The Oncologist –Clinical Trial Results – Open Access
Michael Frass
Peter Lechleitner
Christa Gründling
Claudia Pirker
Erwin Grasmuk‐Siegl
Julian Domayer
Maximilian Hochmair
Katharina Gaertner
Cornelia Duscheck
Ilse Muchitsch
Christine Marosi
Michael Schumacher
Sabine Zöchbauer‐Müller
Raj K. Manchanda
Andrea Schrott
Otto Burghuber
-
- Volume 26Issue 3The Oncologist
- pages: e523-e523
- First Published online: March 3, 2021
Abstract
Lessons Learned
- Conventional medicine and homeopathy work well together.
- Quality of life improves with additive homeopathy in patients with non‐small cell lung cancer (NSCLC).
- Survival improves with additive homeopathy in patients with NSCLC.
Background
Patients with advanced non‐small cell lung cancer (NSCLC) have limited treatment options. Alongside conventional anticancer treatment, additive homeopathy might help to alleviate side effects of conventional therapy. The aim of the present study was to investigate whether additive homeopathy might influence quality of life (QoL) and survival in patients with NSCLC.
Methods
In this prospective, randomized, placebo‐controlled, double‐blind, three‐arm, multicenter, phase III study, we evaluated the possible effects of additive homeopathic treatment compared with placebo in patients with stage IV NSCLC, with respect to QoL in the two randomized groups and survival time in all three groups. Treated patients visited the outpatients’ centers every 9 weeks: 150 patients with stage IV NSCLC were included in the study; 98 received either individualized homeopathic remedies (n = 51) or placebo (n = 47) in a double‐blinded fashion; and 52 control patients without any homeopathic treatment were observed for survival only. The constituents of the different homeopathic remedies were mainly of plant, mineral, or animal origin. The remedies were manufactured by stepwise dilution and succussion, thereby preparing stable Good Manufacturing Practice grade formulations.
Results
QoL as well as functional and symptom scales showed significant improvement in the homeopathy group when compared with placebo after 9 and 18 weeks of homeopathic treatment (p < .001). Median survival time was significantly longer in the homeopathy group (435 days) versus placebo (257 days; p = .010) as well as versus control (228 days; p < .001). Survival rate in the homeopathy group differed significantly from placebo (p = .020) and from control (p < .001).
Conclusion
QoL improved significantly in the homeopathy group compared with placebo. In addition, survival was significantly longer in the homeopathy group versus placebo and control. A higher QoL might have contributed to the prolonged survival. The study suggests that homeopathy positively influences not only QoL but also survival. Further studies including other tumor entities are warranted.
Discussion
Additive homeopathy significantly improved QoL and survival when compared with placebo and control (Fig. 1). A higher QoL might have contributed to the prolonged survival. The results of this study suggest that homeopathy positively influences both QoL and survival. Further studies including other tumor entities are warranted.
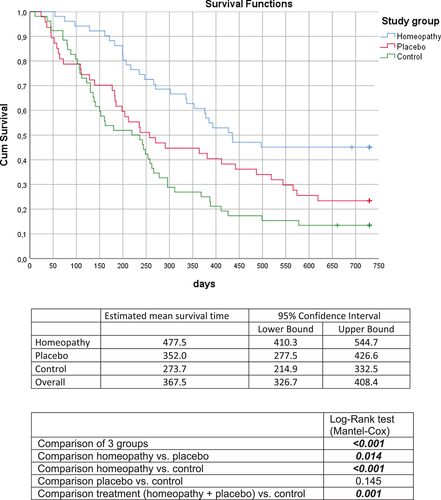
Figure 1
Open in figure viewerPowerPoint
Kaplan‐Meier estimates of overall survival time in the three groups. Crosses indicate time points of censoring (i.e., no patients died after this time point in this study group). Bold italicized values are statistically significant.
Trial Information
| Disease | Advanced cancer/solid tumor only |
| Disease | Lung cancer – NSCLC |
| Stage of Disease/Treatment | Metastatic/advanced |
| Prior Therapy | No Prior Therapy |
| Type of Study | Phase III, randomized |
| Primary Endpoint | Quality of life |
| Secondary Endpoint | Overall Survival |
|
Additional Details of Endpoints or Study Design |
|
| Sample size calculation was based on a significance level of 5% and a median survival of 10.1 months [26]. Furthermore, a 60‐month recruitment period with a 24‐month observational period in each patient was planned. Under these assumptions, 300 patients (corresponding to an average accrual rate of five patients per month) gave 85% power to detect a difference of 10.1 versus 14.5 months. Because the trial duration was quite long, a two‐stage design (O’Brien‐Fleming type with equal information rates) with an interim analysis was planned using the above assumptions (Addplan, version 6.0.8). An interim analysis with nonbinding stopping for futility option was projected after the observation of 140 events. Early rejection of the null hypothesis at interim was planned to be tested at a two‐sided significance level of .0052; the null hypotheses were accepted at interim (stopping for futility) if the p value exceeded .5. The two‐sided significance level for the second stage was .048. | |
| Accrual: After 5 years, enrollment of patients was terminated because the proposed time frame for recruitment and the proposed number for an interim analysis was reached. Baseline patient characteristics are shown in Table 1. From February 2012 to July 2017, 158 White patients with NSCLC stage IV were enrolled in the study, 52 of whom did not consent to participate in the randomized experiment but agreed to observation of their course of disease as controls with no participation in any homeopathic intervention (Fig. 2). The remaining 106 patients were randomized. Eight of the randomized patients had to be excluded because of sensitizing EGFR mutations or ALK translocations reported immediately after randomization (four in the homeopathy group, four in the placebo group). This left 98 patients in total in the two double‐blind groups. Each patient was observed for 24 months independently of the date of his or her study inclusion. Data were sent blinded to the statistician. Tables 2 and 3 show the results of the patient‐completed questionnaires. Table 4 shows the 24‐month mortality in each group. | |
| Investigator’s Analysis | Active and should be pursued further |
Drug Information
| Generic/Working Name | Homeopathic medicinal products |
| Trade Name | Several medicinal products were used in the study |
| Company Name | Maria Treu Pharmacy, Vienna, Austria |
| Drug Type | Homeopathic medicinal products |
| Drug Class | Schedule was individualized |
| Dose | Dosage was individually adjusted as drops or globules |
| Route | Oral (po) |
| Schedule of Administration | |
| Schedule was individually adjusted. Dilutions were taken daily on a 3‐week interval. Homeopathic medicinal products are detailed in Tables 6 and 7. | |
| Treatment Course: Ninety‐eight patients received allocated treatment (51 in the homeopathy group and 47 in the placebo group). Fifty‐two control patients were observed regarding the course of disease without any homeopathic intervention. Altogether, 150 patients were investigated. Seventy‐nine patients (46 patients in the homeopathy group and 33 patients in the placebo group) attended the third visit. All patients were treated as intended (Fig. 2). Figure 3 shows the CONSORT diagram. | |
| There were no statistically significant differences between the groups with respect to gender distribution, age, age groups, Karnofsky index, smoking status, T‐staging, M‐staging, stage groups (IIIB, IIIC, IV), radiotherapy, brain or liver metastases, whole brain radiation therapy, surgery, pneumonectomy, chemotherapy cycles, change to carboplatin, and immuno‐oncological therapy (Table 1). There was a statistically significant difference between treatment groups and the control group with regard to N stage with a higher number of patients with N stages 0 and 1 in the control group and more patients with N stage 3 in the treatment groups (p = .010); however, no difference between the two treatment groups was found. | |
| Histology revealed adenocarcinoma in 39 patients in the homeopathy group and 40 patients in the placebo group, squamous cell carcinoma in 10 patients in the homeopathy group and 4 patients in the placebo group, lung carcinoma not otherwise specified in 1 patient in the homeopathy group and 3 patients in the placebo group, and large‐cell bronchial carcinoma in 1 patient in the homeopathy group and none in the placebo group (p = .219; Table 1). There was also no significant difference from the control group (p = .421). | |
Patient Characteristics
| Number of Patients, Male | 81 |
| Number of Patients, Female | 69 |
| Stage | NSCLC stage IIIB, IIIC and IV |
| Age | Mean (range): 63.2 (33–87) years |
| Number of Prior Systemic Therapies | 0 |
| Karnofsky Index | 85.6 (15.7) |
| Cancer Types or Histologic Subtypes | Adenocarcinoma, 117; squamous cell carcinoma, 27; lung carcinoma not otherwise specified, 5; large‐cell carcinoma, 1 |
Primary Assessment Method: Experimental
| Title | Overall Survival |
| Number of Patients Screened | 55 |
| Number of Patients Enrolled | 51 |
| Number of Patients Evaluated for Efficacy | 51 |
| Evaluation Method | Estimated mean survival time |
| (Median) Duration Assessments OS | 435 |
| Outcome Notes | |
| QoL as well as functional and symptom scales showed significant improvement in the homeopathy group when compared with placebo after 9 and 18 weeks of homeopathic treatment (p < .001; Table 2). Median survival time was significantly longer in the homeopathy group (435 days) versus placebo (257 days; p = .010) as well as versus control (228 days; p < .001; Table 4). Survival rate in the homeopathy group differed significantly from placebo (p = .020) and from control (p < .001). | |
Primary Assessment Method: Placebo
| Title | Overall Survival |
| Number of Patients Screened | 51 |
| Number of Patients Enrolled | 47 |
| Number of Patients Evaluated for Efficacy | 47 |
| Evaluation Method | Estimated mean survival time |
| (Median) Duration Assessments OS | 257 |
Primary Assessment Method: Control
| Title | Overall Survival |
| Number of Patients Screened | 52 |
| Number of Patients Enrolled | 52 |
| Number of Patients Evaluated for Efficacy | 52 |
| Evaluation Method | Estimated mean survival time |
| (Median) Duration Assessments OS | 228 |
Assessment, Analysis, and Discussion
| Completion | Study completed |
| Investigator’s Assessment | Active and should be pursued further |
Lung cancer is the second most common cancer in men and women, as well as the leading cause of cancer‐related mortality in the U.S. [1], accounting for 29% of all cancer‐related mortalities in men and 26% of those in women [2]. More than 85% of lung cancers are non‐small cell lung cancer (NSCLC) [3], for which surgery is the preferred therapy in the early stages. Unfortunately, most patients are diagnosed at stages III or IV, by which time NSCLC is inoperable [4]. Chemotherapy is the standard treatment for unresectable NSCLC [5], but its adverse reactions frequently prevent completion of the recommended number of cycles [6]. Additional approaches to reduce chemotherapy’s toxicity and enhance its clinical efficacy are, therefore, warranted.
Some 90% of patients with advanced NSCLC experience two or more disease‐related symptoms, including pulmonary complaints, such as cough and dyspnea, and general fatigue, pain, and anorexia [7], which can significantly impact the emotional, social, physical, and spiritual well‐being of patients, as well as their functioning [8-12]. One survey found that 68% of patients preferred treatment that eased disease‐related symptoms without prolonging their life [13]. Moreover, shorter overall survival has been associated with poor health‐related quality of life at diagnosis and higher symptom burden at the outset of treatment [14, 15]. There is growing understanding of the extent to which mind and body are connected and awareness that psychosocial characteristics and variables can contribute to both the symptom experiences and to patient outcomes, including survival [16].
Homeopathy is one of the most popular forms of complementary and alternative medicine. It is rooted in two theories: one, that “like cures like”—that is, disease can be cured by a substance that produces similar symptoms in healthy people; and, two, “the law of minimum dose”—the lower the dosage the more effective the medication [17, 18]. A detailed clinical history is recorded by the homeopath, relying on the totality of symptoms described by the patient. The symptoms are then listed and repertorized, which means that the homeopath matches the complete symptom profile of the patient to the symptom profile of the remedy. Then, the homeopath determines the least amount of medicine needed to achieve the desired effect. The medications are not only diluted but also succussed, thereby enhancing the effect of the remedies on the patient.
Homeopathy is a system in which remedies are customized to individuals, based on broad themes characteristics elicited from the totality of the presenting physical, mental, and emotional symptoms [17, 18]. Because homeopathy sees disease as unique to each patient and thus treats it with a unique, specifically tailored medication, it is difficult to perform randomized, controlled trials—a method suited to research on groups rather than individuals.
We published the results of a pragmatic, randomized, controlled trial of 410 cancer patients in which homeopathic treatment was an add‐on therapy to conventional oncological treatment [19]. In this trial, the global health status and subjective well‐being of these patients improved significantly with individualized homeopathic treatment. An oncologist at our medical center noticed that cancer patients treated with adjunct homeopathy seemed to have an improved survival rate. A retrospective evaluation found that homeopathic therapy administered to patients with advanced cancer seemed to provide a statistically significant advantage (p < .001) for survival, compared with control [20].
We decided, therefore, to validate the results of our pragmatic open trial regarding quality of life (QoL) and subjective well‐being, under double‐blind conditions in patients with advanced NSCLC [19]. We also wanted to evaluate whether survival is impacted by homeopathic treatment in patients with stage IV NSCLC in a prospective, randomized, placebo‐controlled, double‐blind, study. Since the beneficial effects of homeopathy are often attributed to a placebo effect, we added a third group of patients, who received no add‐on homeopathic treatment, as a noninterventional control group. Our hypothesis was that there is no significant difference between homeopathy and placebo regarding QoL and survival.
Material and Methods
This phase III study, which examined homeopathic treatment as an add‐on therapy to conventional treatment, was prospective, three‐arm, randomized, double‐blind, placebo‐controlled, stratified, and multicenter; neither the patients nor the managing physicians knew the specific study substances during the study period until the end of the statistical analysis.
The three arms evaluated were additive homeopathy, placebo, and control. Two parallel groups randomized to homeopathy or placebo were compared in a double‐blind fashion. Controls (third group) were patients who refused participation in the randomized trial, but agreed to observation of their course of disease without any homeopathic intervention (Fig. 2). The study was conducted in four outpatients` centers: the Medical University of Vienna (General Hospital of Vienna), Department of Medicine I, Division of Oncology; the Otto Wagner Hospital, Department of Pulmonology I, Vienna; the Hospital of Lienz, Department of Medicine, Tyrol; and the Elisabethinenspital, Department of Medicine, Linz, Austria.
The study protocol was approved by the ethics committees of the participating institutions (Ethical Committee of the Medical University of Vienna No. 709/2010). The study was conducted in accordance with the Declaration of Helsinki, Good Clinical Practice Guidelines, and applicable local regulations, and registered at Clinical Trials.gov (ClinicalTrials.gov Identifier: NCT01509612). All participating patients gave their written informed consent prior to study entry. The full trial protocol can be accessed by contacting the corresponding author.
Regarding eligibility, patients with histologically or cytologically confirmed stage IIIB, IIIC, or IV NSCLC diagnosed within the past 8 weeks were invited to participate in the study. The date of the printed histology/cytology finding was determined as date of diagnosis. Patients were randomly assigned at a ratio of 1:1 to either classical individualized homeopathic treatment or placebo as add‐ons to conventional treatment. Patients who chose not to participate in the study served as a no‐add‐on control group after signing informed consent [21]. This control group was formed to exclude a possible placebo effect of the homeopaths. No homeopathic case histories were taken from them, and they received no treatment in addition to conventional therapy. Patients who refused to be randomized and opted to undergo homeopathic treatment were not considered any further in this study (Fig. 2).
Inclusion criteria were (a) histologically or cytologically confirmed stage IIIB/IIIC or IV NSCLC within the preceding 8 weeks and (b) aged older than 18 years.
Exclusion criteria were numerous: (a) sensitizing mutation of the EGFR gene or translocation of the ALK gene; (b) refusal to sign informed consent; (c) pregnancy; (d) hematological, hepatic, or renal pathology; (e) coronary heart disease; (f) history of secondary tumor; (g) major surgery within 4 weeks prior to study entry; (h) active infection, and symptomatic peripheral neuropathy (National Cancer Institute’s common toxicity criteria, version 2, grade ≥2); (i) central nervous system metastases unless the metastases were treated and stable; (j) active autoimmune disease; (k) use of systemic immunosuppressive treatment; (l) use of systemic treatment during the previous 2 years; (m) active interstitial lung disease, or a history of pneumonitis for which glucocorticoids were prescribed; (n) previous systemic therapy for metastatic disease or previous irradiation; and (o) use of any complementary and/or alternative therapy, including homeopathy other than the research treatment, during the trial.
Chemotherapy
Cisplatin plus gemcitabine or cisplatin plus pemetrexed was the standard regimen for first‐line treatment of advanced squamous or non‐squamous NSCLC at the start of our study [22]. Chemotherapy consisted of cisplatin 80 mg/m2 or carboplatin 5 mg/ml*minute given intravenously on day 1 combined with either gemcitabine 1,000 mg/m2 given intravenously on day 1 and day 8 or with pemetrexed 500 mg/m2 given intravenously on day 1 every 3‐week cycle for up to six cycles.
Homeopathic Medicinal Products
Homeopathy uses a wide variety of source materials, which means that various methods of preparation are necessary depending on the substance being processed. Homeopathic medicines are derived from plants, herbs, minerals, or animal products [23].
All study medications (D, CH, LM, and Q‐Potencies) were prepared (Maria Treu Pharmacy, Vienna, Austria) in accordance with the current version of the European Pharmacopoeia (Ph.Eur.), the German Homoeopathic Pharmacopoeia (GHP), and the Q‐potencies according to Hahnemann’s 6th edition of the Organon of Medicine [17]. In brief, the constituents of the homeopathic medicinal products (HMP) are mainly plants, minerals, or of animal origin. The HMPs are manufactured by stepwise dilution and succussion, thereby preparing stable GMP‐grade formulations.
Homeopathic medicines were produced through sequential agitated dilutions in alcohol/water or by trituration in powdered lactose in decimal (1:10), centesimal (1:100) or quinquaginta millesimal (1:50,000) potencies (Q‐potencies) / LM‐ potencies (Table 5) [23].
All homeopathic therapies started with Q1 potencies of the selected remedies for 3 weeks, and continued in ascending order with Q2, Q3, of either the same remedy or a selected alternative (3 weeks each) toward Q30. Where the study substance was changed, whatever the reason, the new cycle started from the beginning with Q1. A primary reason for changing the study substance was disease deterioration.
The Q‐potencies were applied as liquids, after being shaken and diluted daily by the patient (Table 5). The package leaflet also mentions the number of succussions to be carried out by the patient every day and the number of drops or spoonfuls to be taken [23]. They were used in the trial as constitutional medication based on the mental, emotional, and physical symptoms displayed by any patient. The D‐, C‐, and LM‐potencies were applied as sugar granules. Five pellets comprised one dose, which was taken orally by sucking.
Treatment Protocol
Patients newly diagnosed as suffering from a NSCLC by pulmonal oncologists who did not participate in the study, were invited to participate in the study. At study entry, all eligible patients (those who met all the inclusion and none of the exclusion criteria, and had signed informed consent) were required to complete a series of questionnaires ‐ the EORTC QLQ‐C30 [24], the SF‐36 [25], the Subjective Well‐Being Questionnaire, and one which gauged their attitudes toward homeopathy and CAM. All questionnaires were given and explained to patients and collected from them after completion by individuals uninvolved in the study. Safety was assessed by evaluating the incidence of adverse clinical events and laboratory variables, graded according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.0. Patients in the third non‐randomized, untreated group were compared with the two treatment groups solely with respect to overall survival, and therefore did not complete questionnaires. Demographic information was collected, after which a thorough and detailed homeopathic medical history was taken for all patients [19]. Based on these homeopathic case histories, homeopathic physicians determined the most appropriate constitutional and symptomatic remedies for each individual following the classical principles of homeopathy of individualization.
Constitutional remedies were largely tailored for mental, emotional and general symptoms, regardless of reactions to the anti‐cancer treatment. Symptomatic remedies were given to combat the adverse effects of this treatment. The homeopathic physician explained the patient how to prepare and how to take the prescribed homeopathic Q‐potencies after vigorous succussion, dilution and stirring on a daily basis.
The name and all other details of prescribed medications were faxed to the pharmacy (Maria Treu Pharmacy), where both homeopathic remedies and placebo for the study were prepared.
Method Used to Generate the Random Allocation Sequence
Subjects were randomized for permuted blocks randomization in a 1:1 ratio by a web‐based randomization service (Randomizer, Medical University of Graz, Austria), stratified by age, gender, Karnofsky performance status, and treatment center. Randomization for homeopathy and placebo medication was carried out at the pharmacy by a person not involved in the preparation of the remedies on receipt of the medication name determined and faxed by the physician for each individual patient. Investigators and patients were blinded to treatment allocation until study completion and finish of data analyses.
Following randomization, a pharmacist prepared the prescribed study substances (placebo/homeopathic medicine) for the respective patient. Homeopathic medicine and placebo showed identical appearance. To avoid contact between pharmacist and patients and, thus, any information bias, medications were sent to patients by regular mail together with plastic cups and spoons in neutral wrapping. The study substances were labeled with a code held at the pharmacy not visible to the homeopathic physicians. This design was set up to maintain the double‐blind design of the study; neither the treating physician nor the patient knew which treatment he/she received.
The pharmacy was responsible for both preparing the study medications and dispatching them to patients via regular mail in neutral wrapping. Names of remedies were kept from patients to maintain double‐blind design and to obviate any attempt to avoid the placebo.
Patients were followed up every 9 weeks until death. At each visit, the homeopathic physician evaluated whether to continue with the same remedies or change them, based on patient reporting and routine cancer assessment. Patients were asked to complete again the questionnaires they answered on study entry [19]. The physician completed a case report form (CRF) at every visit. The duration of the first visit at the homeopath’s office was about 60 minutes; the follow‐up visits, which took place every 9 weeks, lasted for approximately 30 minutes. All patient data, including the questionnaires, were sent blinded to two people with no involvement in the study, using a double‐entry method to record data on the “Research, Documentation and Analysis” (RDA) platform of the Medical University of Vienna.
Statistical Analysis and Sample Size Calculation
Sample size calculation was based on a significance level of 5% and a median survival of 10.1 months [26]. Furthermore, a 60‐month recruitment period with a 24‐month observational period in each patient was planned. Under these assumptions, 300 patients (corresponding to an average accrual rate of 5 patients per month) gave 85% power to detect a difference of 10.1 versus 14.5 months. Because the trial duration was quite long, a two‐stage design (O’Brien‐Fleming type with equal information rates) with an interim analysis was planned using the above assumptions (Addplan, Version 6.0.8): An interim analysis with nonbinding stopping for futility option was projected after the observation of 140 events. Early rejection of the null hypothesis at interim was planned to be tested at a two‐sided significance level of .0052, the null hypotheses were accepted at interim (stopping for futility) if the p value exceeded .5. The two‐sided significance level for the second stage was .048. Maximum sample size was estimated to be 302 (corresponding to 279 events), expected (average) number of events was 209 under the null hypothesis and 242 under the alternative.
Efficacy was assessed in the intention‐to‐treat sample, which included all randomized patients. Safety was assessed in the as‐treated sample, which included all randomized patients who had received at least one dose of the assigned therapy. Randomized patients were prescribed at least three Q‐potencies of the assigned homeopathic therapy.
IBM SPSS statistics 26.0 was used for all analyses, α = 5% (two‐sided). Frequencies (n) and valid percentage were used for reporting dichotomous and categorical data; minimum and maximum (range), mean and standard deviation for continuous variables. Group comparisons for 2×2 crosstabs were calculated via Fisher’s exact test, those for larger crosstabs via χ2‐test. Univariate comparisons of two group means were done with t‐test for homogenous respectively heterogenous variances (homogeneity tested by Levene’s test), comparisons of two group medians with Mann‐Whitney‐U‐tests, univariate comparisons of three group means by analyses of variances (ANOVA) and prior testing of homogeneity of variances and co‐variances (Levene test) and pairwise post hoc Scheffé tests. Multivariate comparison of means for multiple assessment scales of psychological tests was done via General Linear Model (multivariate analyses of variances with preceding test for homogeneity of variances and covariances via Box‐M‐Test) respectively via General logistic model for repeated measurements (with preceding test for homogeneity of variances and covariances via Box‐M‐Test test estimation: Wilk’s λ). Kaplan‐Meier curves were used to graphically display the survival comparison between the groups, Log‐Rank‐test (Mantel‐Cox; two‐sided) was used to assess group differences in survival, estimates of mean survival time in days (hazard ratios) and 95% CIs are given overall and as well for each study group. Survival rates for study groups are given in %, Wilcoxon (Gehan) Statistic is used for overall and pairwise comparison of rates.
The obtained and recorded raw data from this three‐arm trial were sent blinded to the statistician and used to compare between the two randomized groups with regard to the following outcomes:
- Primary outcome: QoL as evaluated as global health status and subjective well‐being at 18 weeks (third visit after second prescription) versus base line (EORTC‐QLQ‐C30 remaining dimensions; SF‐36; subjective well‐being) in the two treatment groups using the EORTC QLQ‐C30‐scoring manual.
- Secondary outcome: overall survival time.
- All three groups were compared overall and pair‐wise to each other with respect to overall survival. After statistical analysis, data were unblinded.
We used the CONSORT checklist by Schulz KF et al. for the CONSORT Group 2010 Statement: updated guidelines for reporting parallel group randomized trials, which can be found at https://www.jclinepi.com/article/S0895-4356(10)00079-X/fulltext.
Results
Lists of prescribed homeopathic remedies Q‐, LM‐, C‐ and D‐potencies are shown in Tables 6 and 7. There was no difference between homeopathy and placebo groups with respect to selection of homeopathic remedies or change of Q‐potencies between the two treatment groups (change of Q‐potencies: 11 times (21.6%) in homeopathy patients, 14 times (29.8%) in placebo patients, p = .242).
Patients’ Use of Alternative Treatments (Table 8)
Regarding previous alternative treatments, no statistically significant difference could be found between the two treatment groups. Psychotherapy was the most used alternative method in all groups.
Questionnaire EORTC QLQ‐C30 Data (Table 2)
The changes in global health status, functional scales, and symptom scales at baseline, 9 and 18 weeks (visits 1, 2 and 3) for patients in the treatment groups are summarized in Table 2.
At baseline, there were no significant differences between the treatment groups found apart from constipation (p = .048), which was more common in the homeopathy group. At 9 and 18 weeks, the global health status of the verum group was significantly higher than that of the placebo group (p < .001). According to all the functional scales, apart from cognitive functioning (no difference between groups after 9 weeks), patients in the homeopathy group at both follow‐up visits were significantly more functional than patients in the placebo group, both by univariate analysis of the individual functional scales and by multivariate consideration of all functional scales. Multivariate analysis (general logistic model for repeated measurements, test size: Wilk’s λ) revealed a significant time effect (p < .001) for the functional scales, i.e., the mean scale values change between the survey times, with a significant difference both between baseline and 9 weeks and between baseline and 18 weeks (p < .001 and p < .001, respectively), and a significant group effect (<.001), but additionally a significant interaction between time and group (<.001), caused by an improvement in homeopathic group and worsening in the placebo group.
After 9 weeks, all symptom scales except pain, diarrhea, and financial difficulties scores were significantly lower (i.e., less symptom burden) in the homeopathy group than in the placebo group by both univariate analysis of the individual symptom scales and by multivariate analysis of all symptom scales. After 18 weeks, scores were significantly lower for all symptom scales in the homeopathy group than in the placebo group by both univariate and multivariate analysis. There was also an improvement between visit 1 and visit 2 and between visit 2 and 3 within the homeopathy group, but not in the placebo group. Multivariate analysis revealed a significant time effect both between baseline and 9 weeks (p < .001) and baseline and 18 weeks (p < .001) and a significant group effect (p < .001), and also a significant interaction between time and group (p < .001).
The same effects could be seen for Global Health status, with a significant time effect (p < .001), group effect (p < .001), and interaction between time and group (p = .002).
Questionnaire SF‐36 (Table 3)
The changes in the indices of the SF‐36 questionnaire at baseline, 9 and 18 weeks for the treatment groups are summarized in Table 3.
Attitude Towards Homeopathy (Table 9)
No differences regarding attitude toward homeopathy were detected between the two treatment groups except for referral to previous homeopathic treatment and expectation regarding prognosis: while the majority of homeopathy patients (57.1%) had been referred by practitioners (only 17.6% of placebo patients), placebo patients significantly more often opted for homeopathic treatment themselves (47.1%, only 7.1% in homeopathy patients; p = .039). In the homeopathy group, the expectation of the effect of homeopathy on the prognosis was significantly more negative (p = .010). In the placebo group, patients perceived significantly more minor side effects such as nausea and diarrhea (p = .023).
Survival (Table 4)
The median survival time over the observation period of 730 days for the homeopathy group (435 days) was significantly longer than the placebo group (257 days; p = .010). Median survival of the control group (228 days) was significantly shorter than that of the homeopathy group (p < .001), but not than that of the placebo group (p = .258).
When comparing treatment groups (homeopathy plus placebo group) to control group, there was a significant difference with regard to the pairwise comparison of homeopathy versus placebo (treatment groups vs. control: p = .002).
Regarding patients who died within the 730‐day period, there was no significant difference in the median survival time between the homeopathy and the placebo groups (p = .172); however, the homeopathy group had a significantly longer survival time than the control group (p = .020). There was no significant difference between placebo and control groups (p = .747) or between the two treatment groups and the control group (p = .142).
Estimated survival time (hazard ratio for mean) was 477 (95% CI: 410–545) days in homeopathy group, 352 (95% CI: 278–427) days in placebo group and 274 (95% CI: 215–333) days in control group (p < .001 comparing all 3 groups; p = .014 homeopathy vs. placebo group; p < .001 homeopathy vs. control; p = .145 placebo vs. control group). Survival rate in the homeopathy group was 45.1%, in the placebo group was 23.4%, and in the control group was 13.5%. While survival rate in homeopathy group differed significantly from placebo (p = .020) and from control group (p < .001), the difference between placebo and control (p = .154) was not significant (overall difference was significant at p < .001).
An independent doctor evaluated the cause of death and assured that all patients who died did so as a direct consequence of their underlying cancer; none died because of other reasons such as myocardial infarction, pulmonary embolism, or other major diseases. No severe adverse effects were reported.
Discussion
Similar to our previous open randomized pragmatic study [19], in our present randomized placebo‐controlled, double‐blind study, additive homeopathic treatment in patients with cancer significantly improved global health status, subjective well‐being, and several functional and symptom scales, according to European Organisation for Research and Treatment of Cancer (EORTC) and 36‐Item Short Form Health Survey questionnaires [24, 25].
Unexpectedly, homeopathy also increased survival time. Median survival in the homeopathy group was about 6 months longer than in the placebo group and 7 months longer than in the control group. The median survival in the placebo group was about 8.5 months, close to the data reported in the literature [26]. Furthermore, median survival in patients who died during the observation period was significantly longer in the homeopathy group (8.4 months) than in the placebo group (6.4 months) and the control group (5.2 months). Overall survival was defined as the time from randomization until death or until 2 years later. Date of death was provided by the independent Information Technology Solution Center (ITSC), Austria, and recorded by an individual with no involvement in the study (E.E.).
There were slightly more patients with squamous cell carcinoma in the homeopathy group, but this difference was not significant. Because the prognosis of squamous cell carcinoma is worse than that of adenocarcinoma [27], this might support the results of our study.
The difference between the placebo and control group in survival time in our study is in accordance with the results of Temel et al. [28]. Temel examined the effect of introducing palliative care early after diagnosis on patient‐reported outcomes and end‐of‐life care among ambulatory patients with newly diagnosed disease. Patients with newly diagnosed metastatic NSCLC were randomly assigned to receive either early palliative care integrated with standard oncological care or standard oncological care alone. Despite the fact that fewer patients in the early palliative care group than in the standard care group received aggressive end‐of‐life care (33% vs. 54%, p = .05), median survival was longer among patients receiving early palliative care (11.6 months vs. 8.9 months, p = .02). The authors concluded that among patients with metastatic NSCLC, early palliative care led to significant improvements in both quality of life and mood. Compared with patients receiving standard care, patients receiving early palliative care had less aggressive care at the end of life but had longer survival [28].
Deng et al. reported that physicians are often asked about complementary therapies by patients with cancer, and data show that the interest in and use of these therapies among patients with cancer is common [29]. Therefore, it is important to assess the current evidence base on the benefits and risks of complementary therapies. Several complementary therapy modalities can be helpful in improving the overall care of patients with lung cancer. Placebo effects seem to be of minor influence because homeopathy also works in critically ill patients [30].
Quality of life (QoL) as well as the functional and symptom scales showed a significant improvement in the homeopathy group when compared with the placebo group after 9 and 18 weeks of homeopathic treatment (p < .001). In addition, results show that additive homeopathic treatment produced an overall survival benefit for patients with advanced NSCLC, with a significant reduction of 2‐year mortality from 86.5% in the control group to 76.6% in the placebo group and 54.9% in the homeopathy group. Estimated median survival time was significantly longer in the homeopathic group. These results coincide with the previously reported approximately 50% reduction in the risk of progression or death [20]. This is the first randomized trial to demonstrate a significant overall survival benefit for additive homeopathic therapy.
By including the noninterventional control group, it was possible to assess the real homeopathic effect on the homeopathic cohort, as the real effect will be the natural historical effect minus the placebo effect and the homeopathic effect.
A limitation of the study is the relative long study period. However, during the whole study period, basic conventional therapy remained essentially unchanged. A further limitation is that conventional therapy in this study was performed in almost all patients without the nowadays usual immuno‐oncologic therapy [31] because the study started before 2015. Although EGFR/ALK tyrosine kinase inhibitors have become standard first‐line strategy in patients with advanced, EGFR‐mutation–positive or ALK fusion oncogene–positive NSCLC with improved outcome, standard therapy for control patients suffering from NSCLC without mutations comprised conventional chemotherapy only during the study. The approvals of immune checkpoint inhibitors such as nivolumab, pembrolizumab, and atezolizumab for the second‐line therapy of NSCLC after platinum failure based on the CheckMate 017, CheckMate 057, KEYNOTE 010, and OAK trials might have had an influence on overall survival analysis of this trial. However, this potential bias is likely not relevant, because the number of patients treated with subsequent immuno‐oncological therapy was comparable (p = .942) between the groups (Table 1).
Today, immuno‐oncologic and chemotherapy are established as first‐line therapy. Therefore, further studies with immuno‐oncologic therapy are necessary to investigate the effect of homeopathic therapy with modern forms of therapy. A further limitation is that patients of the control group were not randomized because of patients’ preferences.
Costs for additive homeopathic remedies are very low, compared with immuno‐oncologic approaches. Thereby, additive expenses of homeopathic drugs are negligible in the context of anticancer treatment costs. From a methodological point of view, add‐on homeopathy provides a tool compatible with all other conventional interventions. The advantages are that there are no interactions with other methods and no burden to metabolism of the patients and that the cost is low.
Conclusion
Additive homeopathy significantly improved QoL and survival when compared with placebo and control. A higher QoL might have contributed to the prolonged survival [32].
The results of this study suggest that homeopathy positively influences both QoL and survival. Further studies including other tumor entities are warranted. Our study supports trials in other fields of complementary medicine such as acupuncture for chemotherapy‐induced peripheral neuropathy in breast cancer survivors [33].
Acknowledgments
This work was supported by the Institute for Homeopathic Research, Vienna, Austria.
We thank Dr. Menachem Oberbaum, Head of the Center for Integrative Complementary Medicine at the Shaare‐Zedek Medical Center, Jerusalem, Israel, for providing the impetus to perform this study. He also added significantly to the preparation of the paper and edited the submitted version. We also thank Prof. DDr.h.c. Christoph Zielinski, Head of the Division of Oncology, Department Medicine I, Medical University of Vienna; Prof. Dr. Reinhard Krepler, Director of the General Hospital of Vienna; and Prof. Dr. Wolfgang Schütz, Rector of the Medical University of Vienna, Austria, for establishing the Outpatient Unit “Homeopathy in malignant disease” in March 2004 and for encouraging us to perform this study. We thank Dr. Thorsten Füreder, Medical University of Vienna, Department of Medicine I, Clinical Division of Oncology, Vienna, Austria, for carefully revising the manuscript.
Abbreviations of Authors, Study Contributors, and Study Assistants in Alphabetical Order of Family Names
OB: Otto Burghuber, MD, Department of Respiratory and Critical Care Medicine and Ludwig Boltzmann Institute for Lung Health, Otto Wagner Hospital and Sigmund Freud University, Medical School, Vienna, Austria; JD: Julian Domayer, MD, Department of Respiratory and Critical Care Medicine, Respiratory Oncology Unit, Karl Landsteiner Institute of Lung Research and Pulmonary Oncology, Klinik Floridsdorf, Vienna, Austria; CD: Cornelia Duscheck MD, Hospital Lilienfeld, Lilienfeld, Austria; EE: Ernst Eigenbauer, degreed engineer, chief analyst, Informations Technology Solution Center (ITSC), Research, Documentation and Analysis (RDA), scientific platform, Center for Medical Statistics, Informatics, and Intelligent Systems, Medical University of Vienna; MF: Michael Frass, MD, Medical University of Vienna, Department of Medicine I, Clinical Division of Oncology, Vienna; Institute for Homeopathic Research, Vienna, Austria; MeF: Melanie Fraunschiel, degreed engineer, Research, Documentation and Analysis (RDA), scientific platform, Center for Medical Statistics, Informatics, and Intelligent Systems, Medical University of Vienna; TF: Thorsten Füreder, MD, Medical University of Vienna, Department of Medicine I, Division of Oncology, Vienna, Austria; KG: Katharina Gaertner, MD, Witten/Herdecke University, Faculty of Health Sciences Witten, Germany; AG: Andreas Gleiβ, PhD, Center for Medical Statistics, Informatics, and Intelligent Systems, Medical University of Vienna; EGS: Erwin Grasmuk‐Siegl E, MD, Department of Respiratory and Critical Care Medicine, Respiratory Oncology Unit, Karl Landsteiner Institute of Lung Research and Pulmonary Oncology, Klinik Floridsdorf, Vienna, Austria; CG: Christa Gründling, MD, General Practitioner, Enns; MH: Maximilian Hochmair, MD, Department of Respiratory and Critical Care Medicine, Respiratory Oncology Unit, Karl Landsteiner Institute of Lung Research and Pulmonary Oncology, Klinik Floridsdorf, Vienna, Austria; PL: Peter Lechleitner, MD, Hospital of Lienz, Department of Medicine, Lienz, Austria; RKM: Raj K. Manchanda, MD, Director General, Central Council for Research in Homoeopathy, Ministry of AYUSH, Govt. of India; Secretary Research, LMHI; West Delhi, Delhi, India; IM: Ilse Muchitsch, Mag. pharm., Austrian Chamber of Pharmacists, Department Vienna, HomResearch, Interdisciplinary Homeopathic Research Group, Vienna, Austria; BNB: Birgit Negedly‐Blauensteiner, MD, Traiskirchen, Austria; MO: Menachem Oberbaum, MD, Head of the Department of Complementary Medicine at the Shaare‐Zedek University, Jerusalem, Israel; CP: Claudia Pirker, MD, Department of Respiratory and Critical Care Medicine, Respiratory Oncology Unit, Karl Landsteiner Institute of Lung Research and Pulmonary Oncology, Klinik Floridsdorf, Vienna, Austria; RP: Robert Pirker, MD, Medical University of Vienna, Department of Medicine I, Division of Oncology, Vienna, Austria; WR: Wilhelm Raneburger, MD, Hospital of Lienz, Department of Medicine, Lienz, Austria; AS: Andrea Schrott, MSc, Dr, StatistikAmbulanz, Private Business, Vienna, Austria; MS: Michael Schumacher, MD, Elisabethinenspital, Department of Medicine, Linz, Austria; JS: Judith Stellnberger, Cand. med., Mag. phil., Medical University of Vienna, Department of Medicine I, Division of Oncology; KT: Karin Thieves, Research Fellow, Sola Salus, Geilenkirchen, Germany; PW: Petra Weiermayer, PhD, Veterinarian, Vienna, Austria, and Scientific Society for Homeopathy (WissHom), Koethen, Germany; TW: Thomas Wrba, PhD, Research, Documentation and Analysis (RDA), scientific platform, Center for Medical Statistics, Informatics, and Intelligent Systems, Medical University of Vienna; SZM: Sabine Zöchbauer‐Müller, MD, Medical University of Vienna, Department of Medicine I, Division of Oncology, Vienna, Austria.
Change of address: Department of Respiratory and Critical Care Medicine, Respiratory Oncology Unit, Karl Landsteiner Institute of Lung Research and Pulmonary Oncology, Klinik Floridsdorf, Vienna, Austria: former Otto Wagner Hospital, Department of Respiratory and Critical Care Medicine and Ludwig Boltzmann Institute of COPD and Respiratory Epidemiology, Vienna, Austria.
Disclosures
Sabine Zöchbauer‐Müller: Boehringer Ingelheim, Roche, AstraZeneca, Bristol Myers Squibb, Merck Sharp & Dohme, Amgen (H), Merck Sharpe & Dohme (RF). The other authors indicated no financial relationships.
(C/A) Consulting/advisory relationship; (RF) Research funding; (E) Employment; (ET) Expert testimony; (H) Honoraria received; (OI) Ownership interests; (IP) Intellectual property rights/inventor/patent holder; (SAB) Scientific advisory board
- ClinicalTrials.gov Identifier: NCT01509612
- Sponsor: Michael Frass
- Principal Investigator: Michael Frass
- IRB Approved: Yes
Click here to access other published clinical trials.
References
Figures and Tables
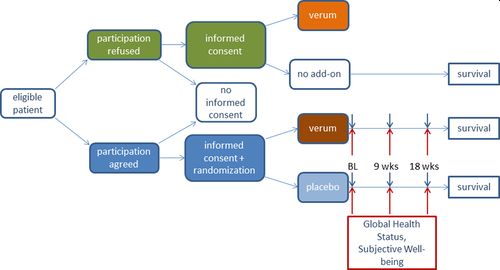
Figure 2
Open in figure viewerPowerPoint
Study protocol.
Abbreviations: BL, baseline; verum, homeopathy; wks, weeks.
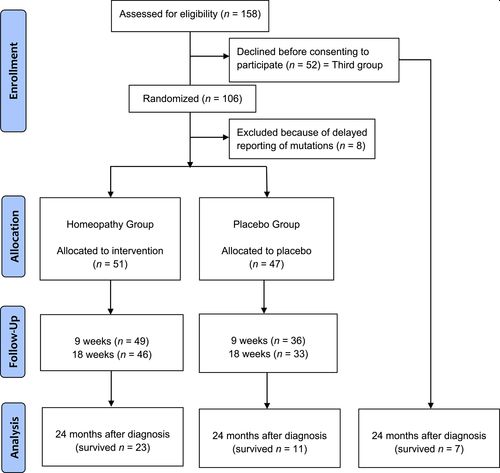
Figure 3
Open in figure viewerPowerPoint
CONSORT diagram.
Table 1. Baseline characteristics and treatment
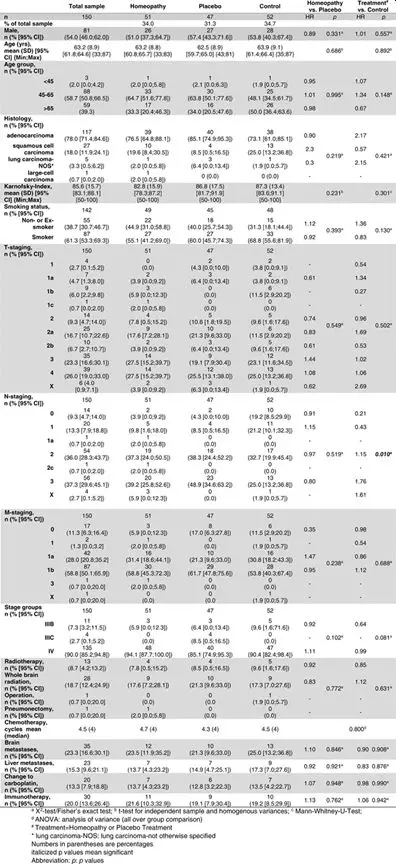
Table 2. Questionnaire EORTC QLQ‐C30
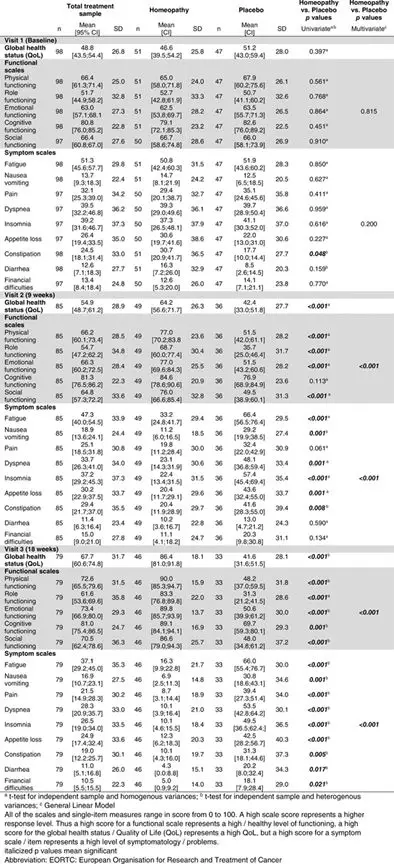
Table 3. Questionnaire SF‐36
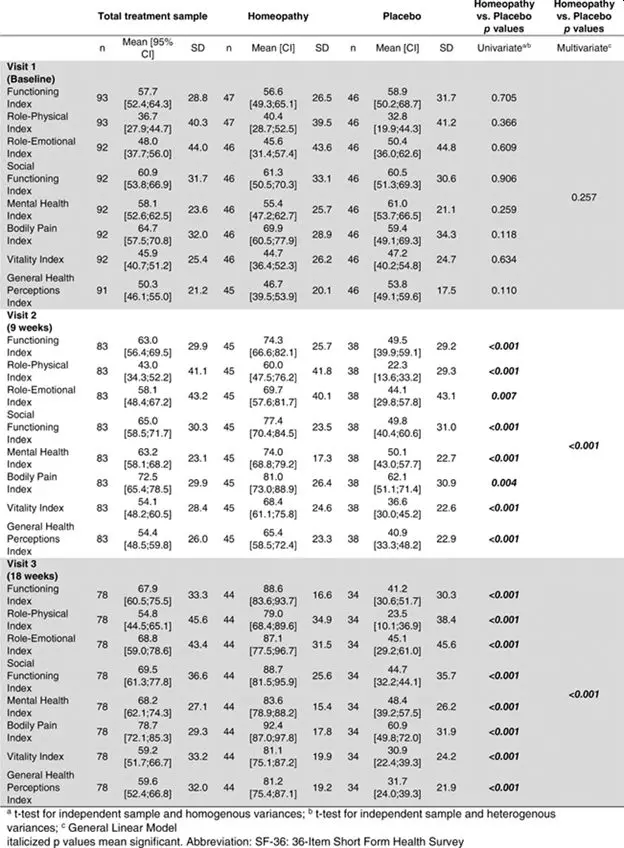
Table 4. 24‐month (730‐day) mortality
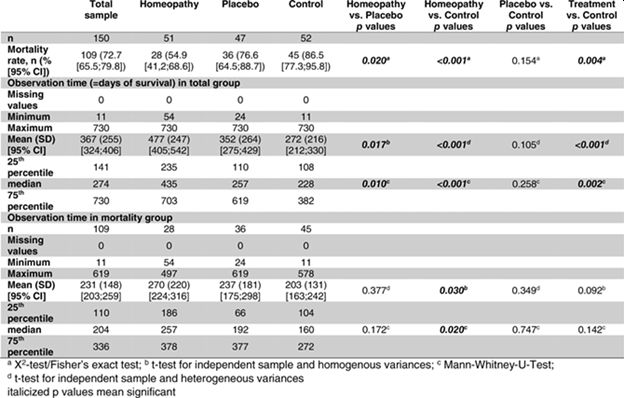
Table 5. Preparation of the Q‐potencies at the pharmacy and instructions for use of the Q‐potencies by the patient
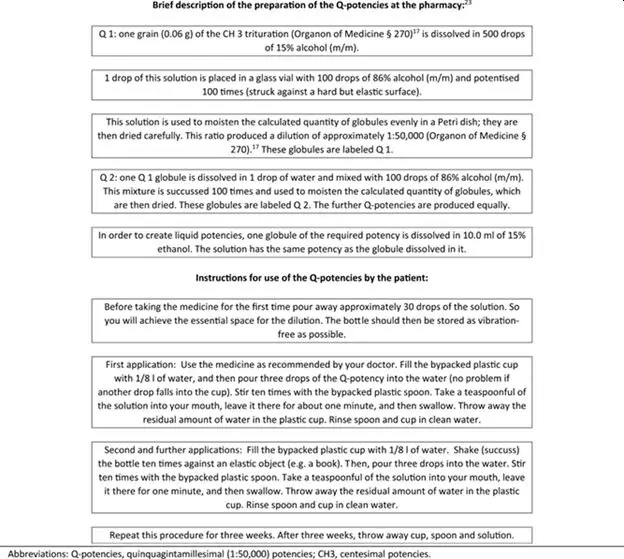
- Abbreviations: Q‐potencies, quinquagintamillesimal (1:50,000) potencies; CH3, centesimal potencies.
Table 6. Homeopathic remedies: Q‐potencies (prescribed in series)
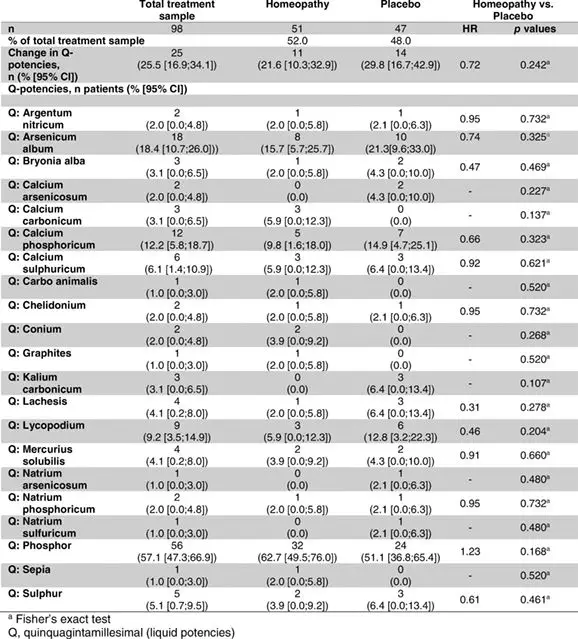
Table 7. Homeopathic remedies, LM‐, C‐, and D‐potencies
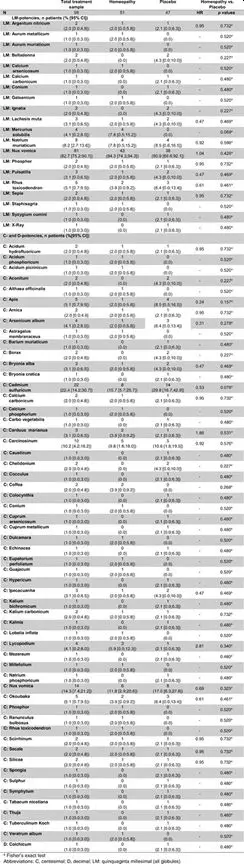
Table 8. Previous alternative treatments
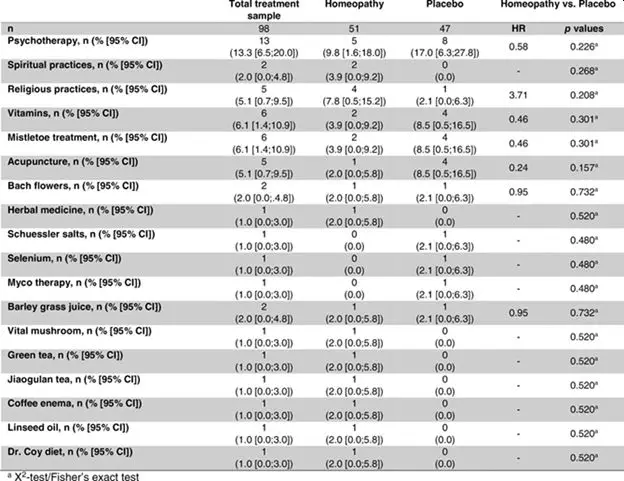
Table 9. Attitude toward homeopathy
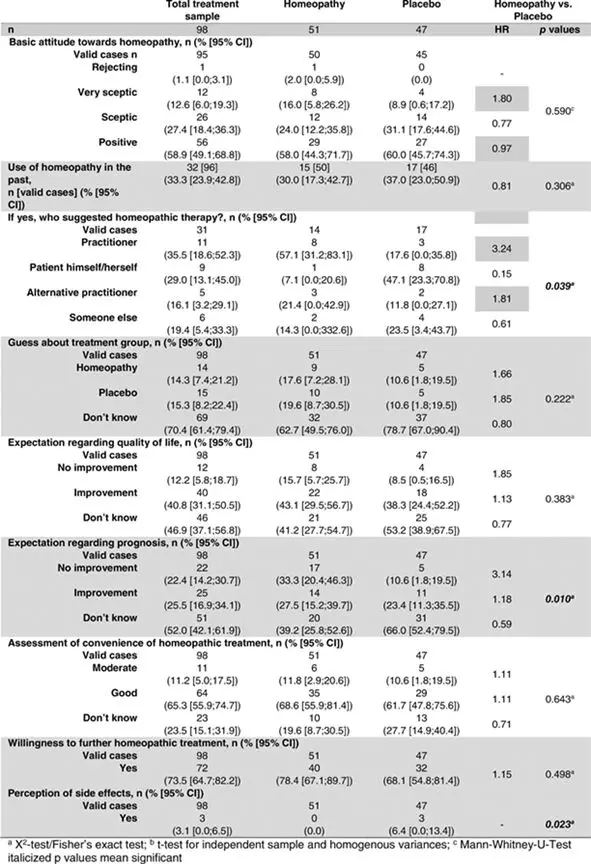
Table 10. Self‐assessment of subjective well‐being (Visual Analog Scalea)

Volume25, Issue12
December 2020
Pages e1930-e1955
© 2020 The Authors. The Oncologist published by Wiley Periodicals LLC on behalf of AlphaMed Press.
This is an open access article under the terms of the Creative Commons Attribution‐NonCommercial‐NoDerivs License, which permits use and distribution in any medium, provided the original work is properly cited, the use is non‐commercial and no modifications or adaptations are made.