This is our 9th paper to explain Homeopathy. Herein we have shown that not only pure water is able to retain electrical energy, but also both ionic (salt solution) & non-ionic solutions (sugar solution) are able to retain electrical energy. The key factor of our research is arrangement and rearrangement of dipoles of water molecules 1, which leads to different energy content of a solution2, ionic or non-ionic.
Before exploring retention of electrical energy by different solutions, let us first note the variation in conduction of current by the referred solutions.
Now to confirm the variation we performed the following experiments:
Experiment 1:
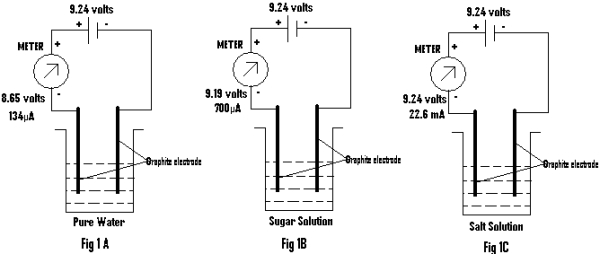
(a) A beaker containing pure water is used wherein two Graphite (Gr) electrodes are dipped. The –ve terminal of a battery having emf of 9.24 volts is connected to one of the Gr. electrodes. The other terminal of the battery is connected to the other Gr through a meter (as shown in Fig.1). The emf and current is measured and recorded in table 1 .
(b) Now the meter is replaced by an LED (light emitting diode) and the same is found to glow considerably (as shown in Fig.2). The degree of brightness is recorded in table 2.
This experiment is repeated by replacing pure water with sugar solution and then salt solution (Sodium Chloride solution) and all observations are recorded at the appropriate place in the table1 & table 2.
Table1:
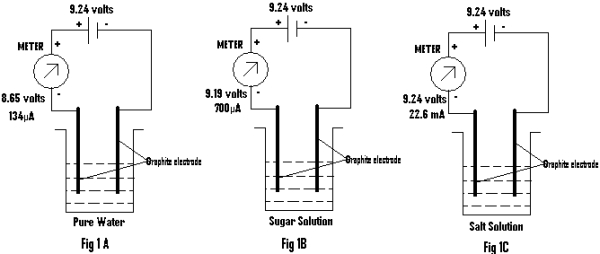
Fig. 1
Table2:
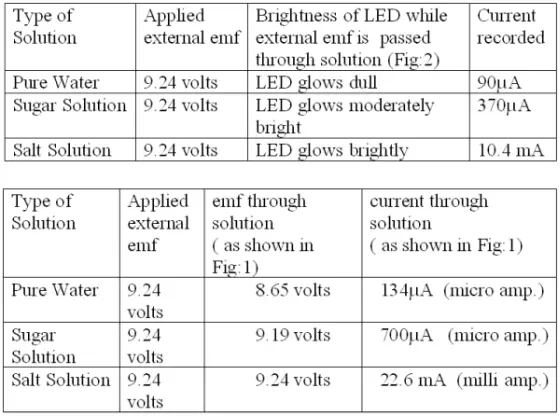
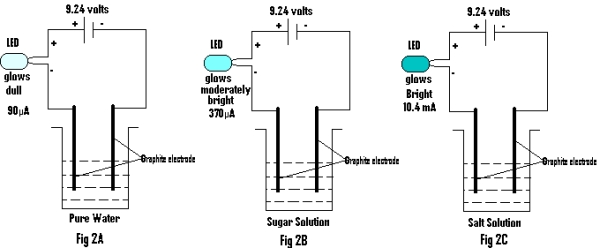
Fig.2
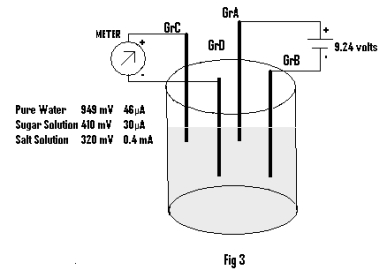
Experiment 2: This experiment is performed by taking pure water in a beaker and placing four Graphite electrodes ( GrA, GrB, GrC,& GrD) such that a pair of electrodes remains just opposite to the other ( as shown in Fig.3). Here GrA & GrB is placed just opposite of GrC & GrD. Now the emf and current is measured at a pair of Gr electrodes (GrC & GrD ) by connecting simultaneously two terminals of a battery (emf=9.24 volts) to the other pair of Gr electrodes (GrA & GrB). The readings of emf & current at the graphites GrC & GrD owing to application of external emf at GrA & GrB is recorded in Table 3.
The same experiment is repeated by replacing Pure Water with a Sugar Solution and Salt solution and observations are recorded in Table 3.
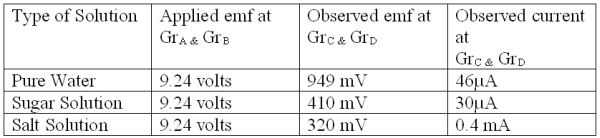
Discussion: From the above two experiments it is found that though the two solutions and pure water conduct electricity, yet the value of the emf and current differs considerably. The conduction of current in ionic solutions or salt solutions may be attributed to the presence of extra ions, Na+ & Cl – in addition to ions present in the water itself (i.e;H3O+ & OH– ions). But the same theory fails to explain conduction in sugar solutions, as being non-ionic they do not render any extra ions in water. As such the values of emf and current as obtained for a sugar solution, should have been same or nearly same as that of pure water. The brightness of the glowing LED should have also been same or nearly same for the sugar solution and pure water. But this is not the case. Rather the emf & current values for the sugar solution is quite higher than pure water (as shown in Table 1). The LED also glows brighter using the sugar solution than in pure water (as shown in Table 2).
As the ionic theory fails to explain such results, we are once again of the opinion that it is change in orientation of dipoles of water molecules (as a strain is always produced whenever any substance is introduced in water 1,2 ) that is responsible for such results.
Now to see the retention of electrical energy by Pure water and other solutions (ionic or non-ionic,) the following experiment is performed:
Experiment 3: A beaker containing pure water is taken and two graphite (Gr) electrodes (GrA & GrB ) are dipped in it. The emf of the two Gr electrodes is measured and recorded in table :4 as initial emf. Then an external source of emf (about 9 volts) is connected to the graphite pair and current is allowed to pass for 3 minutes. After passing of current is stopped, the final readings for the graphite pair (GrA & GrB ) is taken at intervals of 1(one) minute and recorded in Table :4.
This experiment is repeated by replacing pure water with sugar solution & salt solution, and readings are recorded in Table : 5 & Table : 6 respectively.
Table: 4
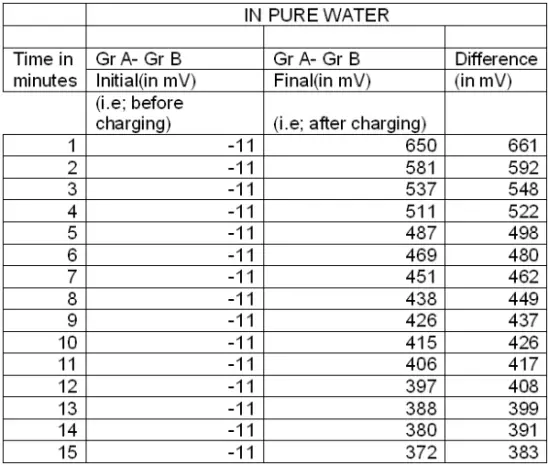
Table:5
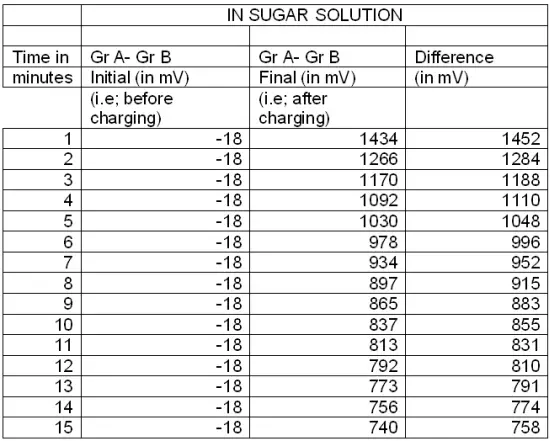
Table:6
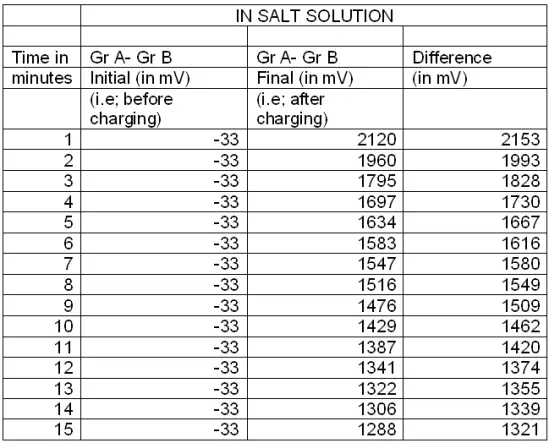
Thereafter a graph is plotted with emf (in mV) vs time (in minutes) for pure water, sugar solution and salt solution.
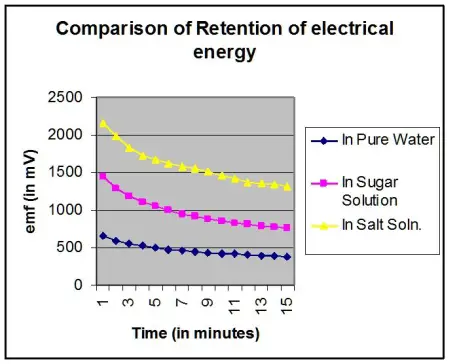
It is found that the rate of decrement of electrical energy with respect to time (i.e –dE/dT) remains almost the same for all the three curves. While the rate of decrement is higher initially, but remarkably lower later.
Discussion: Since retention of electrical energy in pure water cannot be explained by ionization of water3 , it (ionization) cannot explain the retention of electrical energy by Ionic & Non-ionic solutions also. Therefore retention of electrical energy, hence generation of emf4 thereof, may be attributed to the presence of either ions or the orientation of water molecules. Had it been due to the presence of extra ions (i.e. ions other than H3O+ & OH– that already exist in water), there is the possibility of such extra ions in salt solution. But as sugar does not ionize in water, there is no possibility of extra ions in sugar solution. Hence, sugar solution should have behaved like pure water and the curve obtained for pure water and sugar solution should have been the same, or nearly the same. But this is not so. There is great difference between the two.
As it is already stated in our earlier works, that water retains electrical energy by changing its orientation 2 , here also it is confirmed again that not only pure water but also ionic or non-ionic solutions also retain electrical energy by rearrangement of orientations of dipoles of water molecules.
As previously stated, pure water undergoes strain whenever any substance is introduced in it, and to balance the external strain it arranges to a specific orientation. But when the external strain is removed, the water molecules try to get back to their initial orientation. But in a solution of salt (ionic soln) the presence of extra ions stabilizes the orientations due to its charge and restricts the water from getting back to its initial orientation. In spite of the fact that sugar solution is non-ionic, a similar condition prevails therein, due to the fact that being a polar molecule it (sugar) stabilizes the orientation and restricts water molecules from getting back to their initial orientation. So, the curves obtained for retention of electrical energy by pure water, ionic solution and non-ionic solutions differ.
Inference: The electrical energy is not only retained, but also conveyed by the change in orientation of dipoles of water molecules in pure water or solutions (ionic or non-ionic).
Reference:
[1] The Homeopathic Dilution-A New Explanation, Ruhul Amin, Biplab Chakraborty & Farook Rahaman (Simillimum,vol.XXI,Summer Fall/2008. www.hanp.net
[2] The electrical energy of substances –the secret of homeopathy, Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXII, Summer Fall/ 2009. www.hanp.net)
[3]. Water the Magic Liquid: Retaining Electrical Energy, Ruhul Amin & Biplab Chakraborty (Published in Homeopathy for Everyone, April 2012 hpathy.com)
[4]. Generation of electrode Potential of an electrode –an explanation , Ruhul Amin & Biplab Chakraborty (Simillimum,vol.XXIII.2010, www.hanp.net)





Dr. Ruhul Amin MD its a an article which needs some explanations! First of all what is your definition of pure water? Do you mean by this distilled water, deionised water, filtered water or purified water? On the basis of your clarifications then I will see if there are further issues with this paper.
At this moment your paper did not define this very crucial aspect on which your research is relying so I would not further comment on the contents of this paper!
Dear Saqib Rashid,
Here pure water means “Distilled Water”.
The inference drawn from the experiments is that ” the electrical energy is not only retained, but also conveyed by the change in orientation of dipoles of water molecules in pure water or solutions (ionic or non-ionic).” Since here we are concerned with Homeopathy, it would have been helpful if the authors had explained how this finding is related to Homeopathy/Homeopathic medicines.
Dear Sir,
In this regard kindly refer to our earlier works specially the following
(1). The Homeopathic Dilution-A New Explanation, (Simillimum,vol.XXI,Summer Fall/2008. http://www.hanp.net)
(2) The electrical energy of substances –the secret of homeopathy,(Simillimum,vol.XXII, Summer Fall/ 2009. http://www.hanp.net) .