Originally published in Complementary Therapies in Medicine Vol 24, Feb 2016- p111-117 Publisher -Elsevier -Feb 2016
Abstract
Objective
Homeopathic globules are commonly used in clinical practice, while research focuses on liquid potencies. Sequential dilution and succussion in their production process has been proposed to change the physico-chemical properties of the solvent(s). It has been reported that aqueous potencies of various starting materials showed significant differences in ultraviolet light transmission compared to controls and between different dilution levels. The aim of the present study was to repeat and expand these experiments to homeopathic globules.
Methods
Globules were specially produced for this study by Spagyros AG (Gümligen, Switzerland) from 6 starting materials (Aconitum napellus, Atropa belladonna, phosphorus, sulfur, Apis mellifica, quartz) and for 6 dilution levels (6x, 12x, 30c, 200c, 200CF (centesimal discontinuous fluxion), 10,000CF). Native globules and globules impregnated with solvents were used as controls. Globules were dissolved in ultrapure water, and absorbance in the ultraviolet range was measured. The average absorbance from 200 to 340 nm was calculated and corrected for differences between measurement days and instrumental drift.
Results
Statistically significant differences were found for A. napellus, sulfur, and A. mellifica when normalized average absorbance of the various dilution levels from the same starting material (including control and solvent control globules) was compared. Additionally, absorbance within dilution levels was compared among the various starting materials. Statistically significant differences were found among 30c, 200c and 200CF dilutions.
Conclusion
This study has expanded previous findings from aqueous potencies to globules and may indicate that characteristics of aqueous high dilutions may be preserved and detectable in dissolved globules.
Keywords
- UV spectroscopy;
- High dilutions;
- Globules;
- Homeopathy;
- Anthroposophically extended medicine;
- Complementary medicine
1. Introduction
Highly diluted remedies are applied in homeopathy and anthroposophically extended medicine. Several modes of action and models have been discussed on how the production of these aqueous solutions by sequential dilution and succussion, also termed potentization, might feature their structural properties.1 It has been proposed that a network of hydrogen bonds develops around nonpolar solutes that remains with successive dilution, even when the molecules of the starting material have disappeared,2 and 3 thus giving the water a more organized state.4 Additionally, nanobubbles have been discussed to contribute to these supramolecular structures,5 which are assumed to disappear upon heating.4
Differences in the structure of the water in these homeopathic preparations may be reflected in small but measurable changes in physico-chemical properties. Accordingly, methods such as ultraviolet (UV) spectroscopy,2, 6, 7, 8, 9, 10, 11 and 12 nuclear magnetic resonance spectroscopy,4, 13, 14, 15, 16 and 17 calorimetry18 or thermoluminescence19 and 20 have been employed to investigate possible differences between homeopathic preparations and respective controls.
It has been found that dilution and succussion can lead to the introduction of contaminants, e.g., trace elements such as Si, Li, Na, Mg.9 and 21 This can be avoided when dilutions are prepared very carefully (e.g., by washing all flasks and pipettes with the same solution used to prepare the dilutions) or under clean room conditions.16 and 22 This is crucial for experiments testing for differences between high dilutions and controls. Our group reported that high and ultra-high dilutions of various starting materials showed significant differences in UV light transmission to controls and between different dilution levels, all the more so when these dilutions were produced under controlled conditions.10, 11 and 12
In clinical practice, globules of potentized starting materials are commonly applied besides aqueous dilutions. In a preliminary study, we found differences in UV absorbance between verum and placebo globules of Aconitum napellus 30c or calcium carbonate/quercus e cortice 6x dissolved in water. 23 These globules had been produced for clinical trials and not for laboratory experiments. It was unclear whether the differences in UV absorbance originated from specific characteristics of the starting materials, from differences in the production of verum and placebo globules, and/or other unknown interference factors.
Therefore, the aim of the present study was to repeat and expand our previous experiments with globules produced under controlled conditions accurate for comparison of UV absorbance, i.e., sucrose globules and ethanol from the same batch were used to minimize the introduction of possible artifacts.
2. Methods
2.1. Globules
The globules were specially produced for this study by Spagyros AG (Gümligen, Switzerland) and differed only in the starting materials of the potentized dilutions. Sucrose globules and ethanol from the same batch were used to minimize the introduction of possible artifacts. The following starting materials were investigated: A. napellus, Atropa belladonna, phosphorus, sulfur, Apis mellifica, and quartz (2 plants, 2 non-metal elements, 1 animal, and 1 mineral).
Dilution was performed in a 43% ethanol/57% water mixture until the last step, for which 73% ethanol/27% water was used. Manual succussion was performed vertically with 30 strokes in each step.
200CF and 10,000CF dilutions were produced by discontinuous fluxion in a machine with water as dilution medium. In this single glass technique, the potentisation vessel is alternately filled with the dilution medium and emptied again in each step, while a defined amount of liquid remains in the vessel. The operating mode of the machine has been described.24 The final three steps were manually diluted in 30% ethanol/70% water, 43% ethanol/57% water and 73% ethanol/27% water and succussed with 30 strokes.
Two kinds of controls were generated in the experiments: native globules and globules impregnated with a succussed 73% ethanol/27% water mixture (solvent control globules).
The globules were produced in July 2012 and the measurements were carried out from September 2012 to March 2013. Vials were coded to display the starting material but the dilution level and the controls were blinded. Coding was unblinded only after completion of all measurements and preliminary description of the data by box plots. The vials were stored in aluminum boxes, each box containing the same dilution level of the 6 starting materials.
2.2. UV absorbance measurements
Globules were gently dissolved in ultrapure water (arium® pro VF, Sartorius Stedim AG, Goettingen, Germany) at 10 mg/ml in Fiolax® test tubes. Samples were prepared in quadruplicates, 19–22 h prior to the measurements to allow complete dissolution, wrapped individually in aluminum foil and stored in the dark at room temperature. Absorbance of the samples in the UV range (from 190 to 340 nm) was measured in a randomized order with a Shimadzu UV-1800 double beam spectrophotometer (Reinach, Switzerland) equipped with an auto sampler CETAC ASX-260 (Omaha, USA; as described previously in Ref.10). This wavelength range has previously proven to be better suited to detect differences than visible light or near infrared light.25 The spectrophotometer had been switched on 2 h prior to the measurements for sufficient warming up. Globules of each starting material and corresponding controls were freshly dissolved and measured on 5 independent days.
2.3. Data analysis
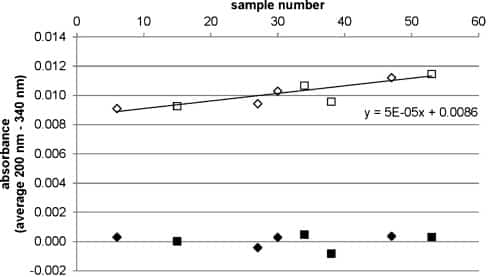
Due to the high absorbance of sucrose below 200 nm, the main component of the globules used, only absorbance values at 200 nm and higher were included in the analysis and one value for each nm was recorded. For each sample the mean of the absorbance values from 200 nm to 340 nm was calculated. Fig. 1 shows how this value was corrected for the differences between measurement days and the instrumental drift during the measurements by linear regression (intercept as measure for the differences between measurement days and slope as measure for the drift), yielding a normalized absorbance. Native and solvent control globules were thereby regarded as references, since they were independent of the starting materials.
Fig. 1.
Obtaining normalized absorbance by correcting for instrumental drift.
A typical example from one measurement day is shown. Diamonds represent native globules, and squares represent solvent control globules. Open symbols show the average absorbance from 200 nm to 340 nm. Samples were measured in a randomized order, and plotting absorbance versus sample number revealed an instrumental drift over time. The intercept of the linear regression (measure for the differences between measurement days) and the slope (measure for the instrumental drift) were used to correct the values, yielding the normalized absorbance (filled symbols). The same equation was then used to normalize the absorbance of the various dilution levels measured on the same day (not shown). A normalized absorbance can thus have positive or negative values.
Figure options
Since some data were not normally distributed, non-parametric statistical tests were applied. The normalized average absorbance from 200 to 340 nm was compared between various starting materials within equal dilution levels and between various dilution levels within equal starting materials using a Kruskal–Wallis test. Within equal starting materials, each dilution level was compared to the solvent control globules using a Mann–Whitney-U test. p ≤ 0.05 was considered statistically significant. Effect sizes (r) were calculated and results were reported according to Ref. 26 no adjustments for multiple comparisons were performed. While such adjustments (e.g., Bonferroni adjustments) decrease the likelihood for type I errors (mistakenly rejecting the null hypothesis), they increase the likelihood for type II errors, i.e., important differences are deemed non-significant. 27 and 28
Statistical analysis was performed with IBM SPSS Statistics 21.0 (Armonk, NY, USA).
3. Results
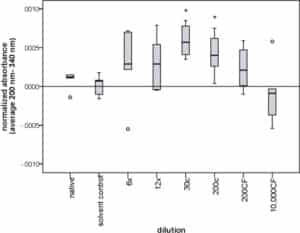
Normalized average absorbances of the various dilution levels and native and solvent control globules from the same starting material were compared by a Kruskal–Wallis test. Statistically significant differences were found for A. napellus, sulfur, and A. mellifica ( Table 1). The example of A. napellus is illustrated in Fig. 2.
Table 1.
Comparisona of normalized absorbanceb of globules of various dilution levelsc for each starting material.
| Including native and solvent control globules | Including solvent control globules | |||
| H (7) | p | H (6) | p | |
| Aconitum napellus | 14.31 | .046 | 11.68 | .070 |
| Atropa belladonna | 4.88 | .675 | 3.73 | .713 |
| Phosphorus | 2.52 | .925 | 1.48 | .961 |
| Sulfur | 15.24 | .033 | 9.52 | .147 |
| Apis mellifica | 14.71 | .040 | 13.59 | .035 |
| Quartz | 4.89 | .673 | 4.12 | .660 |
a
By Kruskal–Wallis test, statistically significant results (p ≤ 0.05) are displayed in bold, H = test statistic with degrees of freedom in parentheses.
b
Average between 200 nm and 340 nm.
c
6x, 12x, 30c, 200c, 200CF, 10,000CF, control(s) as indicated (n = 5 for each starting material and dilution level).
Table options
Fig. 2.
Boxplot showing normalized absorbance of control and Aconitum napellus globules of various dilution levels.
Globules were dissolved in water, their absorbance was measured and corrected for the daily variations and drift in the spectrophotometer, yielding a normalized average absorbance from 200 nm to 340 nm (n = 5 for each dilution level). Circles represent outliers (that lie more than one and a half box lengths above or below from the upper or lower quartile, respectively). *Indicate statistically significant differences (p ≤ 0.05, Mann–Whitney-U test) between the respective dilution level and the solvent control globules.
Native and solvent control globules showed a statistically significant difference from each other (p = 0.035, r = −0.272). Absorbance of dilution levels were then individually compared to the solvent control globules, which were considered a more accurate control than native globules that had not been impregnated with ethanol and water. Statistically significant differences were found between several of the ultra-high dilutions of A. napellus, sulfur, and A. mellifica and the respective solvent control globules ( Table 2).
Table 2.
Comparisona between normalized absorbanceb of solvent control globules and globules of various starting materials and dilution levelsc.
| Aconitum napellus | Atropa belladonna | Phosphorus | Sulfur | Apis mellifica | Quartz | ||
| Solvent control | Median | .00007 | .00003 | −.00016 | −.00001 | −.00038 | −.00003 |
| MAD | .00011 | .00016 | .00004 | .00008 | .00027 | .00015 | |
| 6x | Median | .00029 | .00012 | −.00011 | .00002 | −.00017 | .00005 |
| MAD | .00041 | .00031 | .00023 | .00023 | .00013 | .00003 | |
| p | .117 | .465 | .834 | .602 | .175 | .917 | |
| r | −.496 | −.231 | −.066 | −.165 | −.429 | −.033 | |
| 12x | Median | .00029 | −.00020 | −.00016 | −.00014 | .00039 | −.00011 |
| MAD | .00033 | .00028 | .00035 | .00007 | .00072 | .00033 | |
| p | .175 | .754 | .675 | .602 | .251 | .754 | |
| r | −.429 | −.099 | −.132 | −.165 | −.363 | −.099 | |
| 30c | Median | .00057 | −.00009 | −.00012 | −.00011 | .00020 | −.00006 |
| MAD | .00021 | .00044 | .00006 | .00007 | .00022 | .00011 | |
| p | .009 | .834 | .465 | .344 | .028 | .754 | |
| r | −.826 | −.066 | −.231 | −.299 | −.693 | −.099 | |
| 200c | Median | .00040 | −.00047 | −.00011 | −.00022 | .00036 | .00007 |
| MAD | .00022 | .00039 | .00018 | .00008 | .00017 | .00080 | |
| p | .047 | .249 | .834 | .465 | .028 | .917 | |
| r | −.627 | −.364 | −.066 | −.231 | −.693 | −.033 | |
| 200CF | Median | .00021 | −.00008 | −.00019 | .00022 | .00012 | .00002 |
| MAD | .00026 | .00006 | .00005 | .00006 | .00016 | .00006 | |
| p | .209 | .530 | .754 | .009 | .076 | .917 | |
| r | −.397 | −.199 | −.099 | −.826 | −.562 | −.033 | |
| 10,000CF | Median | −.00009 | −.00042 | −.00020 | −.00009 | .00032 | .00048 |
| MAD | .00028 | .00017 | .00011 | .00024 | .00010 | .00013 | |
| p | .465 | .249 | .675 | .917 | .009 | .117 | |
| r | −.231 | −.364 | −.132 | −.033 | −.826 | −.496 | |
a
By Mann–Whitney-U test, median normalized absorbance with median absolute deviation (MAD) is shown, statistically significant results (p ≤ 0.05) are displayed in bold, r = effect size.
b
Average between 200 nm and 340 nm.
c
n = 5 for each starting material and dilution level.
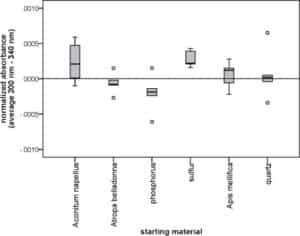
Finally, absorbance within dilution levels were compared between the various starting materials. Statistically significant differences were found between 30c, 200c and 200CF dilutions and between control globules (Table 3). The example of 200CF globules is illustrated in Fig. 3. No differences were found between 6x, 12x and 10,000CF dilutions and among solvent control globules.
Table 3.
Comparisona of normalized absorbanceb of globules of various starting materialsc for each dilution level.
| H (5) | p | |
| Native | 13.59 | .018 |
| Solvent control | 4.93 | .424 |
| 6x | 4.66 | .459 |
| 12x | 3.82 | .575 |
| 30c | 11.36 | .045 |
| 200c | 11.94 | .036 |
| 200CF | 11.95 | .036 |
| 10,000CF | 9.97 | .076 |
a
By Kruskal–Wallis test, statistically significant results (p ≤ 0.05) are displayed in bold, H = test statistic with degrees of freedom in parentheses. Pairwise comparisons of the 6 starting materials were not performed due to the high probability of a type I error when doing 15 tests.
b
Average between 200 nm and 340 nm.
c
Aconitum napellus, Atropa belladonna, phosphorus, sulfur, Apis mellifica, quartz (n = 5 for each starting material and dilution level).
Fig. 3.
Boxplot showing normalized absorbance of 200CF globules from various starting materials.
Globules were dissolved in water, their absorbance was measured and corrected for the daily variations and drift in the spectrophotometer, yielding a normalized average absorbance from 200 nm to 340 nm (n = 5 for each starting material). Circles represent outliers (that lie more than one and a half box lengths above or below from the upper or lower quartile, respectively). Comparison between the starting materials by Kruskal–Wallis test revealed a statistically significant difference (p = 0.036). Pairwise comparisons of the 6 starting materials were not performed due to the high probability of a type I error when executing 15 tests.
4. Discussion
To the best of our knowledge, this is a logical development of our previous study,23 the first report on differences in UV absorbance between globules prepared from various starting materials and various dilution levels. Apart from differences between control globules and globules impregnated with defined dilution levels, we found significant differences for 30c, 200c and 200CF from various starting materials, which indicates that the starting material is important in the potentization process, although at these high dilutions a presence of molecules of the starting material is not expected. This is in agreement with Rey, who was able to distinguish between lithium chloride 15c and sodium chloride 15c by thermoluminescence.19
Limitations of UV absorbance measurements of high dilutions in general include e.g., the detection limit of the spectrophotometer or differences in absolute absorbance between measurement days, and they have been discussed previously.12 In the present study, we corrected for the daily variations of the instrument as well as the drift during the measurements. Additionally, the globules were produced under controlled state-of-the-art conditions and in accordance with legal regulations by a pharmaceutical company. The results were generally consistent with former studies of aqueous dilutions, although by freshly weighing in and dissolving the globules for each measurement, an additional preparatory step was introduced, which may have added to the variations between measurements.
Each series of dilutions from each starting material was measured on 5 different days. Thus, we were limited in statistical testing, i.e., pairwise comparisons between starting materials were not performed and Bonferroni corrections were not applied. In future studies, one could focus on fewer starting materials and dilution levels and perform more measurements.
We used high dilutions to impregnate globules, which were then dissolved in water. In a future study it would be worthwhile to not only measure absorbance of the dissolved globules but also of the original high dilutions, i.e., the solutions with which the globules were impregnated, in order to investigate whether comparable differences are apparent in both. Additionally, a reproduction of these experiments by another and independent research group would be desirable.
In accordance with our preliminary results,23 we found a statistically significant difference between the normalized absorbance of solvent control and A. napellus 30c globules. The differences in uncorrected absorbance in the present study were less pronounced than in the previous study (data not shown). While the globules in the previous study had been produced for a clinical trial, the ones in the present study were specifically produced for our laboratory study, i.e., control globules and globules impregnated with high dilutions were from the same batch. Thus, if minor impurities existed they would be equally distributed in the globules of all groups and could not account for the differences in absorbance. Since more samples were analyzed, an auto sampler was used in the present study.
In previous studies, we compared UV absorbance of high dilutions of sulfur and quartz to succussed but not diluted water controls. For sulfur, we found statistically significant differences between the high dilutions (6c to 30c combined) and controls on 2 out of 5 measurement days.12 When each dilution level from 6c to 30c was tested individually against the controls, 16c and 29c had significantly different absorbance even after Bonferroni correction for multiple testing. In the present study, sulfur 200CF differed significantly from the solvent control globules. For quartz, we found no differences between any of the dilution levels and the solvent control. Similarly, in a previous experiment there was no difference between quartz and the respective control.11 Also in accordance with our previous studies,10 and 11 we found no significant differences between UV absorbance of decimal dilutions and controls.
In clinical studies, placebos are used as control interventions and are defined as substances with no pharmacological effect. In homeopathic in vitro, plant and animal studies several possible controls can be used: the same solvents with which the high dilutions were prepared (e.g., water or water/ethanol),29, 30 and 31 succussed but not diluted solvents,10 and 30 or diluted and succussed solvents can serve as controls.30, 32 and 33 Particularly, the succussion may lead to several effects such as ion leaching and gas exchange and in turn altering the properties of the dilutions. Therefore, pure, i.e., unsuccussed solvent is not regarded as adequate when used as single control.30 and 34 Diluted and succussed solvents also show effects different from the undiluted solvents.29 and 31
Two different controls were used in this study, native globules and globules impregnated with an ethanol/water mixture that had been succussed (solvent control globules). The normalized average absorbance of these controls differed between the two. Solvent control globules were considered as the more appropriate control and thus used for comparisons with the high dilutions (e.g., in Table 2).
Physico-chemical properties of globules have rarely been investigated. Aabel et al. compared T1 relaxation times of globules impregnated with Betula alba 30c and dissolved in water to control globules and found no differences between the two.35 Sukul et al. prepared potassium bromide pellets from several high dilutions and solvents as replacement for sucrose globules and reported significant differences in Fourier transform infrared spectra between the high dilutions and between the high dilutions and the solvents.36 Detecting delayed luminescence, Lenger et al. showed differences between homeopathic argentum metallicum globules and controls37 and found that the stability of ethanolic argentum metallicum declined within a month, while globules were stable over 1 year.38
While we found significant differences between potencies of 3 starting materials and controls in this study, 3 other starting materials did not produce such differences. This might not seem to be in accordance with the hypothesis of a change in the solvent’s structure; however, we would not expect the same resulting structures for all starting materials. Therefore, it may well be that some changes are not detectable with our measurement system.
Unexpectedly, we found differences in absorbance between native globules used as controls for the various starting materials, while no difference was detected between solvent control globules (Table 3). This can presently not be explained and may be a result obtained by chance. It may also raise questions about the possible transfer of a therapeutically active ingredient from high dilutions to a control, a phenomenon that has been observed but not yet investigated systematically.
For example, Endler et al.39 observed an effect of thyroxine 30c on the development of frogs, even when the animals were in the water and had no direct contact to the thyroxine dilution. Variations in controls have been observed before in one of our previous studies with liquid high dilutions.12 However, the variations in the present and previous studies occurred irregularly and did not indicate a systematic influence of high dilutions on controls.
As described above, several modes of action were proposed that lead to a change in water structure during potentization, but it is yet unclear how a changed structure may be preserved on globules. As an explanation the importance of lattice defects in lactose monohydrate or small pores on sucrose globules to transfer therapeutically active ingredients from high dilutions has been pointed out.40 It has also been suggested that in the solid phase, nanostructures may retain their properties without dissipating energy from the environment and could return to their preceding state when water became available,41 but precise models of this process are lacking. Therefore, it is important to gain insights in physical properties of globules as these may facilitate to developing models to better understanding their effects and modes of action. Finally, one of the next achievements will be to transfer findings from such physico-chemical measurements on effects observed in plants, animals and humans.
5. Conclusions
Globules prepared from high dilutions of A. napellus, sulfur and A. mellifica showed significantly different UV absorbance compared to solvent control globules when dissolved in water. This study has expanded our findings from aqueous high dilutions to globules, and it suggests that characteristics of aqueous high dilutions may be preserved and detectable in dissolved globules.
Conflict of interest
None.
Funding
The study was financially supported by Spagyros AG (Gümligen, Switzerland) and the Swiss Medical Society for Homeopathy smsh. The sponsors had no influence whatsoever on the design, collection, analysis and interpretation of the data, or in the writing of the manuscript or in the decision to submit the manuscript for publication.
Figures and tables
Table 1
Table 2
Table 3
Acknowledgements
Globules were specially produced and provided by Spagyros AG (Gümligen, Switzerland). We gratefully acknowledge the sponsors of the study.
We also thank the Department of Clinical Research, University of Bern, for providing laboratory space and Stephan Baumgartner for helpful discussion.
References
Corresponding author at: Institute of Complementary Medicine, University of Bern, Fabrikstrasse 8, 3012 Bern, Switzerland. Fax: +41 31 6324262.
Copyright © 2016 The Authors. Published by Elsevier Ltd.
1. Chaplin MF. The memory of water: an overview. Homeopathy.
2007;96:143–150.
2. Sukul NC, De A, Dutta R, Sukul A, Sinhababu SP. Nux vomica 30 prepared with
and without succession shows antialcoholic effect on toads and distinctive
molecular association. Br Homeopath J. 2001;90:79–85.
3. Rey L. Thermoluminescence of ultra-high dilutions of lithium chloride and
sodium chloride. Phys A. 2003;323:67–74.
4. Demangeat JL. Nanosized solvent superstructures in ultramolecular aqueous
dilutions: twenty years’ research using water proton NMR relaxation.
Homeopathy. 2013;102:87–105.
5. Roy R, Tiller WA, Bell I, Hoover MR. The structure of liquid water; novel
insights from materials research; potential relevance to homeopathy. Mater
Res Innov. 2005;9:577–608.
6. Ludwig W. Physikalische Grundlagenforschung in Bezug auf
Informationsspeicherung in lebenden Systemen und homöopathischen
Medikamenten. Erfahrungsheilkunde. 1991;4:293–295.
7. Korenbaum VI, Chernysheva TN, Apukhtina TP, Sovetnikova LN. Absorption
spectra of electronic-homoeopathic copies of homoeopathic nosodes and
placebo have essential differences. Forsch Komplementmed. 2006;13:294–297.
8. Rao ML, Roy R, Bell IR, Hoover R. The defining role of structure (including
epitaxy) in the plausibility of homeopathy. Homeopathy. 2007;96:175–182.
9. Zacharias CR. Contaminants in commercial homoeopathic medicines: a
spectroscopic determination. Br Homeopath J. 1995;84:71–74.
10. Marschollek B, Nelle M, Wolf M, Baumgartner S, Heusser P, Wolf U. Effects of
exposure to physical factors on homeopathic preparations as determined by
ultraviolet light spectroscopy. ScientificWorldJournal. 2010;10:49–61.
11. Wolf U, Wolf M, Heusser P, Thurneysen A, Baumgartner S. Homeopathic
preparations of quartz, sulfur and copper sulfate assessed by
UV-spectroscopy. Evid Based Complement Altern Med. 2011;2011, 692798.
12. Klein SD, Sandig A, Baumgartner S, Wolf U. Differences in median ultraviolet
light transmissions of serial homeopathic dilutions of copper sulfate,
Hypericum perforatum, and sulfur. Evid Based Complement Altern Med.
2013;2013, 370609.
13. Demangeat JL, Gries P, Poitevin B, et al. Low-field NMR water proton
longitudinal relaxation in ultrahighly diluted aqueous solutions of
silica-lactose prepared in glass material for pharmaceutical use. Appl Magn
Reson. 2004;26:465–481.
14. Aabel A, Fossheim S, Rise F. Nuclear magnetic resonance (NMR) studies of
homeopathic solutions. Br Homeopath J. 2001;90:14–20.
15. Anick DJ. High sensitivity 1H-NMR spectroscopy of homeopathic remedies
made in water. BMC Complement Altern Med. 2004;4:15.
16. Baumgartner S, Wolf M, Skrabal P, et al. High-field 1H T1 and T2 NMR
relaxation time measurements of H2O in homeopathic preparations of quartz,
sulfur, and copper sulfate. Naturwissenschaften. 2009;96:1079–1089.
17. Demangeat JL. NMR water proton relaxation in unheated and heated
ultrahigh aqueous dilutions of histamine: evidence for an air-dependent
supramolecular organization of water. J Mol Liq. 2009;144:32–39.
18. Elia V, Niccoli M. New physico-chemical properties of water induced by
mechanical treatments: a calorimetric study at 25 ◦C. J Therm Anal Calorim.
2000;61:527–537.
19. Rey L. Thermoluminescence of ultra-high dilutions of lithium chloride and
sodium chloride. Phys A. 2003;323:67–74.
20. van Wijk R, Bosman S, van Wijk EPA. Thermoluminescence in ultra-high
dilution research. J Altern Complement Med. 2006;12:437–443.
21. Witt CM, Lüdtke R, Weisshuhn TE, Quint P, Willich SN. The role of trace
elements in homeopathic preparations and the influence of container
material, storage duration, and potentisation. Forsch Komplementmed.
2006;13:15–21.
22. Zacharias CR. Implications of contaminants to scientific research in
homoeopathy. Br Homeopath J. 1995;84:3–5.
23. Klein SD, Wolf U. Investigating homeopathic verum and placebo globules
with ultraviolet spectroscopy. Forsch Komplementmed. 2013;20:295–297.
24. Ryffel J. Spagyros-Hochpotenzen. Spagynews. 2006;5:1–3 http://spagyros.ch/
media/docs/infocenter/homoeopathie/downloads/Spagynews 2006
Hochpotenzen Spagyros.pdf.
25. Klein S, Sandig A, Baumgartner S, Wolf U. Investigating homeopathic
preparations with light spectroscopy. Int J High Dilution Res. 2012;11:117.
26. Field A. Discovering Statistics Using SPSS. 3rd edition Los Angeles, London, New
Delhi, Singapore, Washington DC: Sage Publications; 2009.
27. Rothman KJ. No adjustments are needed for multiple comparisons.
Epidemiology. 1990;1:43–46.
28. Perneger TV. What’s wrong with Bonferroni adjustments. BMJ.
1998;316:1236–1238.
29. Brizzi M, Elia V, Trebbi G, Nani D, Peruzzi M, Betti L. The efficacy of
ultramolecular aqueous dilutions on a wheat germination model as a function
of heat and aging-time. Evid Based Complement Altern Med. 2011;2011,
696298.
S.D. Klein, U. Wolf / Complementary Therapies in Medicine 24 (2016) 111–117 117
30. Jäger T, Scherr C, Shah D, et al. Use of homeopathic preparations in
experimental studies with abiotically stressed plants. Homeopathy.
2011;100:275–287.
31. Hostanska K, Rostock M, Melzer J, Baumgartner S, Saller R. A homeopathic
remedy from arnica, marigold, St. John’s wort and comfrey accelerates in vitro
wound scratch closure of NIH 3T3 fibroblasts. BMC Complement Altern Med.
2012;12:100.
32. Welles SU, Endler PC, Scherer-Pongratz W, et al. Pretreatment with thyroxin
10–8 and the effect of homeopathically prepared thyroxin 10–30 on highland
frogs—a multi-researcher study. Forsch Komplementmed. 2007;14:353–357.
33. Baumgartner S, Doesburg P, Scherr C, Andersen JO. Development of a
biocrystallisation assay for examining effects of homeopathic preparations
using cress seedlings. Evid Based Complement Altern Med. 2012;2012, 125945.
34. Baumgartner S, Heusser P, Thurneysen A. Methodological standards and
problems in preclinical homoeopathic potency research. Forsch
Komplementmed. 1998;5:27–32.
35. Aabel S, Fossheim S, Rise F. Nuclear magnetic resonance (NMR) studies of
homeopathic solutions. Br Homeopath J. 2001;90:14–20.
36. Sukul NC, Ghosh S, Sukul A, Sinhababu SP. Variation in Fourier transform
infrared spectra of some homeopathic potencies and their diluent media. J
Altern Complement Med. 2005;11:807–812.
37. Lenger K, Bajpai RP, Drexel M. Delayed luminescence of high homeopathic
potencies on sugar globuli. Homeopathy. 2008;97:134–140.
38. Lenger K, Bajpai RP, Spielmann M. Identification of unknown homeopathic
remedies by delayed luminescence. Cell Biochem Biophys. 2014;68:321–334.
39. Endler PC, Pongratz W, Smith CW, Schulte J. Non-molecular information
transfer from thyroxine to frogs with regard to homeopathic toxicology. Vet
Hum Toxicol. 1995;37:259–260.
40. Suess WG, Radau K. Mechanism of the transfer of the therapeutically active
ingredient (TAI) from a homeopathic liquid dilution to a solid substance. AHZ.
2005;250:58.
41. Elia V. Physico-chemical properties of perturbed water: facts and enigmas. Int
J High Dilution Res. 2012;11:110–112.




Dejar Mrs Sabine:
About of the paper.
Semi critical comment for “skeptical” physicist of Granada Unkversity, Spain:
http://elprofedefisica.naukas.com/2016/02/15/otra-prueba-de-las-bases-fisicas-de-la-homeopatia-o-no/
Debunked the above semi critical comment from enginner:
http://explicandoalexplicador.blogspot.com 2016/03/el-sacerdocio-de-la-ciencia-xxxix-el.html?m=1
What is tour opinion?
Sorry the link is this:
http://explicandoalexplicador.blogspot.com/2016/03/el-sacerdocio-de-la-ciencia-xxxix-el.html?m=1