinguish it from sleep apnea), and obstructive sleep apnea all arise from partial airway blockage. As for the various parasomnias, they appear to arise from the direct (inadequate oxygenation) or indirect consequences (disrupted or fragmented sleep) of partial airway blockage. Parasomnias (e.g. PLMS, sleep bruxism, sleep walking or somnambulism, and night terrors) appear to be on the same nosologic continuum. All may arise from thesame partial airway blockage problem but manifest in different ways due to different patient susceptibility. For instance, inadequate oxygenation during sleep leading to elevated PaCO2levels, in an individual specifically hypersensitive to an increase in PaCO2, may manifest as night terrors, while another patient whose susceptibility lies in the neurological domain, may manifest PLMS instead.
It is known thatpanic disorder patients are specifically hypersensitive to an increase in PaCO2104.Almost a quarter century ago, Klein105 had proposed that “spontaneous panics occur when the brain’s suffocation monitor erroneously signals a lack of useful air, thereby maladaptively triggering an evolved suffocation alarm system.” Klein postulated that panic attacks occur “when the suffocation alarm threshold is lowered, or when psychosocial cues to suffocation are particularly salient”. Researchers Taylor and Rachman’s experimental results106 confirmed Klein’s theory’s main assumption that suffocation alarm hypersensitivity is a risk factor for panic disorder. Other researchers propose that psychological factors play an important role.
Whatever the pathophysiology underlying panic attacks, a hypersensitivity to increased PaCO2 appears to be salient. And what are night terrors but nocturnal panic attacks in sleeping children who are at risk of being suffocated? When we factor in the familial tendency of SDB and parasomnias like sleep walking and night terrors, it is not unreasonable to confer an inherited hypersensitivity to PaCO2upon these children. It now seems obvious that parasomnias arise from SDB which in turn arises from an inherited predisposition. From a homeopathic perspective, there is aknown predisposition to breathing disorders. Let us consider the signs and symptoms commonly found in children with SDB or OSA.
Common symptoms of OSA in children
- Habitual, frequent or loud snoring.
- Laboured breathing during sleep (e.g. mouth breathing, gasping, flaring nostrils, heaving chests, hyperextension of neck
- Excessive sweating during sleep (night sweats).
- Restless sleep (changing position frequently).
· Longer total sleep time than normal.
- Morning headaches.
- Excessive daytime sleepiness.
- Poor appetite, feeding problems in infants, failure to thrive, to grow and to gain weight.
- Behavioral problems, such as hyperactivity and inattentiveness (ADD/ADHD).
- Mood problems, e.g. irritability, emotional instability.
- Co-morbidities, e.g. enuresis, sleep bruxism, sleepwalking, sleep talking, night terrors.
As can be seen by the repertorization, the symptoms are well represented by phosphorus, a tubercular remedy, as well as by the tubercular nosode, Tuberculinum. Does this mean that sleep apnea, the obstructive type in children, hails from a tubercular diathesis?
What do the following symptoms have in common?
- An allergic disposition or constitution that leads to chronic allergies.
- Hypertrophy of adenoids and tonsils.
- Night sweats.
- Sleep bruxism*.
All these symptoms,which happen to occur in children with OSA, are known to homeopaths as being tubercular in origin. Together with parasomnias such as sleepwalking, night terrors, they form a constellation of symptoms that may well arise from an underlying tubercular miasm.
[*It should also be noted that tooth grinding in children is known in homeopathy to be associated with worm infestation.]
BEYOND SIGNS & SYMPTOMS
In conventional medicine, diagnosis is based on history (symptoms) and clinical findings (signs), and confirmed using special tests(e.g. polysomnography). In contrast, homeopaths do not assign a diagnosis but rather focus in case-taking on the patient’s symptom picture—the totality of the symptoms. The most characteristic and unusual (strange, rare, and peculiar or SRP) symptoms take precedence over common symptoms of the disease. This must be kept in mind when we are treating patients with sleep apnea because many of the symptoms (e.g. unrefreshed on waking, daytime sleepiness, cognitive difficulties) are likely to be due to poor quality sleep and therefore of limited value in repertorization.
Miasmatic Analysis
There is thread running through sleep apnea and its co-morbidities that needs to be acknowledged. The tubercular miasm confers a weakness upon the respiratory system as well as an allergic disposition. Chronic allergies give rise to rhinitis and sinusitis and hypertrophy of tonsils and adenoids which in turn cause obstruction of the airway leading to snoring, sleep-disordered breathing or, in severe cases, apneic episodes (i.e. sleep apnea).
In susceptible individuals, elevated PaCO2 levels, via the feedback loop in the respiratory centres in the brainstem, rouse the patient to engage in strange night time behaviour such as sleep bruxism (perhaps to advance the mandible in order to open up the airway106), somnambulism (possibly in a bid to get the patient from a hard-to-breathe supine position into an upright position), or night terrors (elevatedPaCO2 inducing a panic response in a susceptible individual).
Daytime behaviour could either be sleepiness (more often in the obese child) or ADD/ADHD(in the tubercular child) which reflects fragmented sleep (frequent arousals) and altered REM sleep (a shallow sleep stage) and an increased number of shortened sleep cycles. More short sleep cycles translate into reduced total amount of deep Stage 4 sleep which is required for healing and growth in children. Decreased amount of deep sleep can also lead to hormonal dysregulation that can result in failure to thrive or even nocturnal enuresis38.
Conclusion:In children whose sleep apnea is obstructive and due to tonsillar/adenoid hypertrophy, there may be an underlying tubercular constitution that needs to be addressed. Theoretically, the tubercular diathesismay give rise to a constellation of specific symptoms which, if occurring together in a child with a family history of tuberculosis, may serve to help the homeopath differentiate among indicated remedies, as well as consider the nosode Tuberculinum, especially if the symptoms match, or as an intercurrent when appropriate.
The following flow-chart illustrates the author’s proposed mechanism for a constellation of symptoms arising from a tubercular diathesis:
|
|
Causation
In the case of CSA or mixed SA, if the well-indicated remedy fails to work, we may need to take causation into account. In other words, we may need to go beyond the signs and symptoms in order to treat the underlying cause or NWS (Never Well Since) factor that may, in fact, be the obstacle to cure.A good, thorough, history-taking that elicits causation is imperative. In the case of head trauma or breathing-related trauma, the patient is often happy to disclose relevant details when prompted to do so. In cases of pre-natal, intra-uterine, or early childhood trauma, they may be able to get the relevant information from parents or older siblings. In the case of abuse, sensitivity on the part of the clinician is, of course, of the utmost importance.
In the case of CSA secondary to serious diseases such as heart failure, Cheyne-Stokes respiration (a repertorizable symptom) may be present. Reported initially in patients with heart failure or stroke, Cheyne-Stokes respiration was then recognized both in other diseases and as a component of the sleep apnea syndrome.26 The prevalence of Cheyne-Stokes respiration in patients with congestive heart failure ranges from 30% to 50%.
Cheyne-Stokes respiration (CSR)
an abnormalpattern of respiration,characterized by alternatingperiods of apneaanddeep,rapidbreathing.Therespiratorycyclebeginswithslow,shallowbreathsthatgraduallybecomeabnormallyrapidanddeep.Breathinggraduallybecomesslowerandshallowerand is followed by 10 to 20 seconds of apneabeforethecycle is repeated.Eachepisodemaylastfrom 45 seconds to 3 minutes.UnderlyingCSR is a complex alteration in the functioning of the respiratory center in the brain,caused by dysfunction of thediencephalon or by bilateral hemispheric lesions.Therespiratorycentermayhave a reducedsensitivity to the concentrations of blood gases, as is seen in cerebrovasculardisease, in tumors of the brainstem,and in severe head injury.CSRmay be triggered by changes in blood chemical processes,especially in elderly patientswithdegenerative arterial disease or respiratory diseases,such as bronchopneumonia. In an otherwisehealthyperson,CSRmay be caused by hyperventilation,exposure to highaltitudes, or an overdose of a narcotic or hypnoticdrug.CSRoccursmorefrequentlyduringsleep.Mosby’s Medical Dictionary, 9th edition. © 2009, Elsevier.
Cardiac asthma
In the materia medicas, cardiac asthma is the term used to describe the wheezing, coughing or shortness of breath due to congestive heart failure. It is known as cardiac asthma because the symptoms mimic ordinary bronchial asthma. When remedies are described as having cardiac asthma, the point of weakness or origin of disease is in the heart and not the respiratory system. This is of significance when we differentiate between indicated remedies.
CHAPTER 4: RUBRICS & REPERTORIZATION
RUBRICS for Symptoms of Sleep Apnea
Rubrics for causation will be considered after more general sleep rubrics suitable for sleep apnea patients. These are organized according to Mentals, Generals, and Particulars.
Obviously, each case will be different and each homeopathic practitioner will have to choose the most characteristic signs and symptoms of their patient, find the rubrics that most closely and accurately match these, and repertorize accordingly.
MENTALS
- MIND – DELUSIONS – suffocation; as if – sleep; on going to
- MIND – FEAR – suffocation, of
- MIND – FEAR- suffocation, of – closing eyes
- MIND – FEAR- suffocation, of – lying, while
- MIND – FEAR- suffocation, of – night
- MIND – FEAR- suffocation, of – sleep, during
- MIND – STARTING – sleep – from – suffocative breathing, from
GENERALS
- GENERALS- CHEYNE-STOKES respiration
- GENERALS- OBESITY – accompanied by – respiration – difficult
- GENERALS- OBESITY – accompanied by – respiration – wheezing
PARTICULARS
- RESPIRATION- ARRESTED
- RESPIRATION- ASPHYXIA
- RESPIRATION- GASPING -sleep; during
- RESPIRATION-HYPERVENTILATION
- RESPIRATION- IMPEDED, obstructed
- RESPIRATION- INTERMITTENT, unequal
- RESPIRATION- INTERRUPTED
- RESPIRATION- INTERRUPTED – expiration
- RESPIRATION- INTERRUPTED – inspiration
- RESPIRATION- IRREGULAR
- RESPIRATION- DIFFICULT- evening – bed, in
- RESPIRATION- DIFFICULT- night, during
- RESPIRATION- DIFFICULT- night, during – bed, in
- RESPIRATION- DIFFICULT- midnight – after
- RESPIRATION- DIFFICULT- sleep – falling asleep; when
- RESPIRATION- DIFFICULT- sleep – awakened to avoid suffocation; must be
- RESPIRATION- SNORING
- RESPIRATION- STERTOROUS – puffing expiration; with
- RESPIRATION- STERTOROUS – sleep agg.; during
- RESPIRATION- STRIDULOUS
- RESPIRATION- STRIDULOUS – evening, sleep; when falling asleep
- RESPIRATION- SUFFOCATION; attacks of
- SLEEP – DISTURBED – suffocation, by
- SLEEP – FALLING ASLEEP – suffocation, with
- SLEEP – WAKING – suffocation, from
- SLEEP – WAKING – suffocation, from – sleep; on going to
- LARYNX AND TRACHEA – CONSTRICTION – Larynx – sleep – falling asleep – when
- LARYNX AND TRACHEA- CONSTRICTION – Larynx- sleep- falling asleep – when – lying on either side
- THROAT – CHOKING- sleep – going to sleep; on – agg.
- THROAT – SUFFOCATIVE – sensation – night
- CHEST – OPPRESSION – sleep – during
- CHEST – OPPRESSION – sleep – falling asleep – when
REPERTORIZATION
RUBRICS for CAUSATION
Obviously, the rubrics below may not be suitable for any one case and are not exhaustive, being gleaned from only one (Schroyen’s) repertory.
- Head Trauma
If we were to take the causation of head trauma into consideration, then one or more of the following rubrics may need to be considered according to the details of the case:
- GENERALS – INJURIES – concussion
- HEAD – CONCUSSION of brain
- HEAD – CONCUSSION of brain – ailments after
- HEAD – INJURIES of the head; after
- HEAD – INJURIES of the head; after – children; in – delivery; from
REMEDIES FOR HEAD TRAUMA
Using Radar Opus software and Schroyen’s Synthesis repertory, the following remedies come up high in the reperterization:
Arnica, Cicuta, Glonoine, Helleborus, Hyoscyamus, Hypericum, Natrum sulphuricum
When all the head trauma rubrics are combined, 80 remedies are represented, the top 30 being:
Arnica, Badiaga, Cicuta, Hypericum, Natrum sulphuricum, Aconite, Anacardium, Aurum, Belladonna, Calcarea carbonica, Cocculus, Filix mas, Glonoine, Helleborus, Hyoscyamus, Iodum, Kali phos, Lachesis, Ledum, Lobelia, Manganum, Natrum muriaticum, Nux vomica, Opium, Pulsatilla, Rhus tox, Sepia, Silica, Spigelia, Teucrium, Veratrum album
[NB: Lachesis and Opium are represented in the head trauma rubrics, just as they are in the rubrics for sleep apnea symptoms.]
- Mental/Emotional Trauma
If the causation of mental/emotionaltrauma is taken into consideration, one or more of the following rubrics or similar ones may need to be considered according to the details of the case:
- MIND – AILMENTS FROM – abused; after being
- MIND – AILMENTS FROM – abused; after being – children
- MIND – AILMENTS FROM – abused; after being – marriage; in
- MIND – AILMENTS FROM – abused; after being – sexually
- MIND – AILMENTS FROM – abused; after being – sexually – children; in
- MIND – AILMENTS FROM -fear
- MIND – AILMENTS FROM – fright
- MIND – AILMENTS FROM -fright – long-lasting
- MIND – AILMENTS FROM -fright – old fright
- MIND – AILMENTS FROM -fright – ordeal, of an
- MIND – AILMENTS FROM -fright – shock, in
- MIND – AILMENTS FROM -fright – severe
- MIND – AILMENTS FROM -mental shock; from
As can be seen from the repertorization, most of the rubrics are too small to be useful, but thisis merely an exercise to see which remedies are represented. Interestingly, Opium, which showed up for sleep apnea in general, and for head trauma, is also showing up for emotional trauma.
CHAPTER 5: REMEDIES & DIFFERENTIAL DIAGNOSIS
Of the polycrests, Opium and Lachesis come up strongest, while among the small remedies, Grindelia comes up the strongest.
Opium 18/44, Lachesis 16/37; Spongia 14/23, Ant-t 11/21, Am-c 8/11, Carb-v 9/14, Graph 10/17, Grind 7/16; Samb 10/21, Sulph 12/22, Ars 12/24, Dig (11/17), Ign (11/18), Nux vom (12/20), Phos (13/23). Bell (13/20), Chin (10/15), Laur (10/13), Camph (9/17), Val (8/9)
In Clarke’s materia medica under Grindelia, he gives the following rubric:
[Dyspnoea on falling asleep: Am. c., Ant. t., Arum t., Badiag., Bry., Cadm. s., Carb. an., Carb. v., Graph., Lach., Nux m., Op., Ran. b., and both the Grindelias (i.e. Grindelia robusta and squarrosa).]
Taking Clarke’s rubric into account, as well as the remedies that come up in the repertorization of sleep apnea symptoms, and a close reading of the various materia medicas, the following remedies were chosen as representative of sleep apnea remedies:
Ammonium carb, Antimonium tart, Arsenicum,Badiaga, Cadmium sulphuratum, CamphoraCarbo animalis, Carbo vegetabilis, Cenchris, Graphites, Grindelia, Lachesis, Laurocerasus, Opium, Quebracho, Sambucus, Spongia, Sulphur, Valeriana
AMMONIUM CARBONICA
General Analysis: It produces a violent action upon the heart, in which there is audible palpitation, and every motion aggravates the pulsation. With this is associated great prostration. It is rather a strange coincidence that the ancients know that Amm. carb. would overcome difficult breathing from cardiac attacks and that aqua ammonia or hartshorn is used today to a certain extent in indications similar to those mentioned. They use it as a stimulant. (Kent)
Distinguishing Features:The diseased conditions met by this remedy are such as we find often in rather stout women who are always tired and weary, take cold easily, suffer from cholera-like symptoms before menses, lead a sedentary life, have a slow reaction generally, and are disposed to frequent use of the smelling-bottle.(Boericke)
Respiratory: Much oppression in breathing; worse after any effort, and entering warm room, or ascending even a few steps. Slow labored, stertorous breathing; bubbling sound. (Boericke)In diphtheria, when the nose is stopped, the child starts from sleep gasping for breath. Here again we observe its relation to Lachesis and the ophidia, for soon after the patient drops to sleep he wakes up suffocating, in diphtheria, in chest troubles with great prostration, the patient is worse after sleep.(Kent)
Heart: Audible palpitation with fear, cold sweat, lachrymation, inability to speak, loud breathing and trembling hands. Heart weak, wakes with difficult breathing and palpitation. (Boericke)It belongs to the simple enfeeblement, weak heart, emaciation. There is quite an absence of symptoms and a lack of response to remedies. The patient must lie in bed because of the palpitation and difficult breathing on motion. It is a matter of mere weakness.(Kent)
Asthma: Not an asthmatic dyspnoea; it is a cardiac dyspnoea due to a weak heart; but this remedy has also asthma, and in the asthma there is this peculiarity: if the room is warm the dyspnoea increases until suffocation seems imminent; as if he would die for want of breath. He is compelled to go out into the cold air for relief. While the warm room increases the dyspnoea in asthmatic complaints the bodily state of the patient is worse from cold.(Kent)
Sleep:
- Sleepiness during the day. Starts from sleep strangling. (Boericke)
- Loses breath at moment when falling asleep; wakens to get breath. (Hering)
- Restless, unrefreshing sleep; tosses about. (Hering)
- Frequent violent starting out of sleep, with great fear afterwards. (Hering)
ANTIMONIUM TARTARICUM
General Analysis:Acts directly upon the nerve centers at the base of the brain and in the medulla oblongata; through these Tartar Emetic exerts its power on the mucous membranes of the stomach, lungs, and liver, at the same time through the pneumogastric nerve depressing in a remarkable degree the circulatory and respiratory systems. (Cowperthwaite)
Distinguishing Features: The most essential characteristic of Tartar Emetic is the large accumulation of mucus in the chest, causing much rattling, especially in children, with vomiting and drowsiness.(Cowperthwaite)First, in catarrhal patients, in broken down constitutions, in feeble children, in old people. Catarrhal conditions of the trachea and the bronchial tubes. Our ears being open we hear coarse rattling and bubblings in the chest.(Kent)
Respiratory:
Asthma. Suffocation at 3 a.m., with necessity to sit up (Kali iod.), amel. cough and expectoration; S. in evening in bed, with necessity to sit up all night.Breath arrested. Dyspnœa; with necessity to be supported in a sitting position in bed. (Ars.). Short breath. Slow and labored B. Breathing hindered by swelling of pharynx and accumulation of tough mucus, with fever and delirium. Snoring expiration in sleep, with intermittent inspiration to two jerks. Clinical: Threatening suffocation; with wheezing and rattling and cyanosis, cannot clear the lungs, dyspnœa on falling asleep.(T.F.Allen)
Respiration short, rapid, heavy, anxious, difficult; must be supported in a sitting posture in bed. Suffocated and oppressed; cannot get air; has to sit up inbed. Shortness of breath from suppressed expectoration.Oppression of breathing, relieved by expectoration.Anxious, with oppression of the chest and rising of warmth to the heart; constriction of the chest.(Cowperthwaite)
Rapid, short, difficult breathing; seems as if he would suffocate; must sit up. Dyspnœa relieved by eructation. Cough and dyspnœa better lying on right side–(opposite Badiaga). (Boericke)
Paroxysms of coughing, with suffocating obstruction of respiration (suffocating cough).Dyspnœa, compelling one to sit up. Shortness of breathing from suppressed expectoration Suffocating attacks with sensation of heat at the heart.(Clarke)
Chest: Frequent fits of obstructed respiration, esp. in the evening or in the morning, in bed, almost to the extent of suffocation.Shortness of breath.Difficult respiration.Paralysis of the lungs.Anxious oppression of the chest, with a sensation of heat, which ascends to the heart.Rattling of mucus in the chest when breathing.(Clarke)
Modalities:Worse, in evening; from lying down at night; from warmth.(Boericke)
ARSENICUM
Distinguishing Features:Anxiety, restlessness, prostration, burning and cadaveric odors are prominent characteristics.(Kent)Its general symptoms often alone lead to its successful application. Among these the all-prevailing debility, exhaustion, and restlessness, with nightly aggravation, are most important. Great exhaustion after the slightest exertion. This, with the peculiar irritability of fiber, gives the characteristic irritable weakness. Burning pains. Unquenchable thirst. Burning relieved by heat.(Boericke)
Respiratory: Asthma at midnight, making him spring up; in sleep. Breathing difficult, with anguish; in sleep. Distressed, ending in asphyxia.(T.F.Allen)Unable to lie down; fears suffocation. Air-passages constricted. Asthma worse midnight. Burning in chest. Suffocative catarrh. Cough worse after midnight; worse lying on back. Wheezing respiration.(Boericke)Respiration oppressed, anxious, short. Oppressed, laboured breathing, esp. when ascending a height; in cold air; when turning in bed.(Clarke)
Chest: Shortness of breath, difficulty of respiration, choking, dyspnœa, and attack of suffocation, sometimes with cold sweat, spasmodic constriction of the chest or of the larynx, anguish, great weakness, body cold, pain in the pit of the stomach, and paroxysm of cough. The sufferings occur chiefly in the evening in bed, or at night, when lying down. Respiration anxious, stertorous, and wheezing.(Clarke)
Loss of breath immediately on lying down, in the evening, with whistling and constriction in the trachea. Cough suffocating at night; must sit up. Difficult breathing, with great anguish. Oppression; want of breath; must spring out of bed at midnight. Air passages seem constricted. (Hering)
Sleep: Disturbed, anxious, restless. Must have head raised by pillows. Suffocative fits during sleep.(Boericke)In the night, jerking of the limbs, heat and agitation, burning under the skin, as if there were boiling water in the veins, or cold, with inability to get warm, stifling sensation in the larynx, asthmatic attacks, great agitation, and anguish at the heart.Frequent waking during the night, with difficulty in sleeping again.Sleeplessness, from anguish and restlessness, with tossing about (after midnight). (Clarke)
Modalities: Aggravation.- At night, especially after midnight (1 to 3 A. M.) ; from cold in general, cold air, cold food (ice cream), cold drinks, cold washing ; after eating and drinking ; from lying with head low.(Cowperthwaite)
BADIAGA(Fresh-water sponge)
Distinguishing Features:General soreness of the muscles and integuments of the whole body; flesh sore to touch, even of the clothes; sore as if beaten.(Clarke)Soreness of muscles and whole body, causing a restless and sleepless night.(T.F.Allen)
Respiratory: While lying on right side in bed, and at moment of becoming unconscious by sleep, severe oppressive suffocative attacks from suspended respiration, causing a quick effort to prevent suffocation by changing position. (Clarke)(Hering)
CADMIUM SULPHURATUM
General Analysis:Its pathogenesis gives symptoms corresponding to very low forms of disease, as in cholera, yellow fever, where, with exhaustion, vomiting, and extreme prostration, the disease runs deathward.
Distinguishing Features:Its chief action centres on stomach and respiration. There is extreme exhaustion and prostration, with vomiting. Patient must keep quiet. Icy coldness even near a fire.(Phatak)
Respiratory/Sleep: Stops breathing on going to sleep. Wakes up suffocating. Fears to go to sleep again. Protracted sleeplessness.(Boericke)Interrupted breathing during sleep. On waking, want of air.(Hering) (T.F. Allen)
CAMPHORA
Distinguishing Features:It is characteristic of Camphor that the patient will not be covered, notwithstanding the icy coldness of the body.
RESPIRATION:Suffocative dyspnœa, as if from pressure in pit of stomach. Choking. Hurried respiration and labored. Short and snoring. (Inspiration in sleep shorter than expiration.). Oppressed, anxious, panting. Slow and shallow. Deep and slow. Heavy, slow and difficult. Almost arrested.(T.F. Allen)Præcordial distress. Suffocative dyspnœa. Asthma. Violent, dry, hacking cough. Palpitation. Breath cold. Suspended respiration.(Boericke)Breathing: anxious; oppressed; sighing; quiet; deep and slow; snoring; almost completely arrested, neither audible nor visible. Suffocative oppression. Choking sensation; it threatens to suffocate him and constrict larynx. Inhalation shorter than exhalation, in sleep. Snoring in sleep, with inhalation and exhalation. Asphyxia, or apparent death of newborn children.
Suspended respiration; heart ceases to beat, life seems gone.(Hering)
SLEEP: Snoring.(T.F. Allen)Snoring and tossing during sleep. During sleep, the inspirations are shorter than the expirations. (Clarke)
CARBO ANIMALIS
Distinguishing Features: The Carbon group all have putrid discharges and exhalations. Glandular enlargements and catarrhal states, flatulency and asphyxiation.(Boericke)
Respiratory: Cough, which impedes the breath. Suffocative, in evening after going to sleep. Fear of suffocating in evening while lying with closed eyes, amel. sitting up and opening eyes, it prevented sleep all night, with mucus in throat. Breath seems arrested in chest. Catching of breath on inspiration, with oppression of chest.(T.F. Allen) Dyspnœa with anxiety and low spirits. Oppression in morning and after eating. Panting and rattling breathing. (Hering)
Chest: Constriction; in morning in bed, with suffocation, she thought she would die, with stitches in heart on speaking and sensation on moving arm as if heart and chest would be torn to pieces, with feeling as if whole chest were oppressed or over-fatigued; sudden on deep breathing.(T.F. Allen)
Sleep: Sleep deferred, and nocturnal sleeplessness, caused, by inquietude, anguish, ebullition of the blood, and fear of being stifled. Frightful visions before going to sleep. (Clarke)
CARBO VEGETABILIS
General Analysis:It affects the vascular system especially; more particularly the venous side of the economy, the heart, and the whole venous system. Sluggishness is a good word to think of when examining the pathogenesis of Carbo veg. Everything about the economy is sluggish, turgid, distended and swollen. The Carbo veg. patient suffers from chronic catarrh.(Kent)
Distinguishing Features: The typical Carbo patient is sluggish, fat and lazy and has a tendency to chronicity in his complaints. Blood seems to stagnate in the capillaries, causing blueness, coldness, and ecchymosis. Body becomes blue, icy-cold. Face.–Puffy, cyanotic. Pale, hippocratic, cold with cold sweat; blue. Mottled cheeks and red nose.(Boericke)
Sleep: Loud breathing in sleep. Breath stopped; immediately on falling asleep, with increased vertigo. (T.F. Allen)On going to sleep there is anxiety, suffering, jerking, twitching, and he has the horrors. Horrible visions; sees ghosts.A peculiar sluggish, death-like sleep, with visions. The Carbo veg. patient wakens in anxiety and covered with cold sweat. Exhaustion. Unrefreshed after sleep.And thus the whole patient is prostrated by his sleep. So anxious that he does not want to go to sleep. Anxiety in the dark. Anxiety with dyspnoea as if he would suffocate. Anxiety so great that he cannot lie down. Like Opium; he cannot lift the head from the pillow. Painful throbbing in the head during inspiration. The Carbo veg. patient takes short breaths, quietly, keeping just as still as possible, until finally he is compelled to take a deep breath, and it comes out with a sharp moan.(Kent)
CENCHRIS CONTORTRIX (Copperhead snake)
Distinguishing Features: Cenchris has the main features of all the serpent poisons: Coma; semi-consciousness; insensibility of cornea; swelling of upper lip; general swelling; paralysis; cold, clammy sweat. Distinctive symptoms are: a marked alternation of moods; and dreamy absent-mindedness.(Clarke)
Respiratory: Suffocating feeling after lying down in the evening (7th day). Stops breathing on going to sleep (8th day). After lying down, a suffocating feeling came over her, with anxiety in the chest as if she would die, worse on first lying down. She must lie with head drawn back, as she chokes so (8th day). Dyspnea on lying down and the thought of sleep brings on great anxiety (12th day). She says: “There is no use lying down, that suffocation will come.” (12th day). Great difficulty of breathing at night, she had to gasp and struggle to breathe. (9th day).(Kent)
GRAPHITES
RESPIRATION:Suffocative sensation, waking her at night. Dyspnœa in evening on lying down and cough on deep inspiration; sudden D., with difficult short breath. Difficult breathing. Clinical: Spasmodic asthma, with suffocative paroxysms, which awaken from sleep, amel. eating. (T.F. Allen)Constriction of chest; spasmodic asthma, suffocative attacks wakes from sleep; must eat something.(Boericke)
CHEST: Difficulty of respiration and oppression on the chest. Nocturnal attacks of suffocation, on going to sleep, or on walking in the open air. Suffocative paroxysm at night, awakens him out of sleep, usually after midnight; must quickly jump out of bed, hold himself firmly to something, and quickly eats whatever is at hand, which gives relief; or hoarse cough (asthma).Wheezing respiration. (Clarke)
SLEEP:Great agitation at night, with anxious and frightful dreams, oppression and choking. During sleep, starts, with fright. Wakens at night from a suffocative attack. (Clarke) He suffocates when falling asleep (like Lachesis) and often wakes up in the night gasping for breath; constriction of the chest.(Kent)Suffocative paroxysm at night, awakens him out of sleep, usually after midnight, must quickly jump out of bed, hold himself firmly to something, and quickly eats whatever is on hand, which gives relief; very hoarse cough. θ Asthma spasmodicum.(Hering)
GRINDELIA
Clinical: Paroxysmal dyspnœa, with palpitation, faintness, inability to lie down, starting up with convulsive efforts to breathe, with livid face. Cheyne-Stokes respiration, if the patient drops off to sleep he stops breathing and wakes with a start and gasps for breath (Lach., Gel., Cur.). Œdema of the lungs and broncho-pneumonia in cases of fatty degeneration of the heart, can breathe only when sitting up, has to keep awake to breathe. Chronic pneumonias, with purulent expectoration, great dyspnœa, asthmatic expiration.(T.F. Allen)
RESPIRATION
Stops breathing when falling asleep; wakes with a start, and gasps for breath. Must sit up to breathe. Cannot breathe when lying down. Weak heart and respiration. Cheyne-Stokes respiration.(Boericke)
There was a kind of paralysis of the vagus nerve: on falling asleep the respiratory movement would cease and would not be resumed until he waked up from the resulting suffocation. In the clinical experiences recorded it is the Robusta which is chiefly mentioned, though sometimes it is not specified which. One writer obtained excellent results from “Grindelia” in a number of cases of asthma and emphysema with dilated heart. H. W. Foster reported the cure of a case of asthma with G. robusta which shows that Dr. Bundy’s proving of G. squarrosa is good for each drug. (Hering erroneously gives Bundy’s proving under the heading “Grindelia robusta.”) Dr. Foster’s patient was awakened by asthmatic attacks, the room seeming too small. After getting quiet, just as he would commence to lose himself in sleep he would suddenly awaken again because he seemed to have forgotten to breathe. Grind. r. 30 relieved at once. (Clarke)
SLEEP
Smothering after falling asleep.(Clarke) Feels respiration has stopped.(Clarke)Fear of going to sleep on account of loss of breath which awakens him (S).On falling asleep respiratory movement ceases, and is not resumed until awakened by suffocation resulting. Humid asthma from catarrhal bronchitis. Cardiac asthma.Cough from reflex causes.Much tenacious mucus difficult to detach.(Clarke)When dropping off to sleep the breath stops, the patient awakens catching for it, with a gasp, cannot get to sleep on this account. Stops breathing when falling asleep; wakens with a start.(Nash)After an attack of pneumonia vital forces so greatly reduced that recovery was despaired of; patient almost in a sitting position in bed, with a peculiar interrupted breathing, which entirely prevented sleep; every time he dropped into a doze, breathing would become so interrupted that he would have to be aroused; pneumogastric nerve appeared half paralyzed and action of heart was very weak. Cardiac asthma.(Hering)
LACHESIS
CLINICAL:There is extreme sensitiveness of the larynx to external touch, and especially a feeling of suffocation and constriction, so that the patient cannot bear anything tight about the throat; the cough is spasmodic, suffocative and wakens from sleep. Dyspnœa so great that the patient has to sit up, cannot lie down on account of suffocative fulness in chest, and cannot bear anything tight about neck or chest.(T.F. Allen)
PHARYNX: A feeling of a lump in the throat wakens out of sleep.(T.F. Allen)In the sore throat we have a combination of symptoms. Lachesis has produced this state, going from left to right; but with the sore throat there is a sensation of fullness in the neck and throat, difficult breathing, pallor or plethoric appearance of the face, choking when going into sleep, the peculiar kind of saliva and aggravation of the throat symptoms from warm drinks. (Kent)
RESPIRATION: Breathing almost stops on falling asleep (Grind.).(Boericke) Suddenly something runs from neck to larynx, and interrupts breathing; awakens him at night; spasm of glottis.(Cowperthwaite)Sensation in throat as of choking; springing out of sleep suddenly as if from a dream.(Clarke)
HEART: Prolonged melancholy, mental depression, hysterical symptoms, weeping, mental prostration and despair, with pain in the heart, with a gone sensation or sensation of weakness in the heart, with difficult breathing. (Kent)
SLEEP: The sleep has been disturbed by attacks of suffocation and by awful dreams, and now, after having slept a long time, he arouses with dreadful headaches, with palpitation, with melancholy, with sorrows from head to foot.(Kent)
In many symptoms of Lachesis, there is morning aggravation. This is the well-known Lachesis aggravation after sleep; the patient will sleep into the aggravation. In the milder symptoms this aggravation is mild and is not felt until after the patient wakes up from a long sleep, but if the aggravation is one that is of considerable violence, the patient may feel it immediately on going to sleep, and it arouses him; for instance, the heart symptoms. As soon as he goes into a sleep he rouses up with palpitation, with dyspnoea, with suffocation, with exhaustion, with vertigo, with pain in the back of the head, and many other circulatory disturbances.(Kent)
Desperate fits of suffocation, must sit up in bed. Oppression of chest during sleep. Cannot lie down from a sense of suffocation and must open doors and windows to obtain air. Great dyspnœa,< in afternoon and after sleep; left side. Chest feels constricted. Suddenly something runs from neck to larynx and interrupts breathing completely; it wakens at night. θ Spasmus glottidis. Cannot sleep, as soon as he falls asleep breathing is immediately interrupted. θ Acute rheumatism. Suffocating fits, waking from sleep with throwing arms about; cyanotic symptoms; swelling of liver; black urine. θ Hydrothorax. Awakes with fear of suffocation in middle of night.(Hering)
LAUROCERASUS
Distinguishing Features:Lauro. has cyanosis both of the new-born infant and of heart disease. A peculiar “gasping” is indicative here─ gasping without really breathing. In addition to the blueness there is twitching of the muscles of the face (which is also an indication for Lauro. in chorea). Clubbing of fingers, which is a common feature in cyanosis and phthisis, is a characteristic of Lauro. “Lack of reactive power,” low vitality, is another keynote of Lauro. This is particularly so when occurring in chest affections. Long-lasting faints; seems to have no reactive power; face pale, blue; surface cold.(Clarke)
RESPIRATION
The many strange constitutional symptoms indicate feeble circulation and weak heart.(Kent)
Spells of profound sleep with snoring or stertorous breathing.In the spells of suffocation, he must lie down but the dry hacking cough comes on as soon he lies down.
Difficult breathing. Suffocation, oppression of chest, gasping in heart complaints, ameliorated by lying down. Clutching at the heart and palpitation. It has cured mitral regurgitation many times.(Kent)
Cyanosis and dyspnœa; worse, sitting up. Patient puts hands on heart. Cough, with valvular disease. Exercise causes pain around heart. Tickling, dry cough. Dyspnœa. Constriction of chest. Cough, with copious, jelly-like, or bloody expectoration. Small and feeble pulse. Threatening paralysis of lungs. Gasping for breath; clutches at heart.(Boericke)
HEART
Pains in region of heart. Slow and irregular beating of heart. The patient puts his hand to his heart, as if there was some trouble there; this may result from running a short distance, which puts him completely out of breath; going upstairs, walking, any exercise may bring, this on.(Clarke)Mitral regurgitation. Clutching at heart and palpitation. Cyanosis neonatorum.(Boericke)
SLEEP
Spells of deep sleep, with snoring and stertorous breathing.(Boericke)
Deep snoring sleep.Soporous condition.Somnolence, sometimes like coma vigil.(Clarke)
OPIUM
General Analysis:The leading feature of Opium is its depression of the cerebral functions, indicated by great drowsiness and stupor, with stertorous breathing. (Cowperthwaite)
Clinical: Bronchial catarrh, with dyspnœa, suffocative attacks during sleep, deep snoring respiration, great difficulty in lying down. Bronchial catarrh, wakes with suffocation, great dyspnœa and blueness of the face. (T.F. Allen)
RESPIRATORY: Suffocative attacks; in sleep; and suddenly became blue in face and tried to cough, but had no breath, then deep sleep, with cold sweat. Choking sensation. Rattling. Grunting. Long and sighing; deep and frequent sighing. Interrupted; inspiration. Convulsive, 10 per minute, then did not breathe for a minute. Suspended; for a minute at a time, then begins with a sigh, then short, sudden gasps as if he would suffocate. Slow respiration; and groaning; and sighing; and now and then moaning; and stertorous; and stertorous, irregular; and stertorous, deep, puffing; and stertorous, interrupted, gasping; and sonorous; and loud; and rattling, irregular; with at times deep inspiration; and feeble; almost imperceptible, intermittent; obstructed by mucus; and stertorous, labored, irregular, sometimes more than a minute between the respirations, then sudden snoring inspiration; with rapid, thready pulse.
Breathing stops on going to sleep; must be shaken to start it again. Deep snoring; rattling, stertorous breathing. Difficult, intermittent, deep, unequal respiration.(Boericke)
Rattling breathing.Respiration deep; unequal.Deep snoring breathing, with open mouth. Noisy, stertorous, and rattling inspiration.Where there is continued and steady stertorous breathing. Difficult, slow, and intermittent respiration, as from paralysis of the lungs: pneumonia notha. Obstructed respiration and stifling, with great anguish. (Clarke)
Deep snoring respiration, with open mouth. Frequent involuntary deep breathing; long and sighing respiration. Respiration irregular, slow, and stertorous.(Cowperthwaite)
Paroxysms of suffocation come on during sleep and are apt to be followed by violent fits of dry, racking cough > for a time by drinking water. Loss of breath on going to sleep. Suffocative attacks during sleep, like nightmare.(Hering)
SLEEP: Great drowsiness. Falls into a heavy stupid sleep. Profound coma. Loss of breath on falling asleep.(Boericke)Loud snoring. If he sleeps the sleep is stertorous. Loss of breath on falling asleep. Lethargy, with snoring and mouth open, eyes open and convulsed, face red, and puffed, jaw hanging, loss of consciousness, difficult, slow, or intermittent respiration, pulse slow, or even suppressed, and convulsive movement of muscles of face, corners of mouth, and limbs.Incomplete sleep, without power to wake.Uneasy sleep, with anxious dreams. Stupefying, unrefreshing sleep. (Clarke)
Quebracho (Aspidosperma)
General Analysis:Characteristics.─Quebracho is a Brazilian fever remedy from which the alkaloid Aspidospermine has been isolated. Hale says Queb. produces in animals respiratory paralysis, slowed heart, and paralysis of extremities. It relieves dyspnœa in phthisis and pleurisy, but without influencing the fever. The 1x relieved asthma with livid face; and dyspnœa with cyanosis is frequently relieved by it. Hale gives these cases as relieved by it:
(1) Mitral incompetence and stenosis with severe nocturnal dyspnœa.
(2) Fatty heart (Queb. had no influence on the œdema, which was removed by Dig.).
Jos. P. Cobb (quoted A. H., xxvii. 74) records a case of heart affection of some duration in a man, 24. There was some enlargement, especially of right side, much dyspnœa, and a slight mitral murmur. Following this were signs of emphysema and severe attacks of asthma. Râles were heard, and “pearls” of rounded gelatinous masses were expectorated. Aspidospermine 3x gave more relief than any other remedy. (Clarke)
Picot, Skoda, Krauth, Penzoldt, and many others, have used it in various instances of difficult breathing, with remarkable palliation. In emphysema, spasmodic asthma, uraemic asthma, in spasmodic cough, chronic bronchitis, with asthmatic breathing, it has afforded very considerable relief. It has appeared to be much less useful, if not injurious, in the dyspnoea from valvular disease of the heart, and has had no effect in the dyspnoea of old subjects due to atheroma of the vessels. Krauth, however, has used it with advantage in the dyspnoea due to hypertrophy of the heart and in the difficulty of breathing in a case of albuminuria consecutive to scarlatina. The relief to dyspnoea in all cases, Penzoldt thinks, is referable to the increased consumption of oxygen by the blood, but a more rational explanation would seem to be the action on the respiratory center, and the diminution in the sense of need of air.(Bartholow)
Distinguishing Features:
The digitalis of the lungs (Hale). Removes temporary obstruction to the oxidation of the blood by stimulating respiratory centers, increasing oxidation and excretion of carbonic acid. Pulmonary stenosis. Thrombosis of pulmonary artery. Uræmic dyspnœa. An effective remedy in many cases of asthma. It stimulates the respiratory centers and increases the oxygen in the blood. “Want of breath” during exertion is the guiding symptom. Cardiac asthma.(Boericke)
Clinical:Asthma, Cardiac asthma. Fever.(Clarke)
SAMBUCUS
SLEEP: Starting out of sleep, with anxiety, suffocation and trembling. Slumber, with half-open eyes and mouth, on waking could not catch his breath, obliged to sit up, when the breathing was rapid, with whistling in chest, as if he would suffocate, he beat about with his hands, head and hands bluish and puffy, with heat without thirst, when the attacks came on he cried out, aggravation at night, from 12 till 4.(T.F. Allen) Frequent waking with a start, with anguish, trembling, and obstructed respiration, amounting almost to suffocation.(Clarke)Frequent awaking, as in a fright, with anxiety, trembling, dyspnœa, as if he would suffocate.(Hering)
RESPIRATORY: Acts especially on the respiratory organs. Chest oppressed with pressure in stomach, and nausea. Hoarseness with tenacious mucus in larynx. Paroxysmal, suffocative cough, coming on about midnight, with crying and dyspnœa. Spasmodic croup. Dry coryza. Sniffles of infants; nose dry and obstructed. Loose choking cough. When nursing child must let go of nipple, nose blocked up, cannot breathe. Child awakes suddenly, nearly suffocating, sits up, turns blue. Cannot expire. Millar’s asthma.(Boericke)
Nightly suffocative attacks, with great restlessness; shedding of tears and throwing about of arms. Suffocating attacks after midnight. Child suddenly awakes, nearly suffocated, sits up in bed, turns blue, gasps for breath, which it finally gets; spell passes off; it lies down again, and is aroused sooner or later in the same manner. θ Asthma Millari.
Drowsiness, breathes with mouth open and with a snoring whistling sound, head bent backward, suddenly springs up, tossing arms about, face becoming blue, suffocation seems inevitable when cough recurs, then with rattling breathing child sinks back exhausted; threatened paralysis of lungs. Violent asthmatic attack, respiration rapid and whistling; occasional short but labored coughs; high degree of dyspnœa; constantly pointing to middle of chest; had been suffering twenty-four hours, with intermissions of two or three hours, the free intervals becoming gradually shorter with each attack (which lasted usually from ten to fifteen minutes); threatened suffocation with marked blueness of lips. Roused about 2 or 3 A. M. with sense of stoppage of air tubes as by phlegm. Asthma thymicum; suppressed perspiration; attack comes on suddenly; awakes from a kind of lethargy with eyes and mouth open; raises himself in bed with great anxiety and dyspnœa; respiration oppressed, with wheezing in chest; head and hands puffed and bloated, with dry heat all over body; no thirst; small, irregular and intermittent pulse; no cough; paroxysms occur principally from midnight until 4 A. M.; difficulty of inspiration but not of expiration. Attacks of suffocation, resembling last stage of croup.(Hering)
SPONGIA
CLINICAL: Goitre; hard, with suffocative spells at night, sometimes with hoarse cough or staring eyes. Laryngitis, harsh, croupy cough, with suffocative spells rousing from sleep, larynx sensitive to touch, agg. turning the head ; (in croup Spongia is always indicated by the suffocative attacks, resembling Brom.) Oppression of breathing, amel. eating a little. Bronchial catarrh, with wheezing asthmatic cough, amel. eating or drinking, agg. cold air; sometimes in bronchitis, with profuse expectoration and suffocative attacks, agg. lying with the head low and in a hot room, amel. eating.
SLEEP
Awakes in a fright, and feels as if suffocating. Generally worse after sleep, or sleeps into and aggravation (Lach).(Boericke)
Awakens towards morning from a jerk upwards from the larynx, as if she would suffocate, must sit up, and raises sour, salty mucus.(Clarke)
In cardiac and asthmatic troubles it resembles Lach., inthe rousing up from sleep in suffocation; after the sleep the dyspnoea is worse.(Kent)
Falling asleep early at night; suffocation awakens. Awakens in a fright and feels as if suffocating. Awakening with fear and terror. Frequent waking with a start. Starting out of sleep toward morning from a shock in trachea upward like a suffocative paroxysm, passing off after sitting up in bed. Awakened between 1 and 2 A. M. by sense of suffocation.(Hering)
RESPIRATION
Awakes with suffocative sensation; wakes up choking when falling asleep.Suffocation on moment of falling asleep. Wakes with suffocation about larynx; on falling asleep early at night; fair skin. Suffocative paroxysm, with pain in chest.(Hering)
CHEST
The whole respiratory apparatus is acted upon; cardiac dyspnoea and the most severe forms of asthma. Dryness of the air passages with whistling and wheezing, seldom rattling, must sit up and bend forward; at times after great dyspnoea, white, tough mucus forms in the air passages, difficult to expectorate; it comes up and often has to be swallowed (Arn., Caust., Lach., Kali c., Kali s., Nux mos., Sep., Staph.) Dyspnoea worse lying down.(Kent)
Female: During menses, wakes with suffocative spells (Cupr; Iod; Lach).
Archibel, Keynotes, Radar: DYSPNEA: agg. After midnight, during sleep, during menses, hot room, lying head low.
SULPHUR
RESPIRATION
Oppression, as of a load on chest. Dyspnœa in middle of night, relieved by sitting up.(Boericke)
Nightly suffocative attacks, wants the doors and windows open; becomes suddenly wide awake at night; drowsy in afternoon after sunset, wakefulness the whole night. (H. C. Allen)
Feels suffocated; wants doors and windows wide open; particularly in night.(Hering)
Asthmatic attacks following swelling of hemorrhoids ; chest oppressed, constricted; nightly attack of suffocation ; great anxiety, rush of blood to chest, sudden cessation of respiration, gasps for air, jumps out of bed for relief; several attacks every night, followed by palpitation.(Hering)
CHEST/SLEEP
Shortness of breath; frequent chokings, obstructed respiration, dyspnœa, and fits of suffocation, esp. when lying down at night, and also during sleep. (Clarke)
VALERIANA
General Analysis: Through the cerebro-spinal system Valeriana affects directly the nervous centers, producing a high degree of irritation, which results in excessive nervous excitability, pain and spasms, all partaking of a hysterical character, which latter is the most important feature of the action of the drug.(Clarke)
Distinguishing Features:Hysteria, over-sensitiveness, nervous affections, when apparently well-chosen remedies fail. Hysterical spasms and affections generally.(Boericke)
Respiration: Choking on falling asleep.(Boericke)
Choking in throat-pit on falling asleep; wakens as if suffocating.Inspirations grow less and less deep and more rapid till they cease; then catches her breath by a sobbing effort in spells. Sensation as if something warm were rising from stomach, arresting breathing, with tickling deep in throat and cough.(Clarke)
Choking in throat-pit on point of falling asleep, she awakens as if suffocating.
Inspirations grow less deep and more rapid until they cease; then she catches her breath by a sobbing effort; so on in spells. θ Asthma.(Hering)
DIFFERENTIAL DIAGNOSIS OF REMEDIES FOR SLEEP APNEA
| REMEDY | RESPIRATORY | CARDIAC | HEAD TRAUMA | OSA or CSA or mixed |
| Amm carb | ++ | ++ | OSA, CSA, mixed | |
| Antim tart | +++++ | + | predominantly OSA | |
| Arsenicum | ++++ | + | predominantly OSA | |
| Badiaga | ++ | OSA | ||
| Cad sulph | +++ | OSA | ||
| Camphora | ++++ | + | predominantly OSA | |
| Carbo animalis | ++++ | + | predominantly OSA | |
| Carbo veg | ++ | + | OSA, CSA, mixed | |
| Cenchris | + | CSA | ||
| Graphites | ++++ | + | predominantly OSA | |
| Grindelia | ++ | ++++ | predom. CSA, mixed | |
| Lachesis | + | +++ | +++ | predom. CSA, mixed |
| Laurocerasus | ++ | +++ | predom. CSA, mixed | |
| Opium | ++++ | ++ | ++++ | OSA, CSA, mixed |
| Quebracho | ++ | +++ | OSA, CSA, mixed | |
| Sambucus | ++++ | OSA | ||
| Spongia | ++ | ++ | OSA, CSA, mixed | |
| Sulphur | ++ | + | + | OSA, CSA, mixed |
| Valeriana | + | + | OSA, CSA, mixed |
Scoring:
- respiratory: 1+ for being mentioned for breathing problem (snoring, stertorous breathing, choking, gasping, etc.)in bed, at night, and/or during sleep, in various materia medicas*;
- cardiac: 1+ for being mentioned for cardiac problem e.g. cardiac asthma, or weak heart, or pneumogastric (vagus) nerve,or Cheyne-Stokes respiration, in each materia medica;
- head trauma: 1+ for appearing in plain type, 2+ for appearing in italics, 3+ for appearing in bold, and 4+ for appearing in bold caps in relevant rubrics** in Schroyen’s repertory.
*Materia medicas used: T.F. Allen, Boericke, Clarke, Cowperthwaite, Hering, Kent. Also, Bartholow for Quebracho and Nash for Grindelia.
**Head trauma/concussion rubrics as listed on page 36.
SLEEP SURVEY
Using two slightly different versions of a questionnaire that I created, I surveyed over 230 people. (A copy of each of these questionnaires is provided on the last page as Appendix A & B.)
Questions asked on Sleep Survey included:
- How well do you sleep?
- Do you experience any (specified) symptoms or sleep issues on a regular basis?
- How much stress do you experience on a day-to-day basis?
- Have you been diagnosed with any sleep issues or other (specified) conditions?
- Have you ever experienced any (specified) trauma in your past?
RESULTS OF SLEEP SURVEY
The 238 responders ranged in age from 8 to 82 years old and included students, professionals, white and blue collar workers, homemakers, and retirees. 63 people reported experiencing a lot of stress in their lives. There was nostrong positive correlation between degree of perceived stress and quality of sleep,but there was a strong positive correlation between degree of perceived stress and bruxism (tooth grinding/clenching/TMJ dysfunction or TMD).
| Symptoms | Sleep Disorders | Conditions | History of Trauma |
| Sweating: 29 | Tooth grinding/ clenching/TMD: 52 | Depression: 26 | Losing consciousness: 30 |
| Shortness of breath: 8
|
Nightmares: 29
|
Anxiety: 27
|
Near drowning: 17
|
| Palpitations: 4 | Sleep apnea: 16 | Fibromyalgia or
Chronic fatigue: 7 |
Near death: 16 |
| Night terrors: 2
|
Blue baby: 3 |
The table below showed the number of responders who reported experiencing symptoms during sleep, sleep disorders, medically-diagnosed conditions, and history of specific trauma.
Of the sleep disorders reported, bruxism (tooth grinding or clenching, with or without TMJ dysfunction) was the most common (52), followed by nightmares (29), then sleep apnea (16), and finally, night terrors (2).
Two of the 16 responders who reported sleep apnea had a history of near drowning. Two reported losing consciousness. Two reported other trauma. Three were also bruxists.
| Past Trauma | Sleep Apnoea | Bruxism |
| Lost consciousness | 2+0=2 | 8+6=14 |
| Near drowning/Near death | 2+0=2 | 4+3=7 |
| Other trauma | 0+2=2 | 0+1=1 |
| No past trauma | 4+6=10 | 19+4=23 |
| Total # of sufferers | 8+8=16 | 30+22=52 |
The question about past trauma (losing consciousness, near drowning, near death, blue baby) was included because of the correlation with sleep apnea that Dr. Damaris Drewry discovered. Two of the eight responders who reported sleep apnea had a history of near drowning; two reported losing consciousness; one was also a bruxist.
Unexpectedly, there was also a correlation between sleep bruxism and specific past trauma: 17 of the 52 sleep bruxists (approx. 1 in 3) reported losing consciousness unexpectedly or involuntarily, e.g. knocked out in accident or fight, or going under general anaesthesia as a child (8) or near drowning or near death experience (4).
CONCLUSION
It has long been known that increased upper airway resistance (e.g. due to increased nasal discharge and oedematous nasal mucosa in patients with allergic rhinitis) is responsible for disordered breathing in sleep as well as ten times more micro-arousals than in controls.108 These events do not cause behavioral awakening but can lead to excessive day time sleepiness.109 It has also been determined that sleep bruxism (SB) is associated with micro-arousals and is preceded by an increase in cardiac sympathetic activity.110 In their article, Genesis of sleep bruxism: motor and autonomic-cardiac interactions111, researchers noted that there is a sequence of events that begins in the heart (increase in autonomic sympathetic activity), followed by the brain,in the minutes and seconds, respectively, before the onset of activity in the suprahyoid muscles and finally by rhythmic masticatory muscle activity (RMMA). Results of their study suggest that “the onset of RMMA and SB episodes during sleep are under the influences of brief and transient activity of the brainstem arousal-reticular ascending system contributing to the increase of activity in autonomic-cardiac and motor modulatory networks.”
Certainly, the body remembers, as psychotherapist Babette Rothschild explains in her book The Body Remembers: The Psychophysiology of Trauma and Trauma Treatment. Is it conceivable that a body which, years before, had an experience of trauma, e.g. near-drowning, remembers how dangerous it was to breathe (and thereby inhale water) while losing consciousness, and when this memory is somehow triggered during sleep (the quintessential nightly loss of consciousness), a cascade of events occurs? The body (or more precisely, the heart) remembers that it would have been fatal to breathe in water, and so sends out the alarm to the brain, and the brain responds by stopping breathing. [The heart has a complex neural network of ganglia, neurotransmitters, proteins and support cells, a neural circuitry that enables the heart to act independently of the brain to learn, remember, and even make decisions. Heart transplant recipients have been known to take on characteristics and even memories of their donors.112,113]
As the conscious mind relaxes it hold with the approach of sleep, the trauma is re-lived and the body responds in a replay of the original freeze response or perhaps a misguided attempt to save itself (a “stop breathing to stay alive” program in a case of near-drowning, or breath-holding in a young child who learned to do so in a desperate attempt to remain under the radar of an abusive parent), but the coping mechanism proves to be more harmful than helpful in the long run.
Building on the work of John and Beatrice Lacy in the 1960s and 1970s, researchers from the Heart Math Institute, led by Dr Rollin McCraty, have determined that communication between the heart and brain is a dynamic, two-way process, with one continuously influencing the other. The heart communicates to the brain in four major ways: neurologically (transmission of nerve impulses), biochemically (hormones and neurotransmitters), biophysically (pressure waves) and energetically (electromagnetic field interactions).
In his article, Heart Rhythm Coherence – An Emerging Area of Biofeedback114, Dr McCraty notes that “the analysis of heart rate variability (HRV), or heart rhythms, provides a reliable measure of autonomic nervous system dynamics that is particularly sensitive to changes in psychophysiological state”. Back in 1996, Dr McCraty presented a paper: Head-Heart Entrainment: A Preliminary Survey115 at an Applied Neurophysiology Colloquium, in which he described the degree of entrainment exhibited between heart rate variability (HRV), respiration, and EEG recordings in 5 subjects trained in the use of a self-management technique.
It stands to reason that severe trauma is not simply forgotten but is often stuck in our energy systems, waiting to be triggered once our stress levels go above a critical level. Childhood trauma can be devastating in both the short and long term. The Adverse Childhood Experience (ACE) Study116 carried out by the Centres for Disease Control and Prevention in 1995, which followed 17,000 respondents over fifteen years, found a direct relationship between childhood abuse, neglect, household dysfunction (e.g. divorce, alcoholic parent, violence, substance abuse) and severely compromised health outcomes later in life.
In all cases of sleep disordered breathing, it is imperative to bring to light any history of trauma in both the near and distant past, all the way to the patient as an infant during the birthing process (drugs given to the mother, forceps delivery, umbilical cord around neck), as a newborn soon after birth (separation from mother, unnecessary vaccinations), and even as the unborn foetus sensitive to the mother’s mental and emotional state. All this information is invaluable to the homeopath who understands the need to go beyond the obvious signs and symptoms of the presenting complaint to the causative or NWS factor that may hold the clue to cure.
There is no question that serious consequences can arise when sleep apnea is left untreated or treated inappropriately. While CPAP machines may be life-saving for some, it is palliative at best and so it is vital to keep looking for the underlying cause. In the case of primary or psychophysiological CSA, causation is trauma, either physical or mental/emotional. In the case of OSA, especially in children with adenotonsillar hypertrophy, an underlying tubercular constitution may need to be addressed. These children are expressing an inherited miasm that may be latent in asymptomatic siblings who, incidentally, may also benefit from constitutional treatment that can remove the risk of the latent miasm activating from stressors later on in life.
While genetics certainly play a part in inherited diseases, the study of epigenetics gives us hope that lifestyle changes (e.g. better nutrition, breastfeeding for the first six months) can optimize a child’s health potential, while learnable skills such as biofeedback for heart coherence can greatly improve our resilience to stress. Various energy modalities canhelp defuse the emotional intensity of past trauma, but homeopathy may be the only form of energy medicine able to undo the genetic imprint of inherited miasms that predispose us to conditions such as sleep apnea.
———————–
REFERENCES
BIBLIOGRAPHY
American Academy of Sleep Medicine. (2014). International Classification of Sleep Disorders. 3rd edition. Darian, Ill.
Banerjea, S.K. (2010). Miasmatic Prescribing. 2nd extended Indian ed. B. Jain Publishers (P) Ltd., New Delhi, 110 055.
Barlow, D. (2002). Anxiety and Its Disorders.The Nature and Treatment of Anxiety and Panic. Second Edition. New York, NY: The Guildford Press.
Champagnat, J., Denavit-Saubié, M., Fortin, G., Foutz, A.S.&Thoby-Brisson M. (2005). Post-Genomic Perspectives in Modeling and Control of Breathing. Advances in Experimental Medicine and Biology. Volume 55.
Levine, P.A. (2010). In an unspoken voice: how the body releases trauma and restores goodness.North Atlantic Books, Berkley, California 94712.
McCraty, R. (2015). Science of the Heart. HearthMath Institute. Boulder Creek, CA 95006.
Pien, G.W.&Pack, A.I. (2010). Sleep disordered breathing. In: Mason RJ, Broaddus VC, Martin TR, et al., eds. Murray and Nadel’s Textbook of Respiratory Medicine. 5th ed. Philadelphia, Pa: Elsevier Saunders:chap 79.
Rothschild, B. (2000). The body remembers: the psychophysiology of trauma and trauma treatment. W.W. Norton & Company, Inc., 500 Fifth Avenue, New York, NY 10110.
(2007). Obstructive Sleep Apnea.Diagnosis and Treatment.Ed. C.A. Kushida. Stanford University Stanford, California, USA.Sleep Disorders. Volume 4. Informa Healthcare, New York, NY 10017.
MATERIA MEDICAS
Allen, H. C. (1916). Keynotes and Characteristics with Comparisons of some of the Leading Remedies of the Materia Medica. Fourth Edition, Philadephia, Boericke and Tafel.
Allen, T. F. Hand Book of Materia Medica and Homœopathic Therapeutics.
Allen, T. F. (1874). Encyclopedia of Pure Materia Medica. 10 volumes.
Bartholow, R.(1889). A Practical Treatise on Materia Medica and Therapeutics.
Boericke, W. (1901). Pocket Manual of Homeopathic Materia Medica
Clarke, J. H. (1902). A Dictionary of Practical Materia Medica.
Cowperthwaite, A. C. (1887). A Text-Book of Materia Medica (Characteristic, Analytical, and Comparative.)Fourth edition revised and enlarged with clinical index. Chicago: Gross & Delbridge.
Hering, C. (1879-1891). The Guiding Symptoms of Our Materia Medica.10 volumes.
Kent, J. T. (1904). Lectures on Homeopathic Materia Medica.
Nash, E.B. (1898). Leaders in Homoeopathic Therapeutics.
SCIENTIFIC LITERATURE
- Javaheri, S., Smith, J., & Chung, E. (2009). The prevalence and natural history of complex sleep apnea. J Clin Sleep Med., 5(3):205-11.
- Morgenthaler, T.I., Kagramanov, V., Hanak, V., & Decker, P.A. (2006). Complex sleep apnea syndrome: is it a unique clinical syndrome? Sleep, 29(9):1203–1209.
- Malhotra, A., &Owens, R. L. (2010). What is central sleep apnea? Respir Care, 55(9): 1168–1178.
- Eckert, D.J., Jordan, A.S., Merchia, P., &Malhotra, A. (2007). Central sleep apnea. Pathophysiology and treatment. Chest, 131(2): 595–607. doi: 1378/chest.06.2287
- Heinzer, R. (2009). [Opioids and sleep disordered breathing]. Rev Med Suisse. Nov 18;5(226):2322-4, 2326-8. [Article in French]
- Walker, J.M., Farney, R.J., Rhondeau, S.M., Boyle, K.M., Valentine, K., Cloward, T.V., &Shilling, K.C. (2007). Chronic opioid use is a risk factor for the development of central sleep apnea and ataxic breathing. J Clin Sleep Med. Aug 15;3(5):455-61.
- Dempsey, J.A., Veasey, S. C., Morgan, B.J., &O’Donnell, C.P. (2010). Pathophysiology of Sleep Apnea. Physiol Rev., 90(1): 47–112. doi: 1152/physrev.00043.2008
- Becker, K. (2016). Central Sleep Apnea Syndromes. Retrieved from http://emedicine.medscape.com/article/304967-overview#a5
- Chowdhuri, S., & Badr, M.S. (2010). Central sleep apnoea. Indian J Med Res., (131):150-164. Retrieved from
- Badr, M.S., Toiber, F., Skatrud, J. B., & Dempsey, J. (1995). Pharyngeal narrowing/occlusion during central sleep apnea. Jour App Physiol, 78 (5): 1806-1815
- Verbraecken, J.A., & De Backer, W.A. (2009). Upper airway mechanics. Respiration. 78(2):121-33. doi: 1159/000222508
- Mayo Clinic. (2006). Mayo Clinic Discovers New Type Of Sleep Apnea. ScienceDaily. Retrieved from www.sciencedaily.com/releases/2006/09/060901161349.htm
- Marrone, O., Stallone, A., Salvaggio, A., Milone, F., Bellia, V., & Bonsignore. G. (1991). Occurrence of breathing disorders during CPAP administration in obstructive sleep apnoea syndrome. European Respiratory Journal, 4(6):660–666.
- Kuźniar, T.J., &Morgenthaler, T.I. (2008). Treatment of complex sleep apnea syndrome. Curr Treat Options Neurol.,10(5):336-41.
- Kuźniar, T.J., &Morgenthaler, T.I. (2012). Treatment of complex sleep apnea syndrome. Chest,142(4):1049-57. doi: 10.1378/chest.11-3223.
- Khan, M.T., & Franco, R.A. (2014). Complex sleep apnea syndrome. Sleep Disord. 2014: doi:10.1155/2014/798487. PMID24693440.
- de Godoy, L.B.M., Palombini, L.O., Guilleminault, C., Poyares, D., Tufik, S., &Togeiroa, S.M. (2015). Treatment of upper airway resistance syndrome in adults: Where do we stand? Sleep Sci., 8(1): 42–48. doi: 1016/j.slsci.2015.03.001
- Lee, W., Nagubadi, S., Kryger, M.H., &Mokhlesi, B. (2008). Epidemiology of Obstructive Sleep Apnea: a Population-based Perspective. Expert Rev Respir Med., 2(3): 349–364. doi: 1586/17476348.2.3.349
- Bixler, E.O., Vgontzas, A.N., Ten Have, T., Tyson, K., &Kales, A. (1998). Effects of age on sleep apnea in men: I. Prevalence and severity. Am J Respir Crit Care Med., 157(1):144-8. DOI: 1164/ajrccm.157.1.9706079
- Anttalainen, U., Saaresranta, T., Aittokallio, J., Kalleinen, N., Vahlberg, T., Virtanen, I., &Polo, O. (2006). Impact of menopause on the manifestation and severity of sleep-disordered breathing. Acta Obstet Gynecol Scand.;85(11):1381-8. doi:1080/00016340600935649
- Chang, S.J., &Chae, K.Y. (2010). Obstructive sleep apnea syndrome in children: Epidemiology, pathophysiology, diagnosis and sequelae. Korean J Pediatr.,53(10): 863–871. doi: 3345/kjp.2010.53.10.863
- Young, T., Peppard, P.E., &Gottlieb, D.J. (2002). Epidemiology of obstructive sleep apnea: a population health perspective.Am J Respir Crit Care Med, 165:1217–1239.
- Dehlink, E., Tan, H.L. (2016). Update on paediatric obstructive sleep apnoea. J Thorac Dis.; 8(2): 224–235. doi: 10.3978/j.issn.2072-1439.2015.12.04
- Rosen, C.L., Larkin, E.K., Kirchner, H.L., Emancipator, J.L., Bivins, S.F., Surovec, S.A., ….Redline, S. (2003). Prevalence and risk factors for sleep-disordered breathing in 8- to 11-year-old children: association with race and prematurity. J Pediatr, 142: 383–389.
- Ikävalko, T., Tuomilehto, H., Pahkala, R., Tompuri, T., Laitinen, T., Myllykangas, R., …Lakka, T.A. Craniofacial morphology but not excess body fat is associated with risk of having sleep-disordered breathing–the PANIC Study (a questionnaire-based inquiry in 6-8-year-olds). (2012). Eur J Pediatr, 171(12):1747-52. doi: 10.1007/s00431-012-1757-x
- Cherniack, N.S., Longobardo, G., &Evangelista, C.J. (2005). Causes of Cheyne-Stokes respiration. Neurocrit Care,3(3):271-9.
- Verbraecken, J. (2013). Complex sleep apnoea syndrome. Breathe, 9: 372-380. doi: 10.1183/20734735.042412
- Redline, S., Tishler, P.V., Schluchter, M., Aylor, J., Clark, K., & Graham, G. (1999). Risk factors for sleep-disordered breathing in children. Associations with obesity, race, and respiratory problems. Am J Respir Crit Care Med 159: 1527–1532
- McColley SA, Carroll JL, Curtis S, Loughlin GM. (1997). High prevalence of allergic sensitization in children with habitual snoring and obstructive sleep apnea. Chest, 111:170–3.
- Naresh M. Punjabi, N.M. (2008). The Epidemiology of Adult Obstructive Sleep Apnea. Proc Am Thorac Soc., 5(2): 136–143. doi: 10.1513/pats.200709-155MG
- Redline, S., Tishler, P.V. (2000). The genetics of sleep apnea. Sleep Med Rev.;4(6):583-602.
- Peppard, P.E., Young, T., Palta, M., & Skatrud, J. (2000). Prospective Study of the Association between Sleep-Disordered Breathing and Hypertension. N Engl J Med, 342:1378-1384. doi: 10.1056/NEJM200005113421901
- Punjabi, N.M., Shahar, E., Redline, S., Gottlieb, D.J., Givelber, R., Resnick, H.E. &Sleep Heart Health Study Investigators. (2004). Sleep-disordered breathing, glucose intolerance, and insulin resistance: the Sleep Heart Health Study. Am J Epidemiol.,160(6):521-30.
- Manni R, Terzaghi M, Arbasino C, Sartori I, Galimberti CA, Tartara A. (2003). Obstructive sleep apnea in a clinical series of adult epilepsy patients: frequency and features of the comorbidity. Epilepsia.;44:836-840
- Jackson, M.L., Howard, M.E., &Barnes, M. (2011). Cognition and daytime functioning in sleep-related breathing disorders. Prog Brain Res.,190:53-68. doi: 10.1016/B978-0-444-53817-8.00003-7.
- Park, J.G., Ramar, K., & Olson, E.J. (2011). Updates on Definition, Consequences, and Management of Obstructive Sleep Apnea. Mayo Clin Proc., 86(6): 549–555. doi: 10.4065/mcp.2010.0810
- Thiedke, C.C. (2001). Sleep disorders and sleep problems in children. Am Fam Physician, 63(2):277-285.
- Chan, J., Edman, J.C., Koltai, P.J. (2004). Obstructive Sleep Apnea in Children. Am Fam Physician, 69(5):1147-1155.
- Capdevila, O.S.,Kheirandish-Gozal, L., Dayyat, E., &Gozal, D.(2008). Pediatric Obstructive Sleep Apnea. Complications, Management, and Long-term Outcomes.Proc Am Thorac Soc., (5)274–282, doi: 10.1513/pats.200708-138MG
- Hermann, D.M., &Bassetti, C.L. (2003). Sleep-disordered breathing and stroke. Curr Opin Neurol., 16(1):87-90. doi:1097/01.wco.0000053587.70044.be
- Punjabi, N.M., Caffo, B.S., Goodwin, J.L., Gottlieb, D.J., Newman, A.B., O’Connor, G.T.,….Samet, J.M. (2009). Sleep-Disordered Breathing and Mortality: A Prospective Cohort Study. PLoS Med 6(8): e1000132. doi:10.1371/journal.pmed.1000132 Retrieved from: http://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1000132
- Park, P., Jeon,,H.W., Han, D.H., Won, T.B., Kim, D.Y., Rhee, C.S., Kim, H.J. (2016). Therapeutic outcomes of mandibular advancement devices as an initial treatment modality for obstructive sleep apnea. Medicine (Baltimore),95(46):e5265. doi: 1097/MD.0000000000005265
- Johnson, K.G., & Johnson, D.C. (2005). Bilevel positive airway pressure worsens central apneas during sleep. Chest, 128(4):2141–2150.
- Aurora, R.N., Chowdhuri, S., Ramar, K., Bista, S.B., Casey, K.R., Lamm, C.I.,…Tracy, S.L. (2012). The treatment of central sleep apnea syndromes in adults: practice parameters with an evidence-based literature review and meta-analyses. Sleep, 35:17-40.
- Boyd, S.B., Walters, A.S., Song, Y., &Wang, L. (2013). Comparative effectiveness of maxillomandibular advancement and uvulopalatopharyngoplasty for the treatment of moderate to severe obstructive sleep apnea. J Oral Maxillofac Surg., 71(4): 743–751. doi: 10.1016/j.joms.2012.10.003
- Carvalho, B., Hsia, J., Capasso, R. (2012). Surgical Therapy of Obstructive Sleep Apnea: A Review. Neurotherapeutics, 9(4): 710–716. doi: 10.1007/s13311-012-0141-x
- Dempsey, J.A., Xie, A., Patz, D.S., Wang, D. (2014). Physiology in Medicine: Obstructive sleep apnea pathogenesis and treatment—considerations beyond airway anatomy. Journal of Applied Physiology Published 1 January Vol. 116(1): 3-12. doi: 10.1152/japplphysiol.01054.2013
- Ramar, K., Dort, L.C., Katz, S.G., Lettieri, C.J., Harrod, C.G., Thomas, S.M., &Chervin, R.D. (2015). Clinical Practice Guideline for the Treatment of Obstructive Sleep Apnea and Snoring with Oral Appliance Therapy: An Update for 2015. J Clin Sleep Med., 11(7):773-827. doi: 10.5664/jcsm.4858.
- Marklund, M., Carlberg, B., Forsgren, L., Olsson, T., Stenlund, H., &Franklin, K.A. (2015). Oral Appliance Therapy in Patients With Daytime Sleepiness and Snoring or Mild to Moderate Sleep Apnea: A Randomized Clinical Trial. JAMA Intern Med., 175(8):1278-85. doi: 10.1001/jamainternmed.2015.2051.
- Hoffstein V. (2007). Review of oral appliances for treatment of sleep-disordered breathing. Sleep Breath,.11(1):1-22. doi: 1007/s11325-006-0084-8
- Whitla, L., &Lennon, P. (2016). Non-surgical management of obstructive sleep apnoea: a review. Paediatr Int Child Health, 14:1-5. doi: 1080/20469047.2016.1162391
- Tan, H.L., Gozal, D., Kheirandish-Gozal, L. (2013). Obstructive sleep apnea in children: a critical update. Nat Sci Sleep, 5: 109–123. doi: 10.2147/NSS.S51907
- Kohler M. (2009). Risk factors and treatment for obstructive sleep apnea amongst obese children and adults. Curr Opin Allergy Clin Immunol., (1):4-9. doi: 10.1097/ACI.0b013e32831d8184.
- Guilleminault, C., Huseni, S., Lo, L. (2016). A frequent phenotype for paediatric sleep apnoea: short lingual frenulum. ERJ Open Research 2: 00043-2016; doi:1183/23120541.00043-2016
- Schmidt, N.B., Richey, J.A., Zvolensky, M.J., &Maner, J.K. (2008) Exploring Human Freeze Responses to a Threat Stressor. J Behav Ther Exp Psychiatry, 39(3): 292–304. doi: 1016/j.jbtep.2007.08.002
- Sharafkhaneh, A., Giray, N., Richardson, P., Young, T., &Hirshkowitz, M. (2005). Association of psychiatric disorders and sleep apnea in a large cohort. Sleep, 28(11):1405-11.
- Colvonen, P.J., Masino, T., Drummond, S.P., Myers, U.S., Angkaw, A.C., &Norman, S.B. (2015). Obstructive Sleep Apnea and Posttraumatic Stress Disorder among OEF/OIF/OND Veterans.Journal of Clinical Sleep Medicine; 11(5):513-8. doi: 10.5664/jcsm.4692
- Heidt, J.M., Marx, B.P., &Forsyth, J.P. (2005). Tonic immobility and childhood sexual abuse: a preliminary report evaluating the sequela of rape-induced paralysis. BehavResTher43(9):1157-71.
- Krakow,, Germain, A., Tandberg, D., Koss, M., Schrader, R., Hollifield, M., Cheng, D., &Edmond, T. (2000). Sleep breathing and sleep movement disorders masquerading as insomnia in sexual-assault survivors. Comprehensive Psychiatry,41(1):49–56. http://dx.doi.org/10.1016/S0010-440X(00)90131-7
- Spoormaker, V.I., & Montgomery, P. (2008). Disturbed sleep in post-traumatic stress disorder: Secondary symptom or core feature? Sleep Medicine Reviews,12(3):169-184. doi: http://dx.doi.org/10.1016/j.smrv.2007.08.008
- Drewry, D. (2017). Central Nervous System Apnea Can Be Caused by Traumatizing Events and It Can Be Resolved.International Journal of Healing and Caring.Vol. 17, No. 1; ijhc.org
- Masaoka, Y., &Homma, I. (2004). Amygdala and Emotional Breathing in Humans. Advances in Experimental Medicine and Biology 551:9-14. doi: 10.1007/0-387-27023-X_2
- Kahn, A., Groswasser, J., Sottiaux, M., Kelmanson, I., Rebuffat, E., Franco, P., Dramaix, M., &Wayenberg, J.L. (1994). Prenatal exposure to cigarettes in infants with obstructive sleep apneas. Pediatrics, 93(5):778-83.
- Flores, Á., Saravia, R., Maldonado, R., &Berrendero, F. (2015).Orexins and fear: implications for the treatment of anxiety disorders. Trends Neurosci., 38(9):550-9. doi: 10.1016/j.tins.2015.06.005.
- Williams, R.H., &Burdakov, D. (2008). Hypothalamic orexins/hypocretins as regulators of breathing. Expert Rev Mol Med.,10:e28. doi: 10.1017/S1462399408000823
- Burdakov, D. (2008). Orchestrating brain activity. Retrieved from http://www.cam.ac.uk/research/news/orchestrating-brain-activity
- Gorman, J.M., &Uy, J. (1987). Respiratory physiology and pathological anxiety. Gen Hosp Psychiatry, 9(6):410-9.
- Leibold, N.K., van den Hove, D.L., Esquivel, G., De Cort, K., Goossens, L., Strackx, E., …Schruers KR. (2015). The brain acid-base homeostasis and serotonin: A perspective on the use of carbon dioxide as human and rodent experimental model of panic.Prog Neurobiol., 129:58-78.
- Esquivel G, Schruers KR, Maddock RJ, Colasanti A, &Griez EJ. (2010). Acids in the brain: a factor in panic? J Psychopharmacol., 24(5):639-47. doi: 10.1177/0269881109104847
- Nattie, E., &Li, A. (2012). Respiration and autonomic regulation and orexin. Prog Brain Res.,198:25-46. doi: 10.1016/B978-0-444-59489-1.00004-5
- Johnson, P.L., Molosh, A., Truitt, W. A., Fitz, S. D., & Shekhar, A. (2012). Orexin, Stress and Anxiety/Panic states. Progress in Brain Research, 198, 133-161. doi:10.1016/B978-0-444-59489-1.00009-4
- Ohno, K., &Sakurai, T. (2008). Orexin neuronal circuitry: role in the regulation of sleep and wakefulness. Front Neuroendocrinol., 29(1):70-87.
- Castriotta, R.J., Atanasov, S., Wilde, M.C., Masel, B.E., Lai, J.M., & Kuna, S.T. (2009). Treatment of sleep disorders after traumatic brain injury. J Clin Sleep Med, 5(2):137-144
- Castriotta, R.J.; Wilde, M.C.; Lai, J.M., Atanasov, S., Masel, B.E., &Kuna, S.T. (2007). Prevalence and consequences of sleep disorders in traumatic brain injury. J Clin Sleep Med 2007;3(4):349-356.
- Castriotta, R.J., & Lai, J.M. (2001). Sleep disorders associated with traumatic brain injury. Arch Phys Med Rehabil, 82:1403-1406.
- Verma A, Anand V, Verma NP. Sleep disorders in chronic traumatic brain injury. J Clin Sleep Med. 2007;3:357-362.
- Collen, J., Orr, N., Lettieri, C.J., Carter, K.A., & Holley, A.B. (2012). Sleep disturbances among soldiers with combat-related traumatic brain injury. Chest, 142:622-630.
- Viola-Saltzman, M., &Watson, N.F. (2012). Traumatic Brain Injury and Sleep Disorders. Neurol Clin., 30(4): 1299–1312. doi: 1016/j.ncl.2012.08.008
- George, C.F.P., Kab, V., Levy, A.M. (2003). Increased prevalence of sleep-disordered breathing among professional football players. N Engl J Med, 348:367-368. doi: 10.1056/NEJM200301233480422
- Crisco, J.J., Wilcox, B.J., Beckwith, J.G., Chu, J.J., Duhaime, A.C., Rowson, S., Duma, S.M., Maerlender, A.C., McAllister, T.W., &Greenwald, R.M. (2011). Head impact exposure in collegiate football players. J Biomech., 44(15):2673-8. doi: 10.1016/j.jbiomech.2011.08.003.
- Wilde, M.C., Castriotta, R.J., Lai, J.M., Atanasov, S., Masel, B.E., & Kuna, S.T. (2007). Cognitive impairment in patients with traumatic brain injury and obstructive sleep apnea. Arch Phys Med Rehabil, 88:1284-1288.
- Lettieri, C.J. (2013). Recognizing Obstructive Sleep Apnea in Patients With Traumatic Brain Injury. Medscape Pulmonary Medicine. Retrieved from: http://www.medscape.com/viewarticle/813907
- Gozal, D., Kheirandish-Gozal, L. (2009). Obesity and excessive daytime sleepiness in prepubertal children with obstructive sleepapnea. Pediatrics,123(1):13-8. doi: 10.1542/peds.2008-0228.
- Gozal, D., Wang, M., Pope, D.W. Jr. (2001). Objective sleepiness measures in pediatric obstructive sleep apnea. Pediatrics, 108:693–7.
- Chervin, R.D., Dillon, J.E., Bassetti, C., Ganoczy, D.A., &Pituch, K.J. (1997). Symptoms of sleep disorders, inattention, and hyperactivity in children.Sleep, 20(12):1185-92.
- Sung, V.,Hiscock, H., Sciberras, E.,&Efron, D. (2008). Sleep problems in children with attention-deficit/hyperactivity disorder: prevalence and the effect on the child and family. Arch Pediatr Adolesc Med., 162(4):336-342. doi:10.1001/archpedi.162.4.336
- Yürümez, E., &Kılıç, B.G. (2016). Relationship Between Sleep Problems and Quality of Life in Children With ADHD.J Atten Disord., 20(1):34-40. doi: 10.1177/1087054713479666.
- Huang, Y.S., Guilleminault, C., Li, H.Y., Yang, C.M., Wu, Y.Y., &Chen, N.H. (2007). Attention-deficit/hyperactivity disorder with obstructive sleep apnea: a treatment outcome study. Sleep, 8(1):18-30. doi: 10.1016/j.sleep.2006.05.016
- Silvestri, R., Gagliano, A., Aricò, I., Calarese, T., Cedro, C., Bruni, O.,….Bramanti P. (2009). Sleep disorders in children with Attention-Deficit/Hyperactivity Disorder recorded overnight by video-polysomnography. Sleep, 10(10):1132-8. doi: 10.1016/j.sleep.2009.04.003.
- Sadeh, A., Pergamin, L., &Bar-Haim, Y. (2006). Sleep in children with attention-deficit hyperactivity disorder: a meta-analysis of polysomnographic studies. Sleep Med Rev., 10(6):381-98. doi:1016/j.smrv.2006.03.004
- Crabtree, V.M., Ivanenko, A., O’Brien, L.M., &Gozal, D. (2003). Periodic limb movement disorder of sleep in children. J Sleep, 12(1):73-81.
- Al-Alawi, A., Mulgrew, A., Tench, E., Ryan, C.F. (2006).Prevalence, risk factors and impact on daytime sleepiness and hypertension of periodic leg movements with arousals in patients with obstructive sleep J Clin Sleep Med., 2(3):281-7.
- Chervin, R.D., Chung, S., O’Brien, L.M., Hoban, T.F., Garetz, S.L., Ruzicka, D.L., …Dillon JE. (2014).Periodic leg movements during sleep in children scheduled for adenotonsillectomy: frequency, persistence, and impact. Sleep, 15(11):1362-9. doi: 10.1016/j.sleep.2014.05.004.
- Sieminski, M., Pyrzowski, J., Partinen, M. (2017).Periodic limb movements in sleep are followed by increases in EEG activity, blood pressure, and heart rate during sleep. Sleep Breath. doi: 10.1007/s11325-017-1476-7.
- Tachibana, M., Kato, T., Kato-Nishimura, K., Matsuzawa, S., Mohri, I., &Taniike, M. (2016). Associations of sleepbruxism with age, sleep apnea, and daytime problematic behaviors in children. Oral Dis., 22(6):557-65. doi: 10.1111/odi.12492.
- Lam, M.H., Zhang, J., Li, A.M., Wing, Y.K. (2011). A community study of sleepbruxism in Hong Kong children: association with comorbid sleep disorders and neurobehavioral consequences.Sleep, 12(7):641-5. doi: 10.1016/j.sleep.2010.11.013.
- Ferreira, N.M., Dos Santos, J.F., dos Santos, M.B., &Marchini, L. (2015). Sleep bruxism associated with obstructive sleep apnea syndrome in children., 33(4):251-5. doi: 10.1080/08869634.2015.1097299.
- Drumond, C.L., Souza, D.S., Serra-Negra, J.M., Marques, L.S., Ramos-Jorge, M.L., &Ramos-Jorge, J. (2017). Respiratory disorders and the prevalence of sleep bruxism among schoolchildren aged 8 to 11 years. Sleep Breath. doi: 10.1007/s11325-017-1466-9.
- Winck, M., Drummond, M., Viana, P., Pinho, J.C., &Winck, J.C. (2016). Sleep bruxism associated with obstructive sleep apnoea syndrome – A pilot study using a new portable device. Rev Port Pneumol., 23(1):22-26. doi: 10.1016/j.rppnen.2016.07.001.
- Oksenberg, A., &Arons, E. (2002). Sleep bruxism related to obstructive sleep apnea: the effect of continuous positive airway pressure. Sleep Med. Nov;3(6):513-5.
- Durán-Cantolla, J., Alkhraisat, M.H., Martínez-Null, C., Aguirre, J.J., Guinea, E.R., &Anitua, E. (2015). Frequency of obstructive sleep apnea syndrome in dental patients with tooth wear. J Clin Sleep Med.11(4):445-50. doi: 10.5664/jcsm.4602.
- Szelenberger W, Niemcewicz S, &Dabrowska AJ. (2005). Sleepwalking and night terrors: psychopathological and psychophysiological correlates. Int Rev Psychiatry,17(4):263-70.
- Guilleminault C, Palombini L, Pelayo R, &Chervin RD. (2003). Sleepwalking and sleep terrors in prepubertal children: what triggers them? Pediatrics,111(1):e17-25.
- Zandbergen, J., Lousberg, H.H., Pols, H., de Loof, C., &Griez, E.J. (1990). Hypercarbia versus hypocarbia in panic disorder. J Affect Disord., 18(2):75-81.
- Klein, D.F. (1993). False suffocation alarms, spontaneous panics, and related conditions. An integrative hypothesis. Arch Gen Psychiatry,50(4):306-17.
- Taylor, , &Rachman, S. (1994). Klein’s Suffocation Theory of Panic. Arch Gen Psychiatry, 51(6):505-506. doi:10.1001/archpsyc.1994.03950060069011
- Manfredini D, Guarda-Nardini L, Marchese-Ragona R, &Lobbezoo F. (2015). Theories on possible temporal relationships between sleep bruxism and obstructive sleep apnea events. An expert opinion.Sleep Breath,19(4):1459-65. doi: 10.1007/s11325-015-1163-5.
- Lavie, P., Gertner, R., Zomer, J., &Podoshin L. (1981). Breathing disorders in sleep associated with “microarousals” in patients with allergic rhinitis. Acta Otolaryngol., 92(5-6):529-33.
- Agarwal, R. (2005). Automatic detection of micro-arousals. Conf Proc IEEE Eng Med Biol Soc., 2:1158-61. DOI: 1109/IEMBS.2005.1616628
- Huynh, N., Kato, T., Rompré, P.H., Okura, K., Saber, M., Lanfranchi, P.A., Montplaisir, J.Y., &Lavigne, G.J. (2006). Sleep bruxism is associated to micro-arousals and an increase in cardiac sympathetic activity. J Sleep Res., 15(3):339-46. DOI: 1111/j.1365-2869.2006.00536.x
- Lavigne, G.J., Huynh, N., Kato, T., Okura, K., Adachi, K., Yao, D., &Sessle, B. (2007). Genesis of sleep bruxism: motor and autonomic-cardiac interactions. Arch Oral Biol., 52(4):381-4. Epub 2007 Feb 20. DOI: 1016/j.archoralbio.2006.11.017
- Bunzel, B., Schmidl-Mohl, B., Grundböck, A., &Wollenek, G. (1992). Does changing the heart mean changing personality? A retrospective inquiry on 47 heart transplant patients. Qual Life Res., 1(4):251-6.
- Inspector, Y., Kutz, I., &David, D. (2004). Another person’s heart: magical and rational thinking in the psychological adaptation to heart transplantation. Isr J Psychiatry Relat. Sci., 41(3):161-73.
- McCraty, R. (2002). Heart Rhythm Coherence – An Emerging Area of Biofeedback. Biofeedback, 30(1):23-25.
- McCraty, R., Tiller, W. A., &Atkinson, M. (1996).Head-Heart Entrainment: A Preliminary Survey. Paper presented at the Key West Brain-Mind, Applied Neurophysiology, EEG Biofeedback 4th Annual Advanced Colloquium, Key West, FL. Retrieved from https://www.heartmath.org/assets/uploads/2015/03/head-heart-entrainment.pdf
- Felitti, V.J., Anda, R.F., Nordenberg,, Williamson, D.F. , Spitz, A.M., Edwards, V. , Koss, M.P., &Marks, J.S. (1998). Relationship of Childhood Abuse and Household Dysfunction to Many of the Leading Causes of Death in Adults. The Adverse Childhood Experiences (ACE) Study. American Journal of Preventive Medicine in, 14(245–258). DOI: http://dx.doi.org/10.1016/S0749-3797(98)00017-8





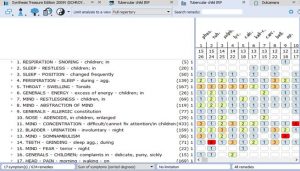
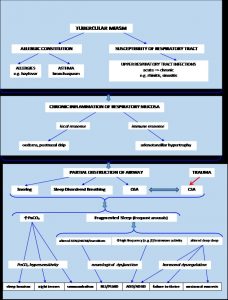
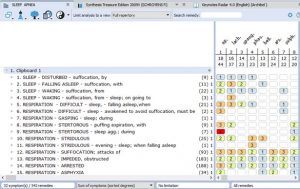

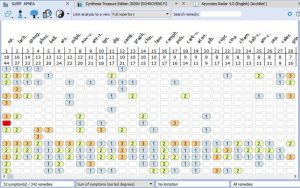

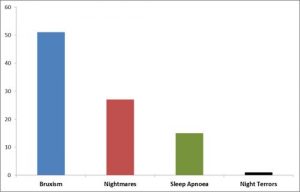


Wonderfully comprehensive survey of sleep apnoea. A valuable reference.
I am so glad that I was alerted to this valuable overview of sleep apnea, different types and causations, and differential description of remedies. I was not at all aware of the connection between sleep apnea and trauma, nor did I know how the condition commonly manifests in children. I will refer back to this paper many times in the future when treating children with sleeping and breathing difficulties.
Thank you for this in-depth and extremely well researched article. I feel it to be an excellent resource.